Abstract
Basal breast carcinomas triple negative for estrogen receptors, progesterone receptors and Her2/neu breast carcinomas are more aggressive than conventional neoplasms. We studied 64 cases with immunohistochemistry, using 23 antibodies, to characterize diverse pathological pathways. A basal cytokeratin was identified in 81% of tumors and vimentin was identified in 55%. The mean Ki67 index was 46% (range, 10–90%). Coincident expression of p50 and p65, which suggests an active nuclear factor-κB factor, was present in 13% of neoplasms. Epithelial growth factor receptor (EGFR), insulin-like growth factor-I receptor (IGF-IR) or c-kit (CD117) was identified in 77% of tumors. Loss of protein tyrosine phosphatase was found in 14%, whereas Akt activation was present in 28%. Several differences were identified between two subtypes of basal breast carcinomas: the pure variant (negative S-100 and actin) was more frequently associated with ‘in situ carcinoma’ (P=0.019) and pBad overexpression (P=0.098), whereas the myoepithelial variant (positive S-100 or actin) showed more frequent tumor necrosis (P=0.048), vimentin expression (P=0.0001), CD117 expression (P=0.001) and activated caspase-3 (P=0.089). IGF-IR could be as important as EGFR for the growth of these neoplasms. Basal cell carcinoma has at least two subtypes with distinct microscopic and immunohistochemical features.
Similar content being viewed by others
Main
Breast cancer is one of the most frequent malignancies in women and ranks second among cancer deaths in adult females.1 At diagnosis, approximately 70–80% of breast carcinoma are positive for estrogen receptors (ERs) and/or progesterone receptors (PgRs), and ∼20% overexpress Her-2/neu.2, 3, 4 These findings could be related with gene expression-profiling studies,5, 6, 7, 8, 9, 10, 11, 12, 13, 14 which identify diverse varieties of breast carcinoma, mainly the ‘luminal’ one associated with hormone receptors, the ‘Her-2/neu-positive’ and the ‘basal’ or ‘triple negative’ breast carcinoma (negative for ERs, PgRs and Her-2/neu).
Basal breast carcinoma includes 15–25% of breast cancers, and it has been correlated with aggressive behavior, poor prognosis and better response to chemotherapy than conventional breast carcinoma. This variety of breast carcinoma was characterized by genetic studies and by immunohistochemical expression of basal cytokeratins, mainly types 5, 14 and 17, epidermal growth factor receptor (EGFR) and p53,15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30 but results from diverse studies are not always concordant. Recently, ‘pure’ and ‘myoepithelial’ variants of basal breast carcinoma were described, based on S-100, actin or p63 expression. Although these subtypes share some concordances in gene profiles and cytokeratin expressions, only the pure variant was associated with shorter survival in the multivariate analyses.17 Based on the above-reported heterogeneity of basal breast carcinomas, further studies seem necessary to better describe the characteristics of this neoplasm, which in turn may improve the knowledge of its growth and dissemination pathways. In this study, we analyze the role and relationship of several factors related with the cell cycle (Ki67, p53, p27, cyclin D1, p50, p52 and p65), membrane growth factor receptors other than Her2 (EGFR, insulin-like growth factor-I receptor (IGF-IR), platelet-derived growth factor receptor-α (PDGFRα) and CD117) and proteins involved in the phosphoinositide-3 kinase (PI3K)/protein tyrosine phosphatase (PTEN)/Akt signaling pathway (PTEN, Akt, and Bad and caspase-3) in a series of basal breast carcinomas.
Materials and methods
Sixty-four breast carcinomas that were triple negative for ERs, PgRs and Her-2/neu were diagnosed between 2000 and 2006. All hematoxylin- and eosin-stained sections of these tumors were reviewed to confirm conventional morphological parameters. Histological grades were assigned using modified Elston & Ellis criteria.31 Three representative areas of each tumor were then selected to build three tissue microarrays. Serial, 5-μm- thick tissue sections were used for immunohistochemical staining with the Envision method (Dako). Antibodies, dilutions, antigen retrieval methods and suppliers are listed in Table 1. Then, we studied the expression of S-100 and smooth muscle actin (SMA) to identify myoepithelial differentiation, basal cytokeratins (types 5/6, 14, 17), luminal cytokeratins (types 8/18) and vimentin. Ki67, p53, p27 and cyclin D1 were studied among the cell cycle-related proteins. Analyses of the nuclear factor-κB (NF-κB) pathway included the study of p50, p52, p65 and IKKα. In addition, diverse membrane receptors other than Her-2 were studied: Her-1(EGFR), platelet-derived growth factor receptor-α (PDGFRα), IGF-IR and c-kit (CD117). Investigation of the PI3K/PTEN/Akt pathway included the analysis of PTEN, phosphorylated Akt (pAkt), phosphorylated Bad (pBad) and activated caspase-3 expression.
