Abstract
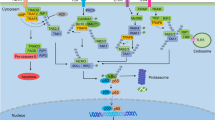
NF-κB, a family of related transcription factors, has been a focus of intense scientific research during the past decade. Multiple stimuli, both extracellular and intracellular, lead to its activation. The NF-κB pathway regulates expression of a diverse array of genes involved in different biological processes. Various pathological states are characterized by the dysregulated NF-κB pathway. Recently, NF-κB activation has been connected with multiple aspects of oncogenesis and serves as an important mechanism to regulate cell survival in response to chemotherapy by activating different genes that inhibit apoptosis. Several methods of inhibiting NF-κB activation, such as antisense oligonucleotides, proteosome inhibitors and RNA interference (RNAi) are currently under investigation. RNAi represents a powerful tool to better define the role of specific genes in different signal transduction pathways and has recently been used to define the function of genes that regulate the NF-κB pathway. This review discusses the emerging role of RNAi to dissect the function of regulatory factors in the NF-κB pathway and its potential use as a targeted therapy.
Similar content being viewed by others
Article PDF
References
Perkins ND . The Rel/NF-kappa B family: friend and foe. Trends Biochem Sci 2000; 25: 434–40.
Baldwin AS . Control of oncogenesis and cancer therapy resistance by the transcription factor NF-kappaB. J Clin Invest 2001; 107: 241–6.
Baeuerle PA, Baltimore D . κB: Ten years after. Cell 1996, 87: 13–20.
Ghosh S, May MJ, Kopp EB . κB and Rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol 1998; 16: 225–60.
Ghosh S, Karin M . Missing pieces in the NF-kappaB puzzle. Cell 2002; 109: S81–S96.
Verma IM, Stevenson JK, Schwartz EM, Van Antwerp D, Miyamoto S . κB/IκB family: intimate tales of association and dissociation. Genes Dev 1995; 9: 2723–35.
DiDonato JA, Hayakawa M, Rothwarf DM, Zandi E, Karin M . κB kinase that activates the transcription factor NF-κB. Nature 1997; 388: 548–54.
Mercurio F, Zhu H, Murray BW, Shevchenko A, Bennett BL, Li J, et al. κB kinases essential for NF-κB activation. Science 1997; 278: 860–6.
Li Y, Kang J, Friedman J, Tarassishin L, Ye J, Kovalenko A, et al. κB activity and as a target of an adenovirus inhibitor of tumor necrosis factor α-induced apoptosis. Proc Natl Acad Sci USA 1999; 96: 1042–7.
Stancovski I, Baltimore D . NF-kappaB activation: the I kappaB kinase revealed? Cell 1997; 91: 299–303.
Baud V, Liu ZG, Bennett B, Suzuki N, Xia Y, Karin M . Signaling by proinflammatory cytokines: oligomerization of TRAF2 and TRAF6 is sufficient for JNK and IKK activation and target gene induction via an amino-terminal effector domain. Genes Dev 1999; 13: 1297–308.
Sun L, Chen ZJ . The novel functions of ubiquitination in signaling. Curr Opin Cell Biol 2004; 16: 339–40.
Wajant H, Scheurich P . Analogies between Drosophila and mammalian TRAF pathways. Prog Mol Subcell Biol 2004; 34: 47–72.
Lee FS, Peters RT, Dang LC, Maniatis T . MEKK1 activates both IkappaB kinase alpha and IkappaB kinase beta. Proc Natl Acad Sci USA 1998; 95: 9319–24.
Zhao Q, Lee FS . Mitogen-activated protein kinase/ERK kinase kinases 2 and 3 activate nuclear factor-kappaB through IkappaB kinase-alpha and IkappaB kinase-beta. J Biol Chem 1999; 274: 8355–8.
Ninomiya-Tsuji J, Kishimoto K, Hiyama A, Inoue J, Cao Z, Matsumoto K . The kinase TAK1 can activate the NIK-I kappaB as well as the MAP kinase cascade in the IL-1 signalling pathway. Nature 1999; 398: 252–6.
