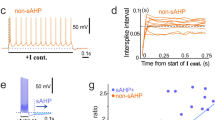
Abstract
Glutamate, the neurotransmitter at most excitatory synapses in the brain, activates a variety of receptor subtypes that can broadly be divided into ionotropic (ligand-gated ion channels) and metabotropic (G-protein-coupled) receptors. Ionotropic receptors mediate fast excitatory synaptic transmission, and based on pharmacological and molecular biological studies are divided into NMDA and non-NMDA subtypes. The non-NMDA receptor group is further divided into AMPA and kainate subtypes1. Virtually all fast excitatory postsynaptic currents studied so far in the central nervous system are mediated by the AMPA and NMDA subtypes of receptors. Surprisingly, despite extensive analysis of their structure, biophysical properties and anatomical distribution, a synaptic role for kainate receptors in the brain has not been found2. Here we report that repetitive activation of the hippocampal mossy fibre pathway, which is associated with high-affinity kainate binding3 and many of the kainate receptor subtypes4,5,6,7,8, generates a slow excitatory synaptic current with all of the properties expected of a kainate receptor. This activity-dependent synaptic current greatly augments the excitatory drive of CA3 pyramidal cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bettler, B. & Mulle, C. Review: Neurotransmitter receptors II. AMPA and kainate receptors. Neuropharmacology 34, 123–139 (1995).
Lerma, J., Morales, M., Vicente, M. A. & Herreras, O. Glutamate receptors of the kainate type and synaptic transmission. Trends Neurosci. 20, 9–12 (1997).
Foster, A. C., Mena, E. E., Monaghan, D. T. & Cotman, C. W. Synaptic localization of kainic acid binding sites. Nature 289, 73–75 (1981).
Werner, P., Voigt, M., Keinänen, K., Wisden, W. & Seeburg, P. H. Cloning of a putative high-affinity kainate receptor expressed predominantly in hippocampal CA3 cells. Nature 351, 742–744 (1991).
Egebjerg, J., Bettler, B., Hermans-Borgmeyer, I. & Heinemann, S. Cloning of a cDNA for a glutamate receptor subunit activated by kainate but not AMPA. Nature 351, 745–748 (1991).
Herb, A. et al. The KA-2 subunit of excitatory amino acid receptors shows widespread expression in brain and forms ion channels with distantly related subunits. Neuron 8, 775–785 (1992).
Petralia, R. S., Wang, Y.-X. & Wenthold, R. J. Histological and ulstrastructural localization of the kainate receptor subunits, KA2 and GluR6/7, in the rat nervous system using selective antipeptide antibodies. J. Comp. Neurol. 349, 85–110 (1994).
Siegel, S. J. et al. Distribution of the excitatory amino acid receptor subunits GluR2(4) in monkey hippocampus and colocalization with subunits GluR5-7 and NMDAR1. J. Neurosci. 15, 2707–2719 (1995).
Donevan, S. D. & Rogawski, M. A. GYKI 52466, a 2.3-benzodiazepine, is a highly selective, noncompetitive antagonist of AMPA/kainate receptor responses. Neuron 10, 51–59 (1993).
Zorumski, C. F., Yamada, K. A., Price, M. T. & Olney, J. W. Abenzodiazepine recognition site associated with the non-NMDA glutamate receptor. Neuron 10, 61–67 (1993).
Paternain, A. V., Morales, M. & Lerma, J. Selective antagonism of AMPA receptors unmasks kainate receptor-mediated responses in hippocampal neurons. Neuron 14, 185–189 (1995).
Wilding, T. J. & Huettner, J. E. Differential antagonism of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid-preferring and kainate-preferring receptors by 2,3-benzodiazepines. Mol. Pharmacol. 47, 582–587 (1995).
Kamiya, H., Shinozaki, H. & Yamamoto, C. Activation of metabotropic glutamate receptor type 2/3 suppresses transmission at rat hippocampal mossy fibre synapses. J. Physiol. (Lond.) 493, 447–455 (1996).
Verdoorn, T. A., Johansen, T. H., Drejer, J. & Nielsen, E. O. Selective block of recombinant glur6 receptors by NS-102, a novel non-NMDA receptor antagonist. Eur. J. Pharmacol. 269, 43–49 (1994).
Lerma, J., Paternain, A. V., Naranjo, J. R. & Mellstrom, B. Functional kainate-selective glutamate receptors in cultured hippocampal neurons. Proc. Natl Acad. Sci. USA 90, 11688–11692 (1993).
Salin, P. A., Scanziani, M., Malenka, R. C. & Nicoll, R. A. Distinct short-term plasticity at two excitatory synapses in the hippocampus. Proc. Natl Acad. Sci. USA 93, 13304–13309 (1996).