Two pathologists (EL and SF) evaluated the immunohistochemical stainings blinded to patient outcome. Discordances were reviewed to achieve agreement.
The percentage of stained nuclei was evaluated, independent of the intensity, for Ki67, p53, p27, cyclin D1 and p50, p52 and p65. The percentage of cells with cytoplasmic staining was determined for S-100, actin, CKs 5/6, 14, 17 and 8/18, and vimentin. Distinct membrane staining in >10% of tumor cells for EGFR, IGF-IR and CD117 was counted as positive for expression. PDGFRα was positive when at least 10% of tumor cells showed cytoplasmic and/or membrane stainings. Immunostaining cytoplasmic scores for PTEN, pAkt and pBad were calculated by multiplying the percentage of labeled cells by the intensity of the staining (range 0–300). Scores >150 were considered positive. Cytoplasmic active caspase-3 was rarely detected and was always in less than 5% of cells, and the cutoff was established at 2%.
Clinical followup was reviewed by one of the authors (TR). Mean followup was 21 months (range, 3–70 months), but overall survival and disease-free survival were not conclusive due to the good initial response to treatment and short followup. Finally, the pathological and immunohistochemical findings were related with clinical data and outcome.
Statistical analyses were performed with the SPSS version-4 statistical software package. Continuous and ordinal variables between the two groups were compared using the Student's t-test. Dichotomous variables were compared with the Fisher exact test. The Kaplan–Meier method was used to estimate survival and the log-rank test was performed to assess survival differences between groups. A two-sided P-value of 0.05 was considered significant.
Results
The mean age of the 64 patients was 53 years (range, 25–86 years). Five patients had a history of familial breast carcinoma, and a BRCA1 mutation was detected in another case. Two patients had a previous history of breast cancer, which could not be reviewed.
The most frequent location of tumors was the upper-external quadrant of the breast (36%) as detailed in Table 2. Thirty-eight patients (59%) underwent a tumorectomy and 26 (41%) a modified radical mastectomy. Sentinel lymph node dissection was studied in 17 cases and was positive in 7. Dissection of axillary lymph nodes at levels I and II was performed in 33 patients with a mean of 18 nodes per patient (median: 15; range 5–45), and metastases were identified in 18 (55%) cases.
The pathological stage at diagnosis and followup was determined in 58 patients: 21 cases were in stage I (36%), 13 in IIA (22%), 14 in IIB (24%), 4 in IIIA (7%), 3 in IIIB (5%) and 3 in IV (5%).
Most carcinomas were between 2 and 5 cm (58%) and most were of the ductal type (NOS), although 2 were medullary carcinomas, 14 atypical medullary carcinomas and 1 had extensive papillar features (2%). A ductal ‘in situ’ (DCIS) component >10% was present in 36% of tumors and >25% in 6%. Histological grading was II in 25% of tumors and III in 75%. Other microscopic features are detailed in Table 2.
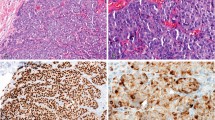
Immunohistochemical data are shown in Table 3 and Figure 1. Briefly, S-100 was positive in 14 (22%) cases and SMA was positive in 5 (8%), but 2 cases co-expressed both Ab. Therefore, 17 (27%) of our basal breast carcinomas had myoepithelial differentiation and were considered of the myoepithelial subtype.
Expressions of CK 5/6, 14, 17 and 8/18 were identified in 50, 56, 58 and 80% of tumors, respectively. Fifty-two (81%) tumors presented one of the basal cytokeratins studied (types 5/6, 14, 17). Expression of cytokeratins 5/6 was often associated with cytokeratin 17 (P=0.002) but not with cytokeratin 14. Vimentin was positive in 55% of tumors.