Woronicz JD, Gao X, Cao Z, Rothe M, Goeddel DV . κB kinase-β: NF-κB activation and complex formation with IκB kinase-α and NIK. Science 1997; 278: 866–9.
Bouwmeester T, Bauch A, Ruffner H, Angrand PO, Bergamini G, Croughton K, et al. A physical and functional map of the human TNF-alpha/NF-kappa B signal transduction pathway. Nat Cell Biol 2004; 6: 97–105.
Malinin NL, Boldin MP, Kovalenko AV, Wallach D . κB induction by TNF, CD95 and IL-1. Nature 1997; 385: 540–8.
Song HY, Regnier CH, Kirschning CJ, Goeddel DV, Rothe M . Tumor necrosis factor (TNF)-mediated kinase cascades: bifurcation of nuclear factor-kappaB and c-jun N-terminal kinase (JNK/SAPK) pathways at TNF receptor-associated factor 2. Proc Natl Acad Sci USA 1997; 94: 9792–6.
Yang J, Lin Y, Guo Z, Cheng J, Huang J, Deng L, et al. The essential role of MEKK3 in TNF-induced NF-kappaB activation. Nat Immunol 2001; 2: 620–4.
Huang Q, Yang J, Lin Y, Walker C, Cheng J, Liu ZG, et al. Differential regulation of interleukin 1 receptor and Toll-like receptor signaling by MEKK3. Nat Immunol 2004; 5: 98–103.
Cheng G, Cleary AM, Ye ZS, Hong DI, Lederman S, Baltimore D . Involvement of CRAF1, a relative of TRAF, in CD40 signaling. Science 1995; 267: 1494–8.
Rothe M, Sarma V, Dixit VM, Goeddel DV . κB by TNF receptor 2 and CD40. Science 1995; 269: 1424–7.
Pickart CM . Mechanisms underlying ubiquitination. Annu Rev Biochem 2001; 70: 503–33.
Takeuchi M, Rothe M, Goeddel DV . Anatomy of TRAF2. Distinct domains for nuclear factor-kappaB activation and association with tumor necrosis factor signaling proteins. J Biol Chem 1996; 271: 19935–42.
Dadgostar H, Cheng G . An intact zinc ring finger is required for tumor necrosis factor receptor-associated factor-mediated nuclear factor-kappaB activation but is dispensable for c-Jun N-terminal kinase signaling. J Biol Chem 1998; 273: 24775–80.
Hu Y, Baud V, Delhase M, Zhang P, Deerinck T, Ellisman M, et al. A subunit of IκB kinase. Science 1999; 284: 316–20.
Deng L, Wang C, Spencer E, Yang L, Braun A, You J, et al. Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 2000; 103: 351–61.
Wajant H, Henkler F, Scheurich P . The TNF-receptor-associated factor family: scaffold molecules for cytokine receptors, kinases and their regulators. Cell Signal 2001; 13: 389–400.
Wilkinson KD . Signal transduction: aspirin, ubiquitin and cancer. Nature 2003; 424: 738–9.
Shibuya H, Yamaguchi K, Shirakabe K, Tonegawa A, Gotoh Y, Ueno N, et al. TAB1: an activator of the TAK1 MAPKKK in TGF-beta signal transduction. Science 1996; 272: 1179–82.
Wang C, Deng L, Hong M, Akkaraju GR, Inoue J, Chen ZJ . TAK1 is a ubiquitin-dependent kinase of MKK and IKK. Nature 2001; 412: 346–51.
Takaesu G, Kishida S, Hiyama A, Yamaguchi K, Shibuya H, Irie K, et al. TAB2, a novel adaptor protein, mediates activation of TAK1 MAPKKK by linking TAK1 to TRAF6 in the IL-1 signal transduction pathway. Mol Cell 2000; 5: 649–58.