Partin, K. M., Patneau, D. K., Winters, C. A., Mayer, M. L. & Buonanno, A. Selective modulation of desensitization at AMPA versus kainate receptors by cyclothiazide and concanavalin A. Neuron 11, 1069–1082 (1993).
Trussell, L. O., Zhang, S. & Raman, I. M. Desensitization of AMPA receptors upon multiquantal neurotransmitter release. Neuron 10, 1185–1196 (1993).
Burnashev, N., Villarroel, A. & Sakmann, B. Dimensions and ion selectivity of recombinant AMPA and kainate receptor channels and their dependence on Q/R site residues. J. Physiol. (Lond.) 496, 165–173 (1996).
Scanziani, J. H. & Salin, P. A., Vogt, K. E., Malenka, R. C. & Nicoll, R. A. Use-dependent increases in glutamate concentration activate presynaptic metabotropic glutamate receptors. Nature 385, 630–634 (1997).
Robinson, J. H. & Deadwyler, S. A. Kainic acid produces depolarisation of CA3 pyramidal cells in the in vitro hippocampal slice. Brain Res. 221, 117–127 (1981).
Sawada, S., Higashima, M. & Yamamoto, C. Kainic acid induces long-lasting depolarizations in hippocampal neurons only when applied to stratum lucidum. Exp. Brain Res. 72, 135–140 (1988).
Sawada, S., Higashima, M. & Yamamoto, C. Inhibitors of high-affinity uptake augment depolarizations of hippocampal neurons induced by glutamate, kainate, and related compounds. Exp. Brain Res. 60, 323–329 (1985).
Represa, A., Tremblay, E. & Ben-Ari, Y. Kainate binding sites in the hippocampal mossy fibers: Localization and plasticity. Neuroscience 20, 739–748 (1987).
Malva, J. O. et al. Afunctionally active presynaptic high-affinity kainate receptor in the rate hippocampal CA3 subregion. Neurosci. Lett. 185, 83–86 (1995).
Chittajallu, R. et al. Regulation of glutamate release by presynaptic kainate receptors in the hippocampus. Nature 379, 78–81 (1996).
Gray, R., Rajan, A. S., Radcliffe, K. A., Yakehiro, M. & Dani, J. A. Hippocampal synaptic transmission enhanced by low concentrations of nicotine. Nature 383, 713–716 (1996).
Swanson, G. T., Feldmeyer, D., Kaneda, M. & Cull-Candy, S. G. Effect of RNA editing and subunit co-assembly on single-channel properties of recombinant kainate receptors. J. Physiol. (Lond.) 492, 129–142 (1996).
Otis, T. S., Wu, Y.-C. & Trussell, L. O. Delayed clearance of transmitter and the role of glutamate transporters at synapses with multiple release sites. J. Neurosci. 16, 1634–1644 (1996).
Weisskopf, M. G. & Nicoll, R. A. Presynaptic changes during mossy fibre LTP revealed by NMDA receptor-mediated synaptic responses. Nature 376, 256–259 (1995).
Huntley, G. W. et al. Selective distribution of kainate receptor subunit immunoreactivity in monkey neocortex revealed by a monoclonal antibody that recognizes glutamate receptor subunits GluR5/6/7. J. Neurosci. 13, 2965–2981 (1993).
Acknowledgements
We thank D. Leander for GYKI 53655; M. Frerking, S. Oliet and K. Vogt for comments on the manuscript; and H. Czerwonka for secretarial assistance. R.A.N. is a member of the Keck Center for Integrative Neuroscience and the Silvio Conte Center for Neuroscience Research. R.C.M. is a member of the Center for Neurobiology and Psychiatry, and the Center for the Neurobiology of Addiction. R.A.N. and R.C.M. are supported by grants from the NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castillo, P., Malenka, R. & Nicoll, R. Kainate receptors mediate a slow postsynaptic current in hippocampal CA3 neurons. Nature 388, 182–186 (1997). https://doi.org/10.1038/40645
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/40645
This article is cited by
-
Behavioral analysis of kainate receptor KO mice and the role of GluK3 subunit in anxiety
Scientific Reports (2024)
-
Sex differences in electrophysiological properties and voltage-gated ion channel expression in the paraventricular thalamic nucleus following repeated stress
Biology of Sex Differences (2022)
-
Intrathecal Injection of GRIP-siRNA Reduces Postoperative Synaptic Abundance of Kainate Receptor GluK2 Subunits in Rat Dorsal Horns and Pain Hypersensitivity
Neurochemical Research (2021)
-
Whole-Transcriptome Analysis of Dermal Fibroblasts, Derived from Three Pairs of Monozygotic Twins, Discordant for Parkinson’s Disease
Journal of Molecular Neuroscience (2020)
-
Stereotypical patterns of epileptiform calcium signal in hippocampal CA1, CA3, dentate gyrus and entorhinal cortex in freely moving mice
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.