The mean percentage of Ki67-positive nuclei was 46% (median of 35 and range 10–90%) and only 20% of tumors had a Ki67<20%. Aberrant p53 expression (>10% of nuclei) was present in 53% of cases, cyclin D1 overexpression (>10% nuclei) was detected in 51% of tumors and loss of p27 was identified in 56%. Nuclear positivity for p50, p52 and p65 was detected in 19, 14 and 13%, respectively. Eight (13%) cases had coincident p50 and p65 expressions, suggesting activation of this NF-κB heterodimer.
Several membrane receptors were also studied: EGFR was identified in 34% of tumors, PDGFRα in 31%, IGF-IR in 36% and CD117 in 11%. Overlapping in the staining of IGF-IR, EGFR and CD117 was very rare (P=0.078), except in one myoepithelial basal breast carcinoma, which stained for EGFR and CD117. Overexpression of at least one of these three membrane receptors was associated with tumor growth in 44 (77%) cases. No relationship was found between PDGFRα and the other growth factor receptors.
Loss of PTEN expression (score <150) was seen in 14% of tumors, whereas pAkt was positive in 28% and pBad in 14%. Activated caspase-3 was detected in 58% and was associated with the presence of pBad (P=0.04).
Some correlation was detected between the expressions of IGF-IR, pAkt and pBad, although this was not statistically significant. Interestingly, loss of p27 appeared only in tumors lacking IGF-IR (P=0.062).
No significant differences were detected between pure and myoepithelial variants of basal breast carcinomas in most clinicopathological features or in the immunohistochemical studies, with the exception that pure basal breast carcinomas were more frequently associated with DCIS (45 vs 12%; P=0.019) and pBad overexpression (19 vs 0%; P=0.098), and myoepithelial basal breast tumors more frequently showed necrosis in >10% of the tumor cells (76 vs 47%; P=0.048), marked inflammatory reaction (59 vs 40%, P=0.19), expression of vimentin (>50% tumor cells) (82 vs 19%; P=0.0001), CD117 (35 vs 2%; P=0.001) and activated caspase-3 (76 vs 51%; P=0.089).
Two patients died from tumor progression: one 4 months after diagnosis of a stage IV pure variant and the second at 15 months after diagnosis of a stage IV myoepithelial variant. Five patients are presently alive with disease, three with pure variants (followup of 32, 30 and 27 months) and two with myoepithelial variants (followup of 27 and 38 months). Another patient died of other causes.
Disease-free survival was calculated according to the main characteristics. No differences were observed between the two groups and survival curves lacked significance due to the low mortality.
Discussion
Triple negative (ER, PgR and Her-2/neu negatives) or basal cell carcinomas are a variety of highly proliferative breast neoplasms that could also be identified by the immunohistochemical expression of basal cytokeratins (type 5, 14 or 17) and by DNA microarray studies.5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30 Table 4 summarizes relevant contributions in the characterization of these neoplasms, which are associated with a high histological grade, metaplastic features, wide tumor necrosis, pushing borders and marked lymphoid reaction.19 Furthermore, these tumors are related with medullary features32 and BRCA1 mutations.18, 28 Similar clinical and pathological findings were found in the present series.
Rakha et al17 have recently subdivided the basal breast carcinomas into two variants: one, ‘pure’, associated with early recurrence and reduced survival, and another with ‘myoepithelial’ differentiation. Approximately, one-fourth of our series presented myoepithelial differentiation, confirmed by immunohistochemical stains for S-100 and SMA, the former being the more sensitive marker. Although most pathological and immunohistochemical results were markedly similar in both subtypes of basal breast carcinomas, we identified several differences: DCIS was more frequent in tumors of the pure variant, whereas necrosis, lymphoid reaction, CD117, activated caspase-3 and vimentin were more common in those with myoepithelial differentiation. Vimentin expression was previously related with some breast carcinomas that today could have been included in the myoepithelial group,33, 35 whereas the remaining breast carcinomas rarely express vimentin.33 Indeed, these tumors can be related to a previously described variety of mammary neoplasms with frequent vascular dissemination, characterized by central acellular areas and expression of cytokeratin 14 and S-100.36
The main objective of this study was the exploration of alternative growth metabolic pathways to hormone receptors and Her2/neu in basal breast carcinomas. Rapid growth of these neoplasms could be related with a high Ki67 index and frequent p53 overexpression. The mean Ki67 index of 46% and the overexpression of p53 in 53% of our cases is much higher than the mean values described in sporadic and familial breast carcinomas.37
Cyclin D1 overexpression has been reported in 60% of DCIS and 70% of invasive cancers,37, 38 in association with a favorable clinical outcome and response to tamoxifen.38 In the present series, cyclin D1 overexpression was present in 52% of tumors in association with loss of p27 nuclear expression, but unrelated to pAkt or ER. In conclusion, cyclin D1 overexpression seems less relevant in the basal subtype than in conventional breast carcinomas.
