Abstract
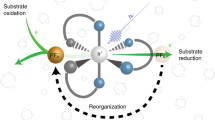
Zeolites are often employed as organizational media or supports for entrapped or adsorbed transition-metal catalysts and photo-catalysts1–6. In such applications, the individual catalytic species have been associated with the framework structure of the zeolite in a purely statistical (randomized) arrangement. A synthetic strategy developed recently7 has shown how a much higher level of organization can be obtained, so pointing the way to the generation of systems in which two or more active components can be arranged—both spatially and in terms of reactivity— within the zeolite host to enhance the efficiency of a desired catalytic reaction. Here we describe an application of this approach to photochemical storage of light energy. Such an application requires efficient photoinduced charge transfer between donor and acceptor molecules to form long-lived charge-separated states: the competing thermal back electron transfer reaction must be minimized. This is achieved in our system by arranging the active components (donor, acceptor and a 'sensitizing' intermediate molecule) such that they occupy adjacent cages within the zeolite framework, and results in unprecedented levels of net charge-separation efficiency.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
1. Breck, D. W. Zeolite Molecular Sieves (Wiley, New York, 1974). 2. Weitkamp, J., Karge, H. G., Pfiefer, H. & Holderich, W. (eds) Zeolites and Related Materials: State of the Art (Elsevier, Amsterdam, 1994). 3. Kalyanasundaram, K. Photochemistry in Microheterogeneous Systems (Academic, New York, 1987). 4. O'Regan, B. & Gratzel, M. Nature 353, 737-740 (1991). 5. Krueger, J. A., Meyer, J. E. & Mallouk, T. E. /. Am. Chem. Soc. 110, 8232-8234 (1988). 6. De Vos, D. E. et al. J. Indus. Phenom. Mol. Recogn. Chem. 21, 185-213 (1995). 7. Sykora, M., Maruszewski, K., Treffert-Ziemelis, S. M. & Kincaid, J. R. /. Am. Chem. Soc. (submitted). 8. DeWilde, W., Peters, G. & Lunsford, L. H. /. Phys. Chem. 84, 2306-2310 (1980). 9. Quayle, W. H. & Lunsford, J. H. Inorg. Chem. 21, 97-103 (1982). 10. Dutta, P. K. & Turbeville, W. /. Phys. Chem. 96, 9410-9416 (1992). 11. Borja, M. & Dutta, P. K. Nature 362, 43-45 (1993). 12. Wiliner, J., Yang, J., Loane, C, Otvos, J. W. & Calvin, M. /. Phys. Chem. 85, 3277-3282 (1981). 13. Slama-Schwork, A., Ottolenghi, M. & Avnir, D. Nature 355, 240-242 (1992). 14. Maruszewski, K., Strommen, D. P., Handrich, K. & Kincaid, J. R. Inorg. Chem. 30,4579-4582 (1991). 15. Maruszewski, K., Strommen, D. P., Handrich, K. & Kincaid, J. R. /. Am. Chem. Soc. 115, 8345-8350 (1993). 16. Maruszewski, K. & Kincaid, J. R. Inorg. Chem. 34, 2002-2006 (1995). 17. Toma, H. E., Auburn, P. R., Dodsworth, E. S., Golovin, M. N. & Lever, A. B. P. Inorg. Chem. 26, 4257-4263(1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sykora, M., Kincaid, J. Photochemical energy storage in a spatially organized zeolite-based photoredox system. Nature 387, 162–164 (1997). https://doi.org/10.1038/387162a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/387162a0
This article is cited by
-
Emerging applications of zeolites in catalysis, separation and host–guest assembly
Nature Reviews Materials (2021)
-
Design and fabrication of semiconductor photocatalyst for photocatalytic reduction of CO2 to solar fuel
Science China Materials (2014)
-
Effect of Method on Monolayer Assembly of Zeolite Microcrystals on Glass with Molecular Linkages
Topics in Catalysis (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.