Shirakabe K, Yamaguchi K, Shibuya H, Irie K, Matsuda S, Moriguchi T, et al. TAK1 mediates the ceramide signaling to stress-activated protein kinase/c-Jun N-terminal kinase. J Biol Chem 1997; 272: 8141–4.
Brummelkamp TR, Nijman SM, Dirac AM, Bernards R . Loss of the cylindromatosis tumour suppressor inhibits apoptosis by activating NF-kappaB. Nature 2003; 424: 797–801.
Trompouki E, Hatzivassiliou E, Tsichritzis T, Farmer H, Ashworth A, Mosialos G . CYLD is a deubiquitinating enzyme that negatively regulates NF-kappaB activation by TNFR family members. Nature 2003; 424: 793–6.
Bignell GR, Warren W, Seal S, Takahashi M, Rapley E, Barfoot R, et al. Identification of the familial cylindromatosis tumour-suppressor gene. Nat Genet 2000; 25: 160–5.
Dave RS, Pomerantz RJ . RNA interference: on the road to an alternate therapeutic strategy. Rev Med Virol 2003; 13: 373–85.
Schmidt CW . Therapeutic interference: small RNA molecules act as blockers of disease metabolism. Modern Drug Disc 2003: 37–42.
Arenz C, Schepers U . RNA interference: from an ancient mechanism to a state of the art therapeutic application? Naturwissenschaften 2003; 90: 345–59.
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC . Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998; 391: 806–11.
Hohmann HP, Brockhaus M, Baeuerle PA, Remy R, Kolbeck R, van Loon AP . Expression of the types A and B tumor necrosis factor (TNF) receptors is independently regulated, and both receptors mediate activation of the transcription factor NF-kappa B. TNF alpha is not needed for induction of a biological effect via TNF receptors. J Biol Chem 1990; 265: 22409–17.
Boutla A, Delidakis C, Livadaras I, Tsagris M, Tabler M . Short 5′-phosphorylated double-stranded RNAs induce RNA interference in Drosophila. Curr Biol 2001; 11: 1776–80.
Brummelkamp TR, Bernards R, Agami R . A system for stable expression of short interfering RNAs in mammalian cells. Science 2002; 296: 550–3.
Waterhouse PM, Wang MB, Lough T . Gene silencing as an adaptive defence against viruses. Nature 2001; 411: 834–42.
Pham JW, Pellino JL, Lee YS, Carthew RW, Sontheimer EJ . A Dicer-2-dependent 80s complex cleaves targeted mRNAs during RNAi in Drosophila. Cell 2004; 117: 83–94.
Medema RH . Optimizing RNA interference for application in mammalian cells. Biochem J 2004; 380: 593–603.
Lee YS, Nakahara K, Pham JW, Kim K, He Z, Sontheimer EJ, et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell 2004; 117: 69–81.
Murchison EP, Hannon GJ . miRNAs on the move: miRNA biogenesis and the RNAi machinery. Curr Opin Cell Biol 2004; 16: 223–9.
Hannon GJ . RNA interference. Nature 2002; 418: 244–51.
Hutvagner G, Zamore PD . A micro RNA in a multiple-turnover RNAi enzyme complex. Science 2002; 297: 2056–60.
Doench JG, Petersen CP, Sharp PA . siRNAs can function as miRNAs. Genes Dev 2003; 17: 438–42.
Liu Q, Rand TA, Kalidas S, Du F, Kim HE, Smith DP, et al. R2D2, a bridge between the initiation and effector steps of the Drosophila RNAi pathway. Science 2003; 301: 1921–5.
Tomari Y, Zamore PD . Perspective: machines for RNAi. Genes Dev 2005; 19: 517–29.
Dodd A, Chambers SP, Love DR . Short interfering RNA-mediated gene targeting in the zebrafish. FEBS Lett 2004; 561: 89–93.
Zender L, Hutker S, Liedtke C, Tillmann HL, Zender S, Mundt B, et al. Caspase 8 small interfering RNA prevents acute liver failure in mice. Proc Natl Acad Sci USA 2003; 100: 7797–802.