NF-κB has an important role in cell cycle regulation39 and may participate in the invasion/metastasis of breast cancer by upregulation of vascular endothelial growth factor mRNA40 and matrix metalloproteinases.41 Coincident expression of p50 and p65 in 13% of our tumors suggests the activation of this NF-κB heterodimer in basal breast carcinomas. The p52–p65 heterodimer was not identified in our series.
Several growth factor receptors were studied in the present series. Epidermal growth factor receptor-1 (EGFR or Her-1) and c-erbB-2 (neu or Her-2) expression had been previously detected in 20 and 32% of breast neoplasms, respectively.15 In basal breast carcinoma, overexpression of EGFR is described between 30 and 54%,15, 17, 21, 24 as in this study. EGFR may relate with intracellular signaling kinases, notably mitogen-activated protein kinase and Akt, and promote ER transcriptional activity in a ligand-independent manner,42 and this cross-linking could exist in basal breast carcinoma. IGF-IR is a member of the type II receptor tyrosine kinase family that regulates cell growth in a variety of neoplasms, including breast cancers. Recently, IGF-IR was identified in ∼50% of breast carcinomas,43 including ER-negative tumors,44 although its presence was unrelated with prognosis or survival.45, 46 IGF-IR was identified in 36% of our series and rarely coincided with other growth factor receptors.
CD117 has been detected in 19–31% of basal breast carcinoma, whereas in conventional breast carcinomas its expression varies from 4 to 14%.21, 24, 34 In this paper, as in others,34 CD117 was mainly present in tumors with myoepithelial differentiation.
Coexpression of EGFR, IGF-IR and CD117 was very rare in our study, and 77% of all basal breast carcinomas showed at least one of these receptors. Our results suggest that IGF-IR may be relevant in the development of basal breast carcinoma without EGFR.
PTEN is a tumor suppressor gene that inhibits PI3K-dependent activation of Akt.47 Lack of PTEN induces Akt phosphorilization (activation), which subsequently promotes cell cycle arrest by decreasing the level and nuclear localization of cyclin D1,48, 49 inactivation of p-Bad, expression of Bcl-2 and inhibition of the Forkhead family of transcription factors.50 Lack of PTEN is found in 8–48% of breast carcinomas and is generally associated with a high grade, necrosis, node metastasis and absence of ER.51, 52, 53, 54, 55, 56 Generally, PTEN absence in breast carcinomas is due to gene promoter hypermethylation51, 52, 53, 54, 55 or loss of heterozygosity,53 whereas PTEN mutations are exceptional in sporadic neoplasms.57 Loss of PTEN is rare in basal breast carcinoma, and its relationship with Akt expression could not always be confirmed,51, 56 as in our series where Akt, present in 28% of tumors, was more frequently associated with growth factor receptors than with the PTEN pathway.52, 57 Recently, pAkt overexpression was described in 33% of in situ and 38% of ductal invasive breast carcinomas, although without any significant clinicopathological relationship,56 as in our cases.
In summary, Ki67 and p53 are higher in basal than in conventional breast carcinomas. The expression of EGFR, IGF-IR and CD117 may have a significant role in basal breast carcinomas, as well the heterodimer p50–p65 (NF-κB) in some cases. PTEN, pAkt, p27 and cyclin D1 alterations occur as in conventional mammary neoplasms. In addition to myoepithelial markers such as S-100 and actin, among others, myoepithelial variant could be differentiated from the pure variant of basal breast carcinomas for more frequent expression of vimentin and CD117.
