Abstract
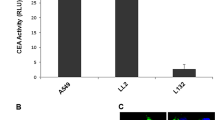
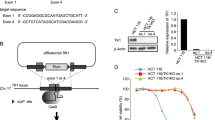
We isolated a 204-base pair carcinoembryonic antigen (CEA) promoter core region from a CEA-producing human colorectal carcinoma (CRC) and constructed retrovirus vectors carrying the expression cassette consisting of the CEA promoter core region and the cytosine deaminase (CD) gene. pCD2 retrovirus carrying the CD gene directed by the retrovirus long terminal repeat promoter served as a control vector. An in vitro study showed that the CEA promoter conferred CEA-producing cell-selective CD expression, specifically when the CD expression cassette was inserted into the 3′ long terminal repeat of the retrovirus vector. CD-modified CRC xenografts in nude mice were sensitive to 5-fluorocytosine and caused a profound bystander effect on the unmodified CRC. When nude mice harboring intraperitoneally disseminated CRCs were injected intraperitoneally with the CD expression cassette-carrying retrovirus-producing cells, CD transduction into the disseminated CRCs and bone marrow (BM) was observed. CD expression was, however, restricted to CRCs, and it was observed in both CRCs and BM of mice injected with pCD2 retrovirus-producing cells, resulting in better therapeutic outcomes without BM suppression. These results indicate that effective and safe in vivo gene therapy for CRC may be feasible by transferring the CD gene controlled by the CEA promoter core region.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cao, G., Kuriyama, S., Cui, L. et al. Analysis of the human carcinoembryonic antigen promoter core region in colorectal carcinoma-selective cytosine deaminase gene therapy. Cancer Gene Ther 6, 572–580 (1999). https://doi.org/10.1038/sj.cgt.7700082
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700082