Abstract
p16INK4A and p57KIP2 are inhibitors of cyclin-dependent kinases and their inactivation by methylation has been reported as a major tumorigenic mechanism in tumors. To examine whether methylation of p16INK4A and p57KIP2 is involved in the development and progression of gastric MALT lymphomas, 24 gastric low-grade lymphomas of MALT, 11 diffuse large B-cell lymphomas, and 10 each case of gastric lymphoid follicles with and without Helicobacter pylori infection were studied. H. pylori infection was positive in 85.7% of the gastric lymphomas. In the gastric lymphoid follicles positive for H. pylori, methylation of p16INK4A was detected in 10% of cases, while methylation of p57KIP2 was not detected. In low-grade MALT lymphomas, p16INK4A and p57KIP2 were methylated in 41.7 and 29.2% of the cases, respectively. In diffuse large B-cell lymphomas, methylation of p16INK4A and p57KIP2 was found in 72.7 and 36.4% of the cases, respectively. All but one case with p16INK4A and p57KIP2 methylation was H. pylori positive and most of them were stage I. Our results indicate that methylation of p16INK4A followed by p57KIP2 methylation involves during the tumorigenesis of gastric MALT lymphomas associated with H. pylori infection. As methylation of these two genes was more frequent in the higher grade (P<0.05), it may contribute to the malignant progression of gastric MALT lymphomas.
Similar content being viewed by others
Main
Hypermethylation of CpG islands within promoter regions of genes is associated with transcriptional inactivation and represents an important mechanism of gene silencing in the pathogenesis of malignancies.1 This epigenetic phenomenon acts as an alternative to mutations and deletions for disrupting the tumor suppressor gene function.2 A large number of genes that involve several fundamental cellular pathways may be affected in virtually all types of human cancers by aberrant CpG island methylation. p16INK4A and p57KIP2 genes are the suppressors of cyclin-dependent kinase (CDK), a member of the tumor suppressor gene, and their inactivation in various tumor cells has been reported.3, 4, 5 p16INK4A is a member of INK4A/ARF family and is inactivated by a loss, deletion, or hypermethylation of the promoter region of the gene. In non-Hodgkin’s lymphomas, inactivation of p16INK4A is primarily due to hypermethylation rather than to genetic alterations. The frequencies of p16INK4A methylation in non-Hodgkin’s B-cell lymphoma have been reported to be 15–46%.6, 7, 8 As the incidence of p16INK4A methylation in diffuse large B-cell lymphomas is higher than in the low-grade lymphomas, this is thought to be involved in the malignant transformation.6, 9, 10 In gastric MALT lymphomas, methylation of p16INK4A is between 58 and 67%, which is higher than other lymphomas. So, inactivation of p16INK4A by methylation is thought to play an important role in the development of MALT lymphomas.7, 10, 11, 12
p57KIP2 is a member of the Cip/Kip family of CDK inhibitors that intervenes in the formation of the cyclin-CDK complex and is considered a putative tumor suppressor gene.5, 13, 14, 15 In mouse, p57KIP2 gene is expressed from the maternal allele only, and the paternal allele is methylated and repressed in all tissues examined.16 In humans, ∼90% of the expression comes from the maternal allele, but no differential methylation between the two parental alleles has been reported.17, 18, 19, 20 Inactivation of p57KIP2 is known to be involved in the development of human malignancies such as hepatocellular carcinoma,21, 22 urinary bladder carcinoma,23 and gastric carcinoma,24 as well as Beckwith–Wiedemann Syndrome.25 Inactivation of p57KIP2 gene expression other than mutation was suggested due to rare occurrence of p57 gene mutations in these cancers. Recently, it has been reported that methylation of p57KIP2 occurs frequently in malignant lymphomas of B-cell type26 and adult acute lymphocytic leukemia.27 However, studies on p57KIP2 methylation in gastric MALT lymphomas are not reported and its clinical significance is not established yet.
Infection with Helicobactor pylori is one of the most important causes of gastric MALT lymphoma.28, 29 In Korea, the infection rate of H. pylori is around 60% in adults.30 Here, we investigated two major inhibitors of CDK in gastric MALT lymphomas to evaluate whether methylation of p16INK4A and p57KIP2 is involved in the development and progression of the gastric MALT lymphomas associated with H. pylori infection.
Materials and methods
Study Population
In total, 35 paraffin-embedded tissue sections of patients from 1996 to 2003 who were diagnosed as having gastric MALT lymphomas were used: 29 cases were biopsy specimens and six cases were surgical samples. The patients’ age ranged from 22 to 80 years with a mean age of 56.3 years. The male to female ratio was 3:2. As a control group, 10 of each case of benign gastric lymphoid hyperplasia negative and positive for H. pylori were used.
Pathologic Examinations
After review of all hematoxylin and eosin-stained sections used for diagnosis, the cases were classified as low-grade lymphoma of MALT (extranodal marginal zone B-cell lymphoma of MALT type) when there are dense diffuse infiltrates of centrocyte-like cells with monocytoid differentiation and typical lymphoepithelial lesion, which eventually leads to eosinophilic degeneration and disintegration of glandular epithelium (Figure 1). Diffuse large B-cell lymphoma was diagnosed if compact clusters, confluent aggregates, or sheets of transformed large centroblast- or immunoblast-like cells with B-cell immunophenotype were found (Figure 2).31, 32
To detect H. pylori, Warthin–Starry silver staining was performed and the grades of infection were defined by the visual analog scales of the updated Sydney system.33
Methylation-Specific PCR for p16 and p57
To assess the methylation status, methylation-specific polymerase chain reaction (MS-PCR) on the p16INK4A and p57KIP2 genes was performed. The paraffin-embedded tissues were sectioned to 10 μm thickness. After they were stained with hematoxylin and eosin, the selected areas were collected with a 26-gauge needle by manual microdissection. The cells attached to the tip of the needle were transferred into a 1.5 ml eppendorf tube containing 20 μl proteinase K buffer solution (0.5% Tween 20, 1 mM EDTA pH 8.0, 50 mM Tris pH 8.5). They were incubated in a 55°C water bath for 12–15 h and next they were incubated at 100°C for 5 min to inactivate proteinase K, and stored at 5°C until further use. DNA (1 μg) was diluted with 50 μl distilled water and 5 μl 0.3 N NaOH was added. The mixture was kept in a 50°C water bath for 10 min; for treating this mix with bisulfite, 550 ml 1 mM hydroquinone (Sigma, St Louis, USA) and 550 ml 3.5 M sodium bisulfite (Sigma, St Louis, USA) were mixed well at pH 5.0 and reacted at 50°C for 16 h. The primer sequences, annealing temperatures, and the sizes of products are summarized in Table 1. The reaction mixture was as follows: 50 ng DNA modified by bisulfite, 5 pmol primers, 4 mM dNTP, 10 × PCR reaction Buffer (20 mM Tris-HCl, 100 mM KCl, 0.1 mM EDTA, and 1 mM DTT), and 1.25 U of Taq polymerase (EX taq, TAKARA, Japan) in a total volume of 20 μl. After utilizing a Thermal cycler (MJ Research PTC-100, Watertown, USA), the PCR products were subjected to 6% polyacrylamide gel electrophoresis, stained with ethidium bromide and assessed under a UV-transilluminator. If only the unmethylated bands were detected, the samples were evaluated as negative for methylation. If unmethylated bands were detected with the methylated bands or if only the methylated bands were detected, the samples were evaluated as positive for methylation.
Statisctical Analyses
Statistical analysis was performed using the SPSS software (SPSS Inc., Chicago, IL, USA) using χ2 test and probability values less than 0.05 were considered statistically significant.
Results
Clinical and Histopathologic Findings
The endoscopic findings included ulcer in 26 cases and erosion in seven cases. Nodular lesion and atrophy were detected in one case each. The location of the lesion was in the antrum in 20 cases, and this was followed by the lower body in eight cases and the midbody in seven cases. The microscopic examination of the gastric tissues revealed infiltration of atypical lymphocytes in the lamina propria and prominent lymphoepithelial lesions. On the histological classification, 24 cases were low-grade MALT lymphomas and 11 cases were diffuse large B-cell lymphomas. Among 11 diffuse large B-cell lymphomas, coexistent low-grade MLAT lymphomas, lymphoepithelial lesion and/or evidence of follicular colonization suggesting MALT origin were observed in eight cases. In the remaining three cases of large B-cell lymphomas, the patients showed no evidences of immunodeficiency.
The disease stage, as classified by the modified Ann Arbor system,34 was stage I in 29 cases, stage II in two cases, stage III in three cases, and stage IV in one case. Bone marrow involvement was detected in the stage IV case. In our study population, four patients died and the remaining patients were still alive for a period of 9–80 months (mean time: 37 months). Four patients with poor outcome consisted of three high-grade and one low-grade lymphomas of MALT type with a mean age of 72.8 years.
With regard to H. pylori infection, 30 cases (85.7%) were H. pylori positive and five cases were H. pylori negative. The degree of H. pylori infection was mild in 19 cases, moderate in four cases, and severe in seven cases. The clinicopathologic characteristics of the patients are summarized in Table 2.
Methylation of p16 and p57 in Gastric MALT Lymphomas
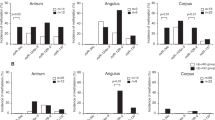
In the control gastric mucosa with lymphoid follicles that was negative for H. pylori, methylation of p16INK4A and p57KIP2 was not detected. In the group positive for H. pylori and containing lymphoid follicles, methylation of p16INK4A gene was found in one case, while p57KIP2 was not methylated. Methylation of p16INK4A was found in 18 of 35 cases (51.4%) and methylation of p57KIP2 was detected in 11 of 35 cases (31.4%) (Figure 3). Methylation of either p16INK4A or p57KIP2 was found in 25 of 35 cases (71.4%) and methylation of both genes was found in four of 35 cases (11.4%). For the grades of lymphomas, in the low-grade lymphomas of MALT, methylation of p16INK4A gene was found in 10 of 24 cases (41.7%) and methylation of p57KIP2 was found in seven cases (29.2%). In the large B-cell lymphomas, methylation of p16INK4A gene was detected in eight of 11 cases (72.7%) and methylation of p57KIP2 was detected in four cases (36.4%) (Table 3). Overall, methylation of p16INK4A and p57KIP2 in the diffuse large B-cell lymphomas was higher than in the low-grade MALT lymphomas (P<0.05).
When comparing methylation status with the modified Ann Arbor stages, 18 cases with p16INK4A methylation consisted 12/29 stage I, 2/2 stage II, 3/3 stage III, and 1/1 stage IV. For p57KIP2 methylation, 11 cases included 9/29 stage I, 1/2 stage II, and 1/3 stage III. Although the number of cases with advanced disease stages was small, most methylation of both genes started to be detected in the early stages. Among four patients who died of gastric lymphomas, three large B-cell lymphomas were methylated in both genes and their clinical stages were I, II, and III in each case. However, methylation of p57KIP2 was not detected in the stage IV patients.
Out of 18 cases with p16INK4A methylation, H. pylori infection was positive in 83.3% of the cases and negative in 16.7% of the cases. In the 11 cases with p57KIP2 methylation, H. pylori infection was found in 90.9% of the cases and it was negative in 9.1% of the cases (Table 4).
Discussion
Altered methylation patterns can be used as biomarkers for cancer detection, for the assessment of prognosis, and for the prediction of response to antitumor treatment. Furthermore, clinical trials using epigenetically targeted therapies have yielded promising results for acute and chronic leukemias as well as for myelodysplastic syndromes. The exploration of our growing knowledge about epigenetic aberrations may help to develop novel strategies for the diagnosis and treatment of hematopoietic malignancies in the future.2 In the non-Hodgkin’s B-cell lymphomas and MALT lymphomas, p16INK4A gene has been reported to be methylated in 15–67% of tumors.7, 10, 11, 12 Martinez-Delgado7 have reported that in MALT lymphomas, p16INK4A gene is methylated in 44% of the low-grade MALT lymphomas and in all the diffuse large B-cell lymphomas; thus, it is associated with the malignant transformation of lymphomas. For our study on 35 cases of gastric MALT lymphomas, methylation of p16INK4A was detected in 51.4% of the cases, which is in agreement with previous reports. In addition, methylation of p16INK4A was detected in 41.7% of the low-grade MALT lymphomas and in 72.7% of the diffuse large B-cell lymphomas. This supports the previous findings that p16INK4A methylation is involved in the development of MALT lymphoma as well as being involved in the malignant progression to a higher grade.7, 8, 10
Although methylation study on p57KIP2 gene has only rarely been investigated, 54.9% of the non-Hodgkin’s lymphomas and 50% of the acute lymphocytic leukemias showed methylation of p57KIP2.26, 27 In our experiment on p57KIP2 gene, the primers described in previous paper26 were modified after checking the sequences from the Genebank (http://www.ncbi.nlm.nih.gov). In this first study of p57KIP2 methylation in the gastric MALT lymphomas, 31.4% of the cases were methylated and the diffuse large B-cell lymphomas showed higher frequency of methylation than the low-grade MALT lymphomas. Similar to p16INK4A, methylation of p57KIP2 may be involved in the development and the malignant transformation of gastric MALT lymphoma.
When comparing methylation status and the disease stages, methylation of p16INK4A and p57KIP2 was detected in the cases with early stage. However, there was no methylation of p57KIP2 in the gastric mucosa with or without H. pylori infection and only rare methylation of p16INK4A in the H. pylori-positive gastric mucosa with lymphoid follicles. The abrupt and frequent occurrence of methylation of those two genes in the stage I MALT lymphomas suggests that this may be involved early in the development of gastric MALT Lymphoma.35
In our study, three out of four patients with poor prognosis showed diffuse large B-cell lymphomas with methylation of both of p16INK4A and p57KIP2 genes although their clinical stages were I in two cases and II in one case. Although limited numbers of cases prohibit further clinical interpretation, methylation of these genes as a predicting factor needs to be evaluated.
For the correlation of methylation and H. pylori infection, 83.3% of the cases with p16INK4A methylation and 90.9% of the cases with p57KIP2 methylation were infected with H. pylori, and this suggests that H. pylori infection may play an important role in the methylation of these genes. It has been reported that in MALT lymphoma, methylation of p16INK4A gene was not detected after treating H. pylori with antibiotics.12, 36, 37 In this study, the control gastric mucosa with lymphoid follicles showed methylation of p16INK4A in 10% of the cases. These findings suggest that infection with H. pylori itself may play an important role in methylation. DNA damage with the considerable production of reactive oxygen species and inducible nitric oxide synthase caused by H. pylori infection will be a potential mechanism,38 but more research is required to elucidate this mechanism. Based on the observation that methylation is involved in the initial stage of the development of MALT lymphomas, efforts have been made to apply methylation of p16INK4A and p57KIP2 genes in clinics. It has been reported that the assessment of methylation of p16INK4A gene may be used as an early diagnostic tool for lung cancer.39 In addition, by using it as a follow-up marker in non-Hodgkin's lymphoma patients, it may be used for evaluating the outcome of therapy and for the early detection of recurrence.11 As MS-PCR is a sensitive, simple, rapid, and inexpensive method to determine the methylation status of CpG islands from very small amounts of DNA, the assessment of methylation may reflect the status of residual cancer cells with more sensitivity than the histological findings and so facilitate detection of recurrence. From such an aspect, our research data also suggest the possible application of p16INK4A and p57KIP2 methylation as a marker for the early diagnosis and detection of residual tumor of gastric MALT lymphomas.
References
Jones PA, Laird PW . Cancer epigenetics comes of age. Nat Genet 1999;21:163–167.
Galm O, Esteller M . Beyond genetics—the emerging role of epigenetic changes in hematopietic malignancies. Int J Hematol 2004;80:120–127.
Kamb A, Gruis NA, Weaver-Feldhaus J, et al. A cell cycle regulator potentially involved in genesis of many tumor types. Science 1994;264:436–440.
Nobori T, Miura K, Wu DJ, et al. Deletions of the cyclin-dependent kinase-4 inhibitor gene in multiple human cancers. Nature 1994;368:753–756.
Lee MH, Reynisdottir I, Massague J . Cloning of p57KIP2, a cyclin-dependent kinase inhibitor with unique domain structure and tissue distribution. Genes Dev 1995;9:639–649.
Herman JG, Civin CI, Issa JJ, et al. Distinct patterns of inactivation of p15INK4b and p16INK4a characterize the major types of hematological malignancies. Cancer Res 1997;57:837–841.
Martinez-Delgado B, Fernandez-Piqueras J, Carcia MJ, et al. Hypermethylation of a 5′ CpG island of p16 is a frequent event in non-Hodgkin's lymphoma. Leukemia 1997;11:425–428.
Baur AS, Shaw P, Burri N, et al. Frequent methylation silencing of p15INK4b(MTS2) and p16INK4a(MTS1) in B-cell and T-cell lymphomas. Blood 1999;94:1773–1781.
Pinyol M, Cobo F, Bea S, et al. p16INK4a gene inactivation by deletions, mutations and hypermethylation is associated with transformed and aggressive variants of Non-Hodgkin's lymphomas. Blood 1998;91:2977–2984.
Villuendas R, Sanchez-Beato M, Martinez JC, et al. Loss of p16/INK4A protein expression in non-Hodgkin's lymphomas is a frequent findings associated with tumor progression. Am J Pathol 1998;153:887–897.
Martinez-Delgado B, Richart B, Garcia MJ, et al. Hypermethylation of p16INK4a and p15INK4b genes as a marker of disease in the follow-up of non-Hodgkin's lymphomas. Br J Haematol 2000;109:97–103.
Kim YS, Kim JS, Jung HC, et al. Regression of low-grade gastric mucosa-associated lymphoid tissue lymphoma after eradication of Helicobacter pylori: possible association with p16 hypermethylation. J Gastroenterol 2002;37:17–22.
EI-Deiry WS, Tokino T, Veculescu VE, et al. WAF1, a potential mediator of p53 tumor suppression. Cell 1993;75:817–825.
Gu Y, Turk CW, Morgan DO . Inhibition of CDK2 activity in vivo by associated 20 K regulatory subunit. Nature 1993;366:707–710.
Toyoshima H, Hunter T . p27, a novel inhibitor of G1 cyclin-cdk protein kinase activity, is related to p21. Cell 1994;78:67–78.
Hatada I, Mukai T . Genomic imprinting of p57(KIP2), cyclin-dependent kinase inhibitor, in mouse. Nat Genet 1995;11:204–206.
Chung WY, Yuan L, Feng L, et al. Chromosome 11p15.5 regional imprinting: comparative analysis of KIP2 and H19 in human tissues and Wilms’ tumors. Hum Mol Genet 1996;5:1101–1108.
Hatada I, Inazawa J, Abe T, et al. Genomic imprinting of human p57(KIP2) and its reduced expression in Wilms’ tumors. Hum Mol Genet 1996;5:783–788.
Kondo M, Matsuoka S, Uchida K, et al. Selective maternal-allele loss in human lung cancers of the maternally expressed p57(KIP2) gene at 11p15.5. Oncogene 1996;12:1365–1368.
Matsuoka S, Thompson JS, Edwards MC, et al. Imprinting of the gene encoding a human cyclin-dependent kinase inhibitor, p57KIP2, on chromosome 11p15. Proc Natl Acad Sci USA 1996;93:3026–3030.
Fujimori M, Tokino T, Hino O, et al. Allelotype study of primary hepatocellular carcinoma. Cancer Res 1991;51:89–93.
Nakai S, Masaki T, Shiratori Y, et al. Expression of p57KIP2 in hepatocellular carcinoma: relationship between tumor differentiation and patient survival. Int J Oncol 2002;20:769–775.
Fearon ER, Feinberg AP, Hamilton SR, et al. Loss of genes on the short arm of chromosome 11 in bladder cancer. Nature 1985;318:377–380.
Shin JY, Kim HS, Lee KS, et al. Mutation and expression of the P27KIP1 and P57KIP2 genes in human gastric cancer. Exp Mol Med 2000;32:79–83.
Koufos A, Grundy P, Morgan K, et al. Familial Wiedemann–Beckwith syndrome and a second Wilms’ tumor locus both map to 11p15.5. Am J Hum Genet 1989;44:711–719.
Li Y, Nagai H, Ohno T, et al. Aberrant DNA methylation of p57KIP2 gene in the promoter region in lymphoid malignancies in B-cell phenotype. Blood 2002;100:2572–2577.
Shen L, Toyota M, Kondo Y, et al. Aberrant DNA methylation of p57KIP2 identifies a cell-cycle regulatory pathway with prognostic impact in adult acute lymphocytic leukemia. Blood 2003;101:4131–4136.
Wotherspoon AC, Ortiz-Hidalgo C, Falzon MR, et al. Helicobacter pylori-associated gastritis and primary B-cell gastric lymphoma. Lancet 1991;338:1175–1176.
Doglioni C, Wotherspoon AC, Moschini A, et al. High incidence of primary gastric lymphoma in northeastern Italy. Lancet 1992;339:834–835.
Ko GH, Park CK, Choi CS, et al. Helicobacter pylori infection and histopathological features of gastric mucosa. Korean J Pathol 1996;30:199–209.
Isaacson PG . Gastrointestinal lymphoma. Hum Pathol 1994;25:1020–1029.
Isaacson PG, Berger F, Muller-Hermelink HK, et al. Extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma). In: Jaffe ES, Harris NL, Stein H, Vardiman JW (eds). World Health Organization Classification of Tumours; Tumours of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press, 2001, pp 157–160.
Dixon MF, Genta RM, Yardley JH, et al. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol 1996;20:1161–1181.
Musshoff K . Clinical staging classification of non-Hodgkin's lymphomas. Straklentherapie 1977;153:218–221.
Nuovo GJ, Plaia TW, Belinsky SA, et al. In situ detection of the hypermethylation-induced inactivation of the p16 gene as an early event in oncogenesis. Proc Natl Acad Sci USA 1999;96:12754–12759.
Wotherspoon AC, Doglioni C, Diss TC, et al. Regression of primary low-grade B cell gastric lymphoma of mucosa-associated lymphoid tissue type after eradication of Helicobacter pylori. Lancet 1993;342:575–577.
Roggero E, Zucca E, Pinotti G, et al. Eradication of Helicobacter pylori infection in primary low-grade gastric lymphoma of mucosa-accociated lymphoid tissue. Ann Intern Med 1995;122:767–769.
O’Rourke EJ, Chevalier C, Pinto AV, et al. Pathogen DNA as target for host-generated oxidative stress: role for repair of bacterial DNA damage in Helicobacter pylori colonization. Proc Natl Acad Sci USA 2003;100:2789–2794.
Belinsky SA, Nikula KJ, Palmisano WA, et al. Aberrant methylation of p16INK4a is an early event in lung cancer and a potential biomarker for early diagnosis. Proc Natl Acad Sci USA 1998;95:11891–11896.
Acknowledgements
This work was supported by a Grant 2004007046 from The Korea Science and Engineering Foundation to Kim KM and Cancer Center of Catholic Medical Center (2003) to Min KO.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Min, K., Seo, E., Kwon, H. et al. Methylation of p16INK4A and p57KIP2 are involved in the development and progression of gastric MALT lymphomas. Mod Pathol 19, 141–148 (2006). https://doi.org/10.1038/modpathol.3800505
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800505
Keywords
This article is cited by
-
RETRACTED ARTICLE: Role of p16 gene promoter methylation in gastric carcinogenesis: a meta-analysis
Molecular Biology Reports (2014)
-
Gastrointestinal lymphomas in a North American population: clinicopathologic features from one major Central-Midwestern United States tertiary care medical center
Diagnostic Pathology (2012)
-
Promoter hypermethylation of p73 and p53 genes in cervical cancer patients among north Indian population
Molecular Biology Reports (2012)
-
Methylation and API2/MALT1 fusion in colorectal extranodal marginal zone lymphoma
Modern Pathology (2009)