Klein C, Bock CT, Wedemeyer H, Wustefeld T, Locarnini S, Dienes HP, et al. Inhibition of hepatitis B virus replication in vivo by nucleoside analogues and siRNA. Gastroenterology 2003; 125: 9–18.
Verma UN, Surabhi RM, Schmaltieg A, Becerra C, Gaynor RB . Small interfering RNAs directed against beta-catenin inhibit the in vitro and in vivo growth of colon cancer cells. Clin Cancer Res 2003; 9: 1291–300.
Forte A, Cipollaro M, Cascino A, Galderisi U . Small interfering RNAs and antisense oligonucleotides for treatment of neurological diseases. Curr Drug Targets 2005; 6: 21–9.
Zhang Y, Boado RJ, Pardridge WM . In vivo knockdown of gene expression in brain cancer with intravenous RNAi in adult rats. J Gene Med 2003; 5: 1039–45.
Gilmore T, Gapuzan ME, Kalaitzidis D, Starczynowski D . Rel/NF-kappa B/I kappa B signal transduction in the generation and treatment of human cancer. Cancer Lett 2002; 181: 1–9.
Greten FR, Karin M . The IKK/NF-kappaB activation pathway-a target for prevention and treatment of cancer. Cancer Lett 2004; 206: 193–9.
Sun L, Deng L, Ea CK, Xia ZP, Chen ZJ . The TRAF6 ubiquitin ligase and TAK1 kinase mediate IKK activation by BCL10 and MALT1 in T lymphocytes. Mol Cell 2004; 14: 289–301.
Guo J, Verma UN, Gaynor RB, Frenkel EP, Becerra CR . Enhanced chemosensitivity to irinotecan by RNA interference-mediated down-regulation of the nuclear factor-kappaB p65 subunit. Clin Cancer Res 2004; 10: 3333–41.
Surabhi RM, Gaynor RB . RNA interference directed against viral and cellular targets inhibits human immunodeficiency virus type 1 replication. J Virol 2002; 76: 12963–73.
Brisibe EA, Okada N, Mizukami H, Okuyama H, Fujii YR . RNA interference: potentials for the prevention of HIV infections and the challenges ahead. Trends Biotechnol 2003; 7: 306–11.
Lee NS, Rossi JJ . Control of HIV-1 replication by RNA interference. Virus Res 2004; 102: 53–8.
Takaesu G, Surabhi RM, Park KJ, Ninomiya-Tsuji J, Matsumoto K, Gaynor RB . TAK1 is critical for IkappaB kinase-mediated activation of the NF-kappaB pathway. J Mol Biol 2003; 326: 105–15.
Takeda K, Takeuchi O, Tsujimura T, Itami S, Adachi O, Kawai T, et al. Science 1999; 284: 313–6.
Li Q, Van Antwerp D, Mercurio F, Lee KF, Verma IM . κB kinase 2 gene. Science 1999; 284: 321–5.
Li ZW, Chu W, Hu Y, Delhase M, Deerinck T, Ellisman M, et al. kB kinase (IKK) is essential for nuclear factor kappa B activation and prevention of apoptosis. J Exp Med 1999; 189: 1839–45.
Willis TG, Jadayel DM, Du MQ, Peng H, Perry AR, Abdul-Rauf M, et al. Bcl10 is involved in t(1;14)(p22;q32) of MALT B cell lymphoma and mutated in multiple tumor types. Cell 1999; 96: 35–45.
Lucas PC, McAllister-Lucas LM, Nunez G . NF-kappaB signaling in lymphocytes: a new cast of characters. J Cell Sci 2004; 117: 31–9.
Zhou H, Wertz I, O'Rourke K, Ultsch M, Seshagiri S, Eby M, et al. Bcl10 activates the NF-kappaB pathway through ubiquitination of NEMO. Nature 2004; 427: 167–71.
Wilkinson KD . Regulation of ubiquitin-dependent processes by deubiquitinating enzymes. FASEB J 1997; 11: 1245–56.