References
Jemal A, Murray T, Ward E, et al. Cancer statistics, 2005. CA Cancer J Clin 2005;55:10–30.
Elledge RM, Fuqua SAW . Estrogen and progesterone receptors. In: Harris H (ed). Diseases of the Breast. Lippincott Williams & Wilkins: Philadelphia, 2000, p 47.
Fitzgibbons FK, Page DL, Weaver D, et al. Prognostic factors in breast cancer: college of American pathologists consensus statement 1999. Arch Pathol Lab Med 2000;124:966–978.
Bardou V-J, Arpino G, Elledge RM, et al. Progesterone receptor status significantly improves outcome prediction over estrogen receptor status alone for adjuvant endocrine therapy in two large breast cancer databases. J Clin Oncol 2003;21:1973–1979.
Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature 2000;406:747–752.
Sorlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subreast carcinomalasses with clinical implications. Proc Natl Acad Sci USA 2001;98:10869–10874.
Sorlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 2003;100:8418–8423.
van't Veer LJ, Dai H, van de Vijver MJ, et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002;415:530–536.
Sotiriou C, Neo SY, McShane LM, et al. Breast cancer classification and prognosis based on gene expression profiles from a population based study. Proc Natl Acad Sci USA 2003;100:10393–10398.
van de Vijver MJ, He YD, van't Veer LJ, et al. A gene expression signature as a predictor of survival in breast cancer. N Engl J Med 2002;347:1999–2009.
Ma XJ, Wang Z, Ryan PD, et al. A two gene expression ratio predicts clinical outcome in breast cancer patients treated with tamoxifen. Cancer Cell 2004;5:607–616.
Huang E, Cheng SH, Dressman H, et al. Gene expression predictors of breast cancer outcomes. Lancet 2003;361:1590–1596.
Jones C, Ford E, Gillett C, et al. Molecular cytogenetic identification of subgroups of grade III invasive ductal breast carcinomas with different clinical outcomes. Clin Cancer Res 2004;10:5988–5997.
Callagy G, Cattaneo E, Diago Y, et al. Molecular classification of breast carcinomas using tissue microarrays. Diag Mol Pathol 2003;12:27–34.
Abd El-Rehim DM, Pinder SE, Paish CE, et al. Expression of luminar and basal cytokeratins in human breast carcinoma. J Pathol 2004;203:661–671.
Abd El-Rehim DM, Ball G, Pinder SE, et al. High-throughput protein expression analysis using microarray technology of a large well characterized series identifies biologically distinct classes of breast cancer confirming recent cDNA expression analyses. Int J Cancer 2005;116:340–350.
Rakha EA, Putti TC, Abd El-Rehim DM, et al. Morphological and immunophenotypic analysis of breast carcinomas with basal and myoepithelial differentiation. J Pathol 2006;208:495–506.
Foulkes WD, Stefansson IM, Chappuis PO, et al. Germline BRCA1 mutations and basal epithelial phenotype in breast cancer. J Natl Cancer Inst 2003;95:1482–1485.
Livasy CA, Karaca G, Nanda R, et al. Phenotypic evaluation of basal-like subtype of invasive breast carcinoma. Mod Pathol 2006;19:264–271.
Gusterson BA, Ross DT, Heath VJ, et al. Basal cytokeratins and their relationship to the cellular origin and functional classification of breast cancer. Breast Cancer Res 2005;7:143–148.
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal like subtype of invasive breast carcinoma. Clin Cancer Res 2004;10:5367–5374.
van de Rijn M, Perou CM, Tibshirani R, et al. Expression of cytokeratins 17 and 5 identifies a group of breast carcinomas with poor clinical outcome. Am J Pathol 2002;161:1991–1996.
Rodriguez-Pinilla SM, Sarrio D, Honrado E, et al. Prognostic significance of basal-like phenotype and fascin expression in node-negative invasive breast carcinomas. Clin Cancer Res 2006;12:1533–1539.
Kim MJ, Ro JY, Ahn SH, et al. Clinicopathologic significance of basal-like subtype of breast cancer: a comparison with hormone receptor and Her2/neu-overexpressing phenotypes. Hum Pathol 2006;37:1217–1226.
Malzahn K, Mitze M, Thoenes M, et al. Biological and prognostic significance of stratified epithelial cytokeratins in infiltrating ductal breast carcinomas. Virchows Arch 1998;433:119–129.
Laakso M, Loman N, Borg A, et al. Cytokeratin 5/14-positive breast cancer: true basal phenotype confined to BRCA-1 tumors. Mod Pathol 2005;18:1321–1328.
Korsching E, Packeisen J, Agelopoulos K, et al. Cytogenetic alterations and cytokeratin expression patterns in breast cancer: integrating a new model of breast differentiation into cytogenetic pathways of breast carcinogenesis. Lab Invest 2002;82:1525–1533.
Haffty BG, Yang Q, Reiss M, et al. Locoregional relapse and distant metastasis in conservative managed triple negative early stage breast cancer. J Clin Oncol 2006;24:5652–5657.
Siziopikou KP, Cobleigh M . The basal subtype of breast carcinoma may represent the group of breast tumors that could benefit from EGFR targeted therapies. Breast 2007;16:104–107.
Rakha EA, El-Sayed ME, Green AR, et al. Prognostic markers in triple negative breast cancer. Cancer 2007;109:25–32.
Elston CW, Ellis IO . Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 1991;19:403–410.
Bertucci F, Finetti P, Cervera N, et al. Gene expression profiling shows medullary breast cancer is a subgroup of basal breast cancers. Cancer Res 2006;66:4636–4644.
Tsuda H, Takarabe T, Hasegawa F, et al. Large, central acellular zones indicating myoepithelial tumor differentiation in high-grade invasive ductal carcinomas as markers of predisposition to lung and brain metastases. Am J Surg Pathol 2000;24:197–202.
Tsuda H, Morita D, Kimura M, et al. Correlation of KIT and EGFR overexpression with ductal breast carcinoma of the solid-tubular subtype, nuclear grade 3, and mesenchymal or myoepithelial differentiation. Cancer Sci 2005;96:48–53.
Hungermann D, Buerger H, Oehlschlegel C, et al. Adenomyoepithelial tumors and myoepithelial carcinomas of the breast—a spectrum of monophasic and biphasic tumours dominated by immature myoepithelial cells. BMC Cancer 2005;5:92 doi:01.1186/1471-2407-5-92.
Korsching E, Packeisen J, Liedtke C, et al. The origin of vimentin expression in invasive breast cancer: epithelial–mesenchymal transition, myoepithelial histogenesis or histogenesis from progenitor cells with bilinear differentiation potential? J Pathol 2005;206:451–457.
Palacios J, Honrado E, Osorio A, et al. Phenotypic characterization of BRCA1 and BRCA2 tumors based in a tissue microarray study with 37 immunohistochemical markers. Breast Cancer Res Treat 2005;90:5–14.
Han S, Park K, Bae BN, et al. Cyclin D1 expression and patient outcome after tamoxifen therapy in estrogen receptor positive metastatic breast cancer. Oncol Rep 2003;10:141–144.
Chen F, Castranova V, Shi X . New insights into the role of nuclear factor-κB in cell growth regulation. Am J Pathol 2001;159:387–397.
Shibata A, Nagaya T, Imai T, et al. Inhibition of NF-kappaB activity decreases the VEGF mRNA expression in MDA-MB-231 breast cancer cells. Breast Cancer Res Treat 2002;73:237–243.
Helbig G, Christopherson II KW, Bhat-Nakshatri P, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem 2003;278:21631–21638.
Clark AS, West K, Streicher S, et al. Constitutive and inducible Akt activity promotes resistence to chemotherapy, transtuzumab, or tamoxifen in breast cancer cells. Mol Cancer Ther 2002;1:707–717.
Peiró G, Aranda FI, Adrover E, et al. Activated IGF1R in breast cancer with neoadjuvant chemotherapy. Lab Invest 2006;86 (Suppl 1):38A.
Gee JM, Robertson JF, Gutteridge E, et al. Epidermal growth factor receptor/Her2/insulin-like growth factor receptor signalling and oestrogen receptor activity in clinical breast cancer. Endocr Relat Cancer 2005;12:S99–S111.
Kostler WJ, Hudelist G, Rabitsch W, et al. Insulin-like growth factor-1 receptor (IGF-1R) expression does not predict for resistance to trastuzumab-based treatment in patients with Her-2/neu overexpressing metastatic breast cancer. J Cancer Res Clin Oncol 2006;132:9–18.
Nahta R, Yuan LX, Zhang B, et al. Insulin-like growth factor-I receptor/human epidermal growth factor receptor 2 heterodimerization contributes to trastuzumab resistance of breast cancer cells. Cancer Res 2005;65:11118–11128.
Li J, Yen C, Liaw D, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast and prostate cancer. Science 1997;275:1943–1947.
Chung JH, Eng C . Nuclear–cytoplasmic partitioning of phosphatase and tensin homologue deleted on chromosome 10 (PTEN) differentially regulates the cell cycle and apoptosis. Cancer Res 2005;65:8096–8100.
Chung JH, Ostrowski MC, Romigh T, et al. The ERK1/2 pathway modulates nuclear PTEN-mediated cell cycle arrest by cyclin D1 transcriptional regulation. Hum Mol Genet 2006;15:2553–2559.
Rhei E, Kang L, Bogomolniy F, et al. Mutation analysis of the putative tumor supressor gene PTEN/MMAC1 in primary breast carcinomas. Cancer Res 1997;57:3657–3659.
Datta K, Franke TF, Chan TO, et al. AH/PH domain mediated interaction between AKT molecules and its potential role in AKT regulation. Mol Cell Biol 1995;15:2304–2310.
Panigrahi AR, Pinder SE, Chan SY, et al. The role of PTEN and its signalling pathways, including AKT, in breast cancer; an assessment of relationships with other prognostic factors and with outcome. J Pathol 2004;204:93–100.
Depowski PL, Rosenthal SI, Ross JS . Loss of expression of the PTEN gene protein product is associated with poor outcome in breast cancer. Mod Pathol 2001;14:672–676.
Perren A, Weng LP, Boag AH, et al. Immunohistochemical evidence of loss of PTEN expression in primary ductal adenocarcinomas of the breast. Am J Pathol 1999;155:1253–1260.
Bose S, Crane A, Hibshoosh H, et al. Reduced expression of PTEN correlates with breast cancer progression. Hum Pathol 2002;33:405–409.
Garcia JM, Silva J, Peña C, et al. Promoter methylation of PTEN gene is a common molecular change in breast cancer. Genes Chromosomes Cancer 2004;41:117–124.
Bose S, Chandran S, Mirocha JM, et al. The Akt pathway in human breast cancer: a tissue-array-based analysis. Mod Pathol 2006;19:238–245.
Acknowledgements
This work was supported by Grants FISSS PI060709 Ministry of Health, Spain, and Pfizer Grant for Breast Cancer Research (2007). We thank Ms Carolyn Newey for her help with the English text.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure/conflict of interest
None.
Rights and permissions
About this article
Cite this article
Lerma, E., Peiro, G., Ramón, T. et al. Immunohistochemical heterogeneity of breast carcinomas negative for estrogen receptors, progesterone receptors and Her2/neu (basal-like breast carcinomas). Mod Pathol 20, 1200–1207 (2007). https://doi.org/10.1038/modpathol.3800961
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800961
Keywords
This article is cited by
-
Mammographic and ultrasonographic features of triple-negative breast cancer compared with non-triple-negative breast cancer
Journal of Ultrasound (2022)
-
Simultaneous targeting of HER family pro-survival signaling with Pan-HER antibody mixture is highly effective in TNBC: a preclinical trial with PDXs
Breast Cancer Research (2020)
-
Machine learning for diagnostic ultrasound of triple-negative breast cancer
Breast Cancer Research and Treatment (2019)
-
Prognostic significance of tumor-infiltrating CD8+ and FOXP3+ lymphocytes in residual tumors and alterations in these parameters after neoadjuvant chemotherapy in triple-negative breast cancer: a retrospective multicenter study
Breast Cancer Research (2015)
-
Tumor-infiltrating CD8+ and FOXP3+ lymphocytes in triple-negative breast cancer: its correlation with pathological complete response to neoadjuvant chemotherapy
Breast Cancer Research and Treatment (2014)