Abstract
OBJECTIVE: Based on recent safety and efficacy data, combined with the known pharmacokinetic parameters of aminoglycosides in the newborn, oncedaily gentamicin should be preferable to the many other dosing regimens currently in use. Although there are growing data to support its use in term newborns, experience with preterm infants is more limited. In our Neonatal Intensive Care Unit, we experienced difficulties regarding complicated dosing regimens, actual dosing errors, and the tendency to check trough and peak levels around the third dose for infants receiving only a 48 hour course. Therefore, we conducted a quality improvement initiative in which we developed and tested a clinical practice guideline for the use of once-daily gentamicin for preterm and term infants that we hoped would yield trough and peak levels in our target range.
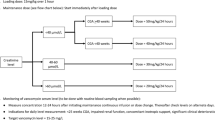
METHODS: We combined a review of the published English language literature with pharmacokinetic analysis of our own data prior to initiation of this new regimen to design the following dosing regimen: <35 weeks gestation: 3 mg/kg q 24 hours, ≥35 weeks gestation: 4 mg/kg q 24 hours. Our goal serum levels were a trough ≤2 μg/ml and a peak between 6 and 12 μg/ml. We collected and analyzed trough and peak levels from all infants receiving this dosing regimen in the first week of life for at least 72 hours between 3/1/99 and 12/31/00.
RESULTS: In total, 214 babies met our inclusion criteria, 75 of whom were <35 weeks gestation. 100% of babies of all gestational ages had a nontoxic trough level. For infants <35 weeks gestation, 79% had a therapeutic peak level, with a mean value of 6.8 μg/ml. For infants of at least 35 weeks gestation, 93% had a therapeutic peak level, with a mean value of 8.4 μg/ml. 92% of nontherapeutic peaks were too low.
CONCLUSION: This study of once-daily gentamicin represents the largest sample size of pre-term infants published to date. The proposed regimen is simple and yields a high proportion of desirable levels. We recommend it for use in preterm and term newborns.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hayani KC, Hatzopoulos FK, Frank AL, et al. Pharmacokinetics of once-daily dosing of gentamicin in neonates. J Pediatr 1997;131:76–80.
Lundergan FS, Glasscock GF, Kim EH, Cohen RS . Once-daily gentamicin dosing in newborn infants. Pediatrics 1999;103:1228–1234.
Skopnik H, Wallraf R, Nies B, Troster K, Heimann G . Pharmacokinetics and antibacterial activity of daily gentamicin. Arch Dis Child 1992;67:57–61.
Miron D . Once daily dosing of gentamicin in infants and children. Pediatr Infect Dis J 2001;20:1169–1173.
Chattopadhyay B . Newborns and gentamicin — how much and how often? J Antimicrob Chemother 2002;49:13–16.
Agarwal G, Rastogi A, Pyati S, Wilks A, Pildes RS . Comparison of once-daily versus twice-daily gentamicin dosing regimens in infants > or =2500 g. J Perinatol 2002;22:268–274.
Verpooten GA, Giuliano RA, Verbist L et al. Once-daily dosing decreases renal accumulation of gentamicin and netilmicin. Clin Pharmacol Ther 1989;45:22–27.
Skopnik H, Heimann G . Once daily aminoglycoside dosing in full term neonates. Pediatr Infect Dis J 1995;14:71–72.
Blaser J, Stone BB, Groner MC, Zinner SH . Comparative study with enoxacin and netilmicin in a pharmacodynamic model to determine importance of ratio of antibiotic peak concentration to MIC for bactericidal activity and emergence of resistance. Antimicrob Agents Chemother 1987;31:1054–1060.
Moore RD, Smith CR, Lietman PS . Association of aminoglycoside plasma levels with therapeutic outcome in Gram-negative pneumonia. Am J Med 1984;77:657–662.
Moore RD, Smith CR, Lietman PS . The association of aminoglycoside plasma levels with mortality in patients with Gram-negative bacteremia. J Infect Dis 1984;443–448.
Moore RD, Lietman PS, Smith PS . Clinical response to aminoglycoside therapy: importance of the ratio of peak concentration to minimal inhibitory concentration. J Infect Dis 1987;155:93–99.
Isaksson B, Hanberger H, Maller R, Nilsson LE, Nilsson M . Synergic post-antibiotic effect of amikacin in combination with beta-lactam antibiotics on Gram-negative bacteria. J Antimicrob Chemother 1991;28:25–34.
Craig WA, Redington J, Ebert SC . Pharmacodynamics of amikacin in vitro and in mouse thigh and lung infections. J Antimicrob Chemother 1991;27 (Suppl C):29–40.
Vogelman B, Gudmundsson S, Turnidge J, Leggett J, Craig WA . In vivo postantibiotic effect in a thigh infection in neutropenic mice. J Infect Dis 1988;157:287–298.
Van der Auwera P, Klastersky J . Serum bactericidal activity and postantibiotic effect in serum of patients with urinary tract infection receiving high-dose amikacin. Antimicrob Agents Chemother 1987;31:1061–1068.
McGrath BJ, Marchbanks CR, Gilbert D, Dudley MN . In vitro postantibiotic effect following repeated exposure to imipenem, temafloxacin, and tobramycin. Antimicrob Agents Chemother 1993;37:1723–1725.
Murphy JE, Austin ML, Frye RF . Evaluation of gentamicin pharmacokinetics and dosing protocols in 195 neonates. Am J Health Syst Pharm 1998;55:2280–2288.
Capers CC, Gregory DF, Herzog CC, Walsh W, Watson B . Optimal gentamicin dosing in neonates. Ann Pharmacother 1995;29:430–431.
de Alba Romero C, Gomez Castillo E, Manzanares Secades C, Rodriguez Lopez J, Arreaza Lopez L, Saenz Valiente P . Once daily gentamicin dosing in neonates. Pediatr Infect Dis J 1998;17:1169–1671.
Krishnan L, George SA . Gentamicin therapy in preterms: a comparison of two dosage regimens. Indian Pediatr 1997;34:1075–1080.
Solomon R, Kuruvilla KA, Job V, et al. Randomized controlled trial of once vs. twice daily gentamicin therapy in newborn. Indian Pediatr 1999;36:133–137.
Thureen PJ, Reiter PD, Gresores A, Stolpman NM, Kawato K, Hall DM . Once- versus twice-daily gentamicin dosing in neonates >/=34 Weeks' gestation: cost-effectiveness analyses. Pediatrics 1999;103:594–598.
Acknowledgements
Ann Stark MD, Eric Eichenwald MD, Lori Wright D. Pharm, Mica Astion.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hansen, A., Forbes, P., Arnold, A. et al. Once-daily Gentamicin Dosing for the Preterm and Term Newborn: Proposal for a Simple Regimen that Achieves Target Levels. J Perinatol 23, 635–639 (2003). https://doi.org/10.1038/sj.jp.7210996
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210996
This article is cited by
-
Gentamicin Dosing in Neonates with Normal Renal Function: Trough and Peak Levels
European Journal of Drug Metabolism and Pharmacokinetics (2021)
-
Optimizing gentamicin conventional and extended interval dosing in neonates using Monte Carlo simulation – a retrospective study
BMC Pediatrics (2019)
-
Should gentamicin trough levels be routinely obtained in term neonates?
Journal of Perinatology (2016)
-
High-dose gentamicin in newborn infants: is it safe?
European Journal of Pediatrics (2014)