D'Andrea A, Pellman D . Deubiquitinating enzymes: a new class of biological regulators. Crit Rev Biochem Mol Biol 1998; 33: 337–52.
Chen G, Goeddel DV . TNF-R1 signaling: a beautiful pathway. Science 2002; 296: 1634–5.
Yoshimura S, Bondeson J, Brennan FM, Foxwell BM, Feldmann M . Role of NFkappaB in antigen presentation and development of regulatory T cells elucidated by treatment of dendritic cells with the proteasome inhibitor PSI. Eur J Immunol 2001; 31: 1883–93.
Yoshimura S, Bondeson J, Foxwell BM, Brennan FM, Feldmann M . Effective antigen presentation by dendritic cells is NF-kappaB dependent: coordinate regulation of MHC, co-stimulatory molecules and cytokines. Int Immunol 2001; 13: 675–83.
O'Sullivan BJ, Thomas R . CD40 ligation conditions dendritic cell antigen-presenting function through sustained activation of NF-kappaB. J Immunol 2002; 168: 5491–8.
Silverman N, Maniatis T . κB signaling pathways in mammalian and insect innate immunity. Genes Dev 2001; 15: 2321–42.
Neumann M, Fries H, Scheicher C, Keikavoussi P, Kolb-Maurer A, Brocker E, et al. Differential expression of Rel/NF-kappaB and octamer factors is a hallmark of the generation and maturation of dendritic cells. Blood 2000; 95: 277–85.
Burkly L, Hession C, Ogata L, Reilly C, Marconi LA, Olson D, et al. Expression of relB is required for the development of thymic medulla and dendritic cells. Nature 1995; 373: 531–6.
Kontgen F, Grumont RJ, Strasser A, Metcalf D, Li R, Tarlinton D, et al. Mice lacking the c-rel proto-oncogene exhibit defects in lymphocyte proliferation, humoral immunity, and interleukin-2 expression. Genes Dev 1995; 9: 1965–77.
Sha WC, Liou HC, Tuomanen EI, Baltimore D . κB leads to multifocal defects in immune responses. Cell 1995; 80: 321–30.
Franzoso G, Carlson L, Poljak L, Shores EW, Epstein S, Leonardi A, et al. κB/p52 present with defects in humoral responses, germinal center reactions, and splenic microarchitecture. J Exp Med 1998; 187: 147–59.
Laderach D, Compagno D, Danos O, Vainchenker W, Galy A . RNA interference shows critical requirement for NF-kappa B p50 in the production of IL-12 by human dendritic cells. J Immunol 2003; 171: 1750–7.
Trinchieri G . Interleukin-12: a cytokine at the interface of inflammation and immunity. Adv Immunol 1998; 70: 83–243.
Wesa AK, Galy A . IL-1 beta induces dendritic cells to produce IL-12. Int Immunol 2001; 13: 1053–61.
Gri G, Savio D, Trinchieri G, Ma X . Synergistic regulation of the human interleukin-12 p40 promoter by NFkappaB and Ets transcription factors in Epstein-Barr virus-transformed B cells and macrophages. J Biol Chem 1998; 273: 6431–8.
Barboric M, Nissen RM, Kanazawa S, Jabrane-Ferrat N, Peterlin BM . κB binds P-TEFβ to stimulate transcriptional elongation by RNA polymerase II. Mol Cell 2001; 8: 327–37.
West MJ, Lowe AD, Karn J . Activation of human immunodeficiency virus transcription in T cells revisited: NF-kappaB p65 stimulates transcriptional elongation. J Virol 2001; 75: 8524–37.
Kimpton J, Emerman M . Detection of replication-competent and pseudotyped human immunodeficiency virus with a sensitive cell line on the basis of activation of an integrated beta-galac tosidase gene. J Virol 1992; 66: 2232–9.
Hannon GJ, Conklin DS . RNA interference by short hairpin RNAs expressed in vertebrate cells. Methods Mol Biol 2004; 257: 255–66.
Hilliard B, Samoilova EB, Liu TS, Rostami A, Chen Y . Experimental autoimmune encephalomyelitis in NF-kappa B-deficient mice: roles of NF-kappa B in the activation and differentiation of autoreactive T cells. J Immunol 1999; 163: 2937–43.
Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS Jr . κB anti-apoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science 1998; 281: 1680–3.
Veiby OP, Read MA . Chemoresistance: impact of nuclear factor (NF)-kappaB inhibition by small interfering RNA. Commentary re Guo J. et al. Enhanced chemosensitivity to irinotecan by RNA interference-mediated down-regulation of the NFkappaB p65 subunit. Clin Cancer Res 2004; 10: 3333–41.
Muratovska A, Eccles MR . Conjugate for efficient delivery of short interfering RNA (siRNA) into mammalian cells. FEBS Lett 2004; 558: 63–8.
Song E, Lee SK, Wang J, Ince N, Ouyang N, Min J, et al. RNA interference targeting Fas protects mice from fulminant hepatitis. Nat Med 2003; 9: 347–51.
Yokota T, Sakamoto N, Enomoto N, Tanabe Y, Miyagishi M, Maekawa S, et al. Inhibition of intracellular hepatitis C virus replication by synthetic and vector-derived small interfering RNAs. EMBO Rep 2003; 4: 602–8.
McCaffrey AP, Meuse L, Pham TT, Conklin DS, Hannon GJ, Kay MA . RNA interference in adult mice. Nature 2002; 418: 38–9.
Lewis DL, Hagstrom JE, Loomis AG, Wolff JA, Herweijer H . Efficient delivery of siRNA for inhibition of gene expression in postnatal mice. Nat Genet 2002; 32: 107–8.
Dillon CP, Sandy P, Nencioni A, Kissler S, Rubinson DA, Van Parijs L . RNAi as an experimental and therapeutic tool to study and regulate physiological and disease processes. Annu Rev Physiol 2005; 67: 147–73.
Achenbach TV, Brunner B, Heermeier K . Oligonucleotide-based knockdown technologies: antisense versus RNA interference. Chembiochemistry 2003; 4: 928–35.
Silvestri LS, Taraporewala ZF, Patton JT . Rotavirus replication: plus-sense templates for double-stranded RNA synthesis are made in viroplasms. J Virol 2004; 78: 7763–74.
Chang J, Provost P, Taylor JM . Resistance of human hepatitis delta virus RNAs to dicer activity. J Virol 2003; 77: 11910–17.
Bitko V, Barik S . Phenotypic silencing of cytoplasmic genes using sequence-specific double-stranded short interfering RNA and its application in the reverse genetics of wild type negative-strand RNA viruses. BMC Microbiol 2001; 1: 34.
Li WX, Li H, Lu R, Li F, Dus M, Atkinson P, et al. Interferon antagonist proteins of influenza and vaccinia viruses are suppressors of RNA silencing. Proc Natl Acad Sci USA 2004; 101: 1350–5.
Lu S, Cullen BR . Adenovirus VA1 noncoding RNA can inhibit small interfering RNA and Micro RNA biogenesis. J Virol 2004; 78: 12868–76.
Shankar P, Manjunath N, Lieberman J . The prospect of silencing disease using RNA interference. JAMA 2005; 293: 1367–73.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Helen and Alden Wagner Cancer Fund and the Nasher Cancer Research Program.
Rights and permissions
About this article
Cite this article
Guo, J., Fu, Yc. & Becerra, C. Dissecting role of regulatory factors in NF-κB pathway with siRNA. Acta Pharmacol Sin 26, 780–788 (2005). https://doi.org/10.1111/j.1745-7254.2005.00140.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00140.x
Keywords
This article is cited by
-
Apoptosis induced by synthetic retinoic acid CD437 on human melanoma A375 cells involves RIG-I pathway
Archives of Dermatological Research (2009)