Abstract
Inflammatory myofibroblastic tumours (IMFT) may arise at any anatomical site, including lung, soft tissues, retroperitoneum and bladder. Although morphologically similar, these lesions encompass a spectrum of entities with differing aetiology, ranging from reactive/regenerative proliferations to low-grade neoplasms with a risk of local recurrence, but no significant metastatic potential. Vesical IMFT usually presents as a polypoid mass with a pale firm cut surface and can be of considerable size, mimicking a malignant tumour clinically and radiologically. Its good outcome, however, warrants conservative surgical excision, emphasising the importance of identification and distinction from malignant tumours of the bladder that may require more radical surgery and/or adjuvant therapy. We conducted a preliminary retrospective, comparative immunocytochemical study of 20 bladder tumours, including nine IMFTs, five spindle cell (sarcomatoid) carcinomas, two rhabdomyosarcomas, two leiomyosarcomas and two neurofibromas. The results confirmed IMFT positivity for smooth muscle actin, desmin and cytokeratin in 78–89% cases, resulting in potential confusion with sarcomatoid carcinoma or leiomyosarcoma. In contrast, cytoplasmic anaplastic lymphoma kinase (ALK 1) staining was present in eight IMFT (89%), but was not seen in any other lesion examined. The ALK 1 staining was confirmed by fluorescence in situ hybridisation, with translocation of the ALK gene present in 15–60% tumour cells in four of six IMFT examined, but not in four cases of sarcomatoid carcinoma or three of leiomyosarcoma. In conclusion, ALK 1 staining may be of value in the distinction of vesical IMFT from morphologically similar entities, and often reflects ALK gene translocations in these lesions.
Similar content being viewed by others
Main
Attributed initially to the lung,1 inflammatory myofibroblastic tumours (IMFT) are now known to arise in a wide variety of body sites, including the abdominal cavity, retroperitoneum, pelvis and bladder.2, 3, 4, 5 In the early literature, these lesions were classified into separate groups based on their anatomical site and postulated aetiology, using terms such as ‘inflammatory pseudotumour’, ‘inflammatory fibromyxoid tumour’, ‘postoperative spindle cell nodule’ and ‘plasma cell granuloma’.6, 7, 8More recently, IMFT has been widely accepted as the terminology for a subset of these lesions that are thought to be neoplastic in nature, while other cases thought to be infective, inflammatory or reactive in origin are called ‘pseudotumours’.9
Studies of IMFT at a variety of anatomical sites have shown that these tumours present most frequently in children and young adults (mean age 9–10 years), with rare cases in middle age.10, 11 They arise slightly more commonly in females (M:F=3 : 4) and may reach up to 17 cm in size (mean 6 cm). Clinical presentation is usually as a mass lesion, with symptoms of pain, fever and weight loss and haematological findings of anaemia, thrombocytosis, increased erythrocyte sedimentation rate (ESR) and raised gamma globulins.12
Three main microscopic patterns are recognised in IMFT: (1) loose stellate cells with a myxoid background including scattered inflammatory cells (nodular fasciitis-like), (2) spindle cells with a compact fascicular pattern, and (3) sparsely cellular collagenised area, and these may be present within the same case.11
As a result of endoscopic and microscopic features, including size, presence of atypical spindle cells, scattered mitoses and an ‘infiltrative’ margin, these entities may be mistaken for malignant spindle cell lesions and the differentials considered vary with the site of the lesion.
In the bladder, the main differential diagnoses for IMFT are spindle cell carcinoma, leiomyosarcoma, neurofibroma13 and, particularly in adolescents or young children, rhabdomyosarcoma. Although, these tumours are classically said to be distinguishable on morphological grounds (Table 1), the features enabling distinction may be focal and thus not apparent throughout the tumour, or may be partially obscured by a range of inflammatory and reactive infiltrates or by artefacts of processing or fixation.
Immunohistochemistry may be helpful in the differential diagnosis of spindle cell lesions of the bladder, with each entity showing a different immunophenotypic profile (Table 1). However, there is a wide spectrum of possible immunophenotypes for each entity and an individual case may not show the expected ‘classical’ immunoprofile. For instance, immunocytochemical studies in recent years have shown that IMFT are strongly positive (usually >90% cases) for vimentin and smooth muscle actin (SMA), and variably positive for desmin (0–69%), cytokeratin (20–40%), macrophage marker (KP 1) and CD30, raising the possibility of confusion with spindle cell carcinoma or sarcoma.2, 10, 14, 15
More recently, the presence of anaplastic lymphoma kinase (ALK 1) staining in some cases has been postulated to be of value in the confirmation of IMFT and in its distinction from other entities. A series of studies examining the expression of ALK 1 protein expression by immunocytochemistry or ALK gene rearrangements by fluorescence in situ hybridisation (FISH) in IMFT is shown in Table 2.5, 11, 16, 17, 18, 19
The presence of ALK gene translocations involving the 2p23 band, and the subsequent identification of ALK fusion proteins with the protein products of the tropomyosin (TPM3, 4) and clathrin heavy chain genes (CLTC) in a proportion of IMFT, is further evidence that a subset of these entities is indeed neoplastic.20, 21, 22
The favourable outcome of IMFT in the lung and bladder23 is well known, however, and even in a wide range of anatomical sites examined, there is a low rate of local recurrence (25%) and no metastases reported (Table 3).2, 10, 15 This indicates that vesical IMFT may be treated with conservative surgical excision compared to that performed with sarcomatous lesions, which may resemble IMFT clinically and radiologically.
We performed a preliminary retrospective immunocytochemical study on nine vesical IMFT and 11 other spindle cell lesions of the bladder, with the aim of assessing ALK1 staining and its value in the distinction of IMFT from other vesical lesions with a similar morphology. Some of the cases were further examined for ALK gene translocations using FISH, and the results were compared with the immunocytochemical expression of the ALK protein.
Materials and methods
Case Selection
Nine cases of vesical IMFT were retrieved from the archives of the histopathology departments involved in the project. A number of other vesical spindle cell lesions, including five spindle cell carcinomas (carcinosarcomas), two leiomyosarcomas, two rhabdomyosarcomas and two neurofibromas were also selected for comparison. The ages and clinical features of the nine vesical IMFT cases are presented in Table 4.
The slides were reviewed by AF and MCP, and one representative block was chosen per case for immunocytochemical examination, including normal urothelium and muscularis propria as internal controls where possible. Serial sections (5 μm) were cut and stained for a pancytokeratin (CAM 5.2 or MNF116), SMA, desmin, ALK 1, calponin and caldesmon and myogenin for the rhabdomyosarcomas, as shown in Table 5.
Immunocytochemical Method
The slides were dewaxed in xylene, rehydrated through graded alcohols and rinsed in water. Antigen retrieval was performed using microwaving or enzyme digestion techniques as shown in Table 4. Endogenous peroxidase activity was blocked for 10 min with DAKO peroxidase block (Catalogue Number S2023). Primary antibodies were then applied for 1 h at room temperature. The stain was amplified using the DAKO ChemMate Peroxidase/DAB kit (Catalogue Number K5001) according to the manufacturer's guidelines. The slides were finally washed in water, taken through graded alcohols and xylene and coverslips applied.
Interpretation of Staining
The staining pattern of the slides was examined using a semiquantitative scoring method by AF and MCP. The slide was scored as ‘positive’ or ‘negative’ according to the pattern of staining seen and the extent quantitated as ‘focal’ or ‘diffuse’. The term ‘focal’ was used to indicate a patchy staining pattern, with <25% of all cells staining with the applied marker, while the term ‘diffuse’ was used to indicate strong staining in over 25% of cells. The entire slide was examined in an attempt to account for tumour heterogeneity. Positive controls were examined with each batch of stained slides for standardisation of scoring, in addition to the urothelium and muscularis internal tissue controls, and negative controls were performed by omitting the primary antibody.
FISH Protocol
Paraffin-embedded tissue sections were processed using the Vysis Paraffin Pretreatment Kit protocol, substituting xylene in place of the Hemo-De clearing agent. Protease times used were between 30 and 100 min. Slides were denatured in 70% formamide/2 × SSC at 75°C for 6 min followed by dehydration in an ethanol series. FISH was performed with the Vysis LSI ALK probe according to the manufacturer's instructions (Vysis). The probe set contains a 250 bp probe telomeric to the breakpoint region, labelled in Spectrum Orange, and a 300 bp probe centromeric to the breakpoint region, labelled in Spectrum Green. Slides were examined using the BX50 Olympus microscope, equipped with appropriate filters. At least 150 interphases were examined per slide and the percentage of cells showing separation of the probes, reflecting ALK gene translocation, was calculated.
When hybridised with the LSI ALK Dual Color, Break Apart Rearrangement Probe, the 2p23 ALK region in its native state will be seen as two immediately adjacent or fused orange/green (yellow) signals (Figure 2b). However, if a t(2;5) or other chromosome rearrangement at the 2p23 ALK breakpoint region has occurred, one orange and one green signal will be seen, while the native ALK region will remain as an orange/green fusion signal (Figure 2a). The hybridisation result of the LSI ALK Dual Color, Break Apart Rearrangement Probe containing the t(2;5) will be the centromeric green probe remaining at 2p23, while the telomeric orange signal that covers the region is translocated to 5q35 on the derivative chromosome.
(a) FISH studies confirm the presence of ALK gene rearrangement in the cells of an IMFT. There is displacement of the green and orange probes against the centromeric and telomeric portion of the common breakpoint region of the ALK gene onto different chromosomes at one allele (white arrow). This shows that part of the ALK gene is translocated from Chromosome 2p onto Chromosome 5. (b) Cells of a leiomyosarcoma show an intact ALK gene at both alleles, with the green and orange probes adjacent to each other (white arrow). This finding confirms that the centromeric and telomeric portion of the ALK gene are on the same chromosome, excluding the presence of a gene translocation at this point.
Results
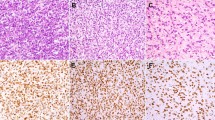
Immunocytochemistry (Table 6, Figure 1)
An IMFT showing compact fascicles of spindle cells, with an inflammatory component of (a) × 200 magnification (b) and × 400 magnification. (c) Areas of looser spindled cells in a myxoid background are focally seen (× 400 magnification). (d) The ALK 1 immunostain shows diffuse cytoplasmic positivity (× 400 magnification) in the neoplastic cells.
A positive cytokeratin immunostain was seen in 8/9 (89%) IMFT, all five spindle cell carcinomas (100%) and 1/2 (50%) leiomyosarcomas. SMA was positive in 8/9 (89%) IMFT, 4/5 (80%) spindle cell carcinomas and 2/2 (100%) leiomyosarcomas (Figure 1, Table 1). In contrast, desmin was positive in 7/9 (78%) IMFT, 2/5 (40%) spindle cell carcinomas and 2/2 (100%) rhabdomyosarcomas, but negative in leiomyosarcoma.
Calponin and caldesmon, novel markers for smooth muscle tumours, were positive in 2/2 (100%) leiomyosarcomas, and but also in 8/9 (89%) and 6/9 (67%) IMFT, respectively, indicating their presence in myofibroblasts as well as smooth muscle fibres.
ALK 1 staining revealed a cytoplasmic pattern of staining in 8/9 IMFT (89%), but was not seen in any of the spindle cell carcinomas, leiomyosarcomas or rhabdomyosarcomas examined.
FISH Results
Six IMFT, four sarcomatoid carcinomas and three leiomyosarcomas were examined for ALK gene rearrangements, using the commercial probe kit from Vysis as explained above.
Four of six IMFT (67%) showed ALK gene rearrangements, as characterised by separation of the centromeric and telomeric probes on either side of the common ALK breakpoint region onto different chromosomes, in a range of between 15 and 60% of the tumour cells examined. In contrast, the sarcomatoid carcinomas and leiomyosarcomas examined were completely negative for ALK translocations (Figure 2).
One of the IMFT cases examined revealed an inversion of Chromosome 2 (inv. 2 p23q35) resulting in a variant fusion protein between the ALK gene and ATIC gene. This translocation has previously been reported in anaplastic large cell lymphoma but not, as far as we are aware, in IMFT.
Discussion
IMFT are a group of lesions that arise in a variety of anatomical sites, and are postulated to be aetiologically composed of a variety of entities that may be either neoplastic or inflammatory in nature. The favourable outcome of most of these lesions, particularly in the lung and bladder is well known, and has implications for initial surgical resection and follow-up. However, the clinical and radiological similarity of vesical IMFT to sarcomas and spindle cell carcinomas may cause a potential diagnostic problem.
At present, the morphological criteria suggestive of IMFT include a variably cellular spindle cell tumour composed of reactive myofibroblastic cells with elongated nuclei, and the presence of stellate cells, with scattered mitoses but no significant atypia or necrosis. These features are not totally specific to IMFT and may be focally seen in some sarcomas. Electron microscopy is also not definitive, although quantitative differences in cytoskeletal filaments between smooth muscle cells and myofibroblasts may be useful in some cases, and the presence of a fibronexus junction, although rarely visualised by electron microscopy, is also indicative of a myofibroblastic phenotype.24, 25
Immunocytochemistry using conventional markers for smooth muscle and epithelial markers has been shown to be positive in a proportion of IMFT, making the evaluation of positive staining for SMA, desmin and cytokeratin unreliable in their distinction from leiomyosarcoma and carcinoma with spindle cell change.
In recent years, ALK 1 has been found to be expressed in the cytoplasm of IMFT in a variable proportion of cases. Studies of ALK 1 protein using immunohistochemical methods have shown positive staining in a range of 0–75% of cases, with high rates of expression in vesical IMFT (33–75%), and complete absence in IMFT of the spleen and lymph nodes.5, 11, 16, 17, 18
The results of our preliminary study using nine vesical IMFT support these findings, with ALK 1 protein immunocytochemically identifiable in eight vesical IMFT (89%), but negative in all 11 sarcomatoid carcinoma, leiomyosarcoma, rhabdomyosarcoma and neurofibromas examined from the bladder. Our results agree with a recent abstract that showed cytoplasmic ALK 1 staining in 12/16 (75%) IMFT, but not in 15 control cases, including leiomyosarcoma, stromal tumours and carcinosarcomas.26
Although a number of cases of rhabdomyosarcoma may show expression of ALK 1, particularly those of alveolar subtype,27 these lesions usually also express desmin and myogenin or myoD1, and show characteristic morphological features that allow identification and distinction from IMFT.
The immunocytochemical ALK 1 protein expression is reflected in the results of the FISH studies. ALK gene translocations were present in a percentage of tumour cells in 4/6 IMFT examined (67%), indicating that a genetic abnormality in the area of Chr. 2p23 often results in overexpression of the ALK 1 protein, and may be an important factor in tumorigenesis of IMFT. In contrast, none of the seven other bladder spindle cell lesions examined showed translocation in the ALK gene, confirming the negative result by immunocytochemistry. The absence of an identifiable ALK translocation in the remaining two IMFT tested may either reflect the sensitivity of the technique, or be attributed to the fact that the commercial probe used in this experiment only identifies translocations at the most common site of the ALK gene and may not show translocations at other sites. Our finding correlates well with the results of another group, who found 47% ALK gene rearrangement in IMFT,16 but is in contrast with those of a recent abstract that shows 0% ALK gene rearrangement in 23 genitourinary cases studied.19 Although the number of cases examined in each category of our study was small, there is no obvious confounding factor in the series of cases examined, and thus no reason why these results may not be extrapolated to a diagnostic setting.
In summary, ALK 1 protein is often upregulated in vesical IMFT by translocation of the ALK gene and is identifiable by immunocytochemistry, making it a valuable marker in cases of IMFT in the bladder. The expression of ALK is not seen in the majority of cases of sarcomatoid carcinomas, leiomyosarcomas or rhabdomyosarcomas of the bladder. Positive ALK 1 immunocytochemical staining, with the conventional morphological and ultrastructural features in current use, may be of value in the distinction of IMFT from other spindle cell lesions of the bladder. The presence of ALK gene translocation may provide further evidence for a neoplastic rather than a reactive or inflammatory nature for the lesion and may be useful in indicating which lesions may show local recurrence potential.
References
Umiker WO, Iverson L . Postinflammatory ‘tumour’ of the lung. Report of four cases simulating xanthoma, fibroma or plasma cell granuloma. J Thorac Cardiovasc Surg 1954;28:55–63.
Jones EC, Clement PB, Young RH . Inflammatory pseudotumour of the bladder: a clinicopathological, immunohistochemical, ultrastructural and flow cytometric study of 13 cases. Am J Surg Pathol 1993;17:264–274.
Coyne JD, Wilson G, Sandhu D, et al. Inflammatory pseudotumour of the urinary bladder. Histopathology 1991;18:261–264.
Coffin CM, Humphrey PA, Dehner LP . Extrapulmonary myofibroblastic tumour: a clinical and pathologic survey. Semin Diagn Pathol 1998;15:85–101.
Chan JKC, Cheuk W, Shimizu M . Anaplastic lymphoma kinase expression in inflammatory pseudotumour. Am J Surg Pathol 2001;25:761–768.
Jufe R, Molinolo AA, Fefer SA, et al. Plasma cell granuloma of the bladder: a case report. J Urol 1984;131:1175–1176.
Nochomovitz LE, Orenstein JH . Inflammatory pseudotumour of the urinary bladder — possible relationship to nodular fasciitis. Two case reports, cytologic observations and ultrastructural observations. Am J Surg Pathol 1985;9:366–373.
Chan JKC . Inflammatory pseudotumour: a family of lesions of diverse nature and aetiologies. Adv Anat Pathol 1996;3:156–171.
Arber DA, Kamel OW, van de Rijn M, et al. Frequent presence of the Epstein–Barr virus in inflammatory pseudotumour. 1995;26:1093–1098.
Coffin CM, Watterson J, Priest JR, et al. Extrapulmonary inflammatory myofibroblastic tumour (inflammatory pseudotumour): a clinicopathologic and immunohistochemical study of 84 cases. Am J Surg Pathol 1995;19:859–872.
Cook JR, Dehner LP, Collins MH, et al. Anaplastic lymphoma kinase (ALK) expression in the inflammatory myofibroblastic tumour. Am J Surg Pathol 2001;25:1364–1371.
Petinato G, Manivel JC, de Rosa N, et al. Inflammatory myofibroblastic tumour (plasma cell granuloma): clinicopathologic study of 20 cases with immunohistochemical and ultrastructural observations. Am J Clin Pathol 1990;94:538–546.
Cheng L, Scheithauer BW, Leibovich BC, et al. Neurofibroma of the urinary bladder. Cancer 1999;86:505–513.
Lundgren L, Aldenborg F, Angervall L, et al. Pseudomalignant spindle cell proliferations of the urinary bladder. Human Pathol 1994;25:181–191.
Iczkowski KA, Shanks JH, Gadaleanu V, et al. Inflammatory pseudotumour and sarcoma of urinary bladder: differential diagnosis and outcome in thirty-eight spindle cell neoplasms. Mod Pathol 2001;14:1043–1051.
Coffin CM, Patel A, Perkins S, et al. ALK and p80 expression and chromosomal rearrangements involving 2p23 in inflammatory myofibroblastic tumour. Mod Pathol 2001;14:569–576.
Kutok JL, Pinkus GS, Dorfman DM, et al. Inflammatory pseudotumour of lymph nodes and spleen: an entity biologically distinct from inflammatory myofibroblastic tumour. Human Pathol 2001;32:1382–1387.
Kapusta LR, Weiss MA, Ramsay J, et al. Inflammatory myofibroblastic tumours of the kidney: a clinicopathologic and immunohistochemical study of 12 cases. Am J Surg Pathol 2003;27:658–666.
Hirsch MS, Cin PD, Fletcher CDM . ALK expression in reactive pseudosarcomatous myofibroblastic proliferations of the genitourinary tract, USCAP Meeting Abstr 697: 2003;153A.
Lawrence B, Perez-Atayde A, Hibbard MK, et al. TPM3-ALK and TPM4-ALK oncogenes in inflammatory myofibroblastic tumours. Am J Pathol 2000;157:377–384.
Bridge JA, Kanamori M, Ma Z, et al. Fusion of the ALK gene to the clathrin heavy chain gene CLTC in inflammatory myofibroblastic tumour. Am J Pathol 2001;159:411–415.
Griffin CA, Hawkins AL, Dvorak C, et al. Recurrent involvement of 2p23 in inflammatory myofibroblastic tumours. Cancer res 1999;59:2776–2780.
Colby TV, Koss MN, Travis WD . Tumours of the lower respiratory tract, In: Colby, Koss, Travis (eds). Atlas of Tumour Pathol Armed Forces Institute of Pathology: Washington, 1995, pp 327–341.
Eyden B . The fibronexus in reactive and tumoral myofibroblasts: further characterisation by electron microscopy. Histol Histopathol 2001;16:57–70.
Eyden B . Electron microscopy in the study of myofibroblastic lesions. Semin Diagn Pathol 2003;20:13–24.
Tsuzuki T, Magi-Galluzzi C, Epstein JI . ALK 1 expression in pseudosarcomatous myofibroblastic tumor (PSFMT) of the urinary bladder, USCAP Meeting Abstr 795: 2003;p174A.
Pillay K, Govender G, Chetty R . ALK protein expression in rhabdomyosarcomas. Histopathology 2002;41:461–467.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Freeman, A., Geddes, N., Munson, P. et al. Anaplastic lymphoma kinase (ALK 1) staining and molecular analysis in inflammatory myofibroblastic tumours of the bladder: a preliminary clinicopathological study of nine cases and review of the literature. Mod Pathol 17, 765–771 (2004). https://doi.org/10.1038/modpathol.3800078
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800078
Keywords
This article is cited by
-
Intra-abdominal Inflammatory Myofibroblastic Tumour (IMFT)—Uncommon Entity
Indian Journal of Surgical Oncology (2024)
-
Inflammatory myofibroblastic tumor with extensive involvement of the bladder in an adolescent: a case report
World Journal of Surgical Oncology (2013)
-
Pseudosarcomatous myofibroblastic lesion of the urinary bladder: A rare entity posing a diagnostic challenge and therapeutic dilemma
Diagnostic Pathology (2008)
-
Pathological classification of hepatic inflammatory pseudotumor with respect to IgG4-related disease
Modern Pathology (2007)