Abstract
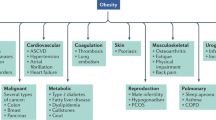
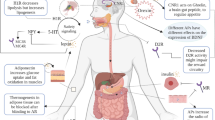
When prevention fails, medicinal treatment of obesity may become a necessity. Any strategic medicinal development must recognize that obesity is a chronic, stigmatized and costly disease that is increasing in prevalence. Because obesity can rarely be cured, treatment strategies are effective only as long as they are used, and combined therapy may be more effective than monotherapy. For a drug to have significant impact on body weight it must ultimately reduce energy intake, increase energy expenditure, or both. Currently approved drugs for long-term treatment of obesity include sibutramine, which inhibits food intake, and orlistat, which blocks fat digestion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
World Health Organization. Obesity: Preventing and Managing the Global Epidemic. (World Health Organization, Geneva, 1998).
National Heart Lung and Blood Institute. Clinical guidelines on the identification evaluation and treatment of overweight and obesity in adults — the evidence report. Obes. Res. 6 (Suppl. 2), 51S–290S ( 1998).
Allison D. B. et al. Annual deaths attributable to obesity in the United States . J. Am. Med. Assoc. 282, 1530– 1538 (1999).
Bray, G. A. Drug treatment of obesity. Don't throw out the baby with the bath water. Am. J. Clin. Nutr. 67, 1–2 (1998).
Bray, G. A. Current and Contemporary Management of Obesity (Handbooks in Health Care, Newtown, PA, 1998).
Bray, G. A. & Greenway, F. L. A review of current and potential drugs for treatment of obesity. Endocr. Rev. 20, 805–875 (1999).
Jequier, E. & Tappy, L. Regulation of body weight in humans . Physiol. Rev. 79, 451– 480 (1999).
Rolls, B. J., Shide, D. J., Thorwart, M. L. & Ulbrecht, J. S. Sibutramine reduces food intake in non-dieting women with obesity. Obes. Res. 6, 1–11 ( 1998).
Hansen, D. L. et al. Thermogenic effects of sibutramine in humans. Am. J. Clin. Nutr. 68, 1180–1186 (1998).
Seagle, H. M., Gessesen, D. H. & Hill, J. O. Effects of sibutramine on resting metabolic rate and weight loss in overweight women. Obes. Res. 6, 115–121 (1998).
Bray, G. A. et al. Sibutramine produces dose-related weight loss. Obes. Res. 7, 189–198 ( 1999).
Apfelbaum, M. et al. Long-term maintenance of weight loss after a very low calorie diet: efficacy and tolerability of sibutramine. Am. J. Med. 106, 179–184 (1999).
Guercolini, R. Mode of action of orlistat. Int. J. Obes. Relat. Metab. Disord . 21, S12–S23 ( 1997).
Hauptman, J. B., Jeunet, F. S. & Hartmann, D. Initial studies in humans with the novel gastrointestinal lipase inhibitor Ro 18-0647 (tetrahydrolipstatin). Am. J. Clin. Nutr. 55, 309S–313S ( 1992).
Sjostrom, L. et al. Randomized placebo-controlled trial of orlistat for weight loss and prevention of weight regain in obese patients. European Multicentre Orlistat Study Group. Lancet 352, 167– 172 (1998).
Davidson, M. H., Hauptman, J. & DiGirolamo, M. Weight control and risk factor reduction in obese subjects treated for 2 years with orlistat. A randomized controlled trial. J. Am. Med. Assoc. 281, 235–242 (1999).
Hollander, P. et al. Role of orlistat in the treatment of obese patients with type 2 diabetes. Diabetes Care 21, 1288– 1294 (1998).
Hill, J. O. et al. Orlistat, a lipase inhibitor, for weight maintenance after conventional dieting — A 1 year study. Am. J. Clin. Nutr. 9, 1108–1116 ( 1999).
Conavatchel, W. Long-term tolerability profile of orlistat, an intestinal lipase inhibitor . Diabetologia 40, A196 ( 1997).
Astrup, A., Lundsgaard, C., Madsen, J., & Christensen, N. J. Enhanced thermogenic responsiveness during chronic ephedrine treatment in man. Am. J. Clin. Nutr. 42, 83– 94 (1985).
Astrup, A., Breum, L., Toubro, S., Hein, P. & Quaade, F. The effect and safety of an ephedrine/caffeine compound compared to ephedrine, caffeine and placebo in obese subjects on an energy-restricted diet. A double-blind trial. Int. J. Obes. Relat. Metab. Disord. 16, 269–277 ( 1992).
Toubro, S., Astrup, L., Breum, L. & Quaade, F. The acute and chronic effects of ephedrine/caffeine mixtures on energy expenditure and glucose metabolism in humans. Int. J. Obes. Relat. Metab. Disord. 17, S73–S77 (1993).
Astrup, A. et al. The effect of ephedrine/caffeine mixture on energy expenditure and body composition in obese women. Metabolism 41, 686–688 (1992).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue . Nature 372, 425–432 (1994).
Tartaglia, L. A. et al. Identification and expression cloning of a leptin receptor OB-R. Cell 83, 1263–1271 (1995).
Chen, H. et al. Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell 84, 491–495 ( 1996).
Lee, G. H. et al. Abnormal splicing of the leptin receptor in diabetic mice . Nature 379, 632–635 (1996).
Chua, S. C. Jr et al. Phenotypes of mouse diabetes and rat fat due to mutations in the OB (leptin) receptor. Science 271, 994–996 (1996).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131– 141 (1997).
Smith, G. P. Satiation: From Gut to Brain 291 (Oxford Univ. Press, New York, 1998).
Gutzwiller J. P. et al. Effect of intravenous human gastrin-releasing peptide on food intake in humans. Gastroenterology 106, 1168–1173 (1994).
Flint, A., Raben, A. l., Astrup, A. & Holst, J. J. Glucagon-like peptide I promotes satiety and suppresses energy intake in humans. J. Clin. