Abstract
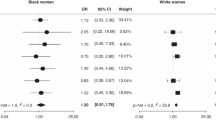
We examined ovarian cancer risk in relation to use of phenolphthalein-containing laxatives in 410 epithelial ovarian cancer cases and 713 controls. Compared to women who never used a laxative, ever use of a phenolphthalein-containing laxative was not associated with an increased risk of ovarian cancer (odds ratio, OR, 1.1, 95% confidence interval, CI, 0.9–1.4). Risk was slightly, but not significantly, higher with more frequent use (OR 1.2 for 75 or more days of use). When women who used non-phenolphthalein containing laxatives was used as the reference group, the associations were slightly, but not significantly larger (OR 1.4 for any use of phenolphthalein-containing laxatives and OR 1.5 for 75 or more days of use) © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bo-Linn GW, Santa Ana CA, Morawski SG and Fordtran JS (1983) Purging and caloric absorption in bulimic patients and normal women. Ann Intern Med 99: 14–17
Bytzer P, Stokholm M, Andersen I, Klitgaard NA and Schaffalitzky de Muckadell OB (1989) Prevalence of surreptitious laxative abuse in patients with diarrhoea of uncertain origin: a cost benefit analysis of a screening procedure. Gut 30: 1379–1384
De Smet PH and Wagenaar HW (1998) Weight reduction preparations from Thailand: unexpected complication for the traveler. Ned Tijdschr Geneeskd 142: 2798–2800
Dunnick JK and Hailey JR (1996) Phenolphthalein exposure causes multiple carcinogenic effects in experimental model systems. Cancer Res 56: 4922–4926
Dunnick JK, Hardisty JF, Herbert RA, Seely JC, Furedi-Machacek EM, Foley JF, Lacks GD, Stasiewicz S and French JE (1997) Phenolphthalein induces thymic lymphomas accompanied by loss of the p53 wild type allele in heterozygous p53-deficient (±) mice. Toxic Pathol 25: 533–540
Food and Drug Administration. Laxative drug products for over-the-counter human use. (1999). Federal Register 64: 4535–4540
National Toxicology Program (1996) NTP technical report on the toxicology and carcinogenesis studies of phenolphthalein (CAS no.77–09–8) in F344/N rats and B6C3F1R mice (feed studies). Research Triangle Park, NC: US Dept of Health and Human Services, Public Health Service, National Institutes of Health; Springfield, VA.: Available from the National Technical Information Service.
Ness RB, Grisso JA, Klapper J, Schlesselman JJ, Silberzweig S, Vergona R, Morgan M and Wheeler JE and the SHARE Study Group. Risk of ovarian cancer in relation to estrogen and progestin dose and use characteristics of oral contraceptives. Am J Epidemiol,
Physicians' Desk Reference for Nonprescription Drugs and Dietary Supplements. 11th edition . Montvale NJ, Medical Economics Company, Inc. 1990
Ravdin PM, van Beurden M and Jordan VC (1987) Estrogenic effects of phenolphthalein on human breast cancer cells in vitro. Breast Cancer Res Treat 9: 151–154
WHO Pharmaceuticals Newsletter (1998) Laxatives containing phenolphthalein – voluntary withdrawal. Nos. 1–2, 4–15
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Cooper, G., Longnecker, M., Sandler, D. et al. Risk of ovarian cancer in relation to use of phenolphthalein-containing laxatives. Br J Cancer 83, 404–406 (2000). https://doi.org/10.1054/bjoc.2000.1250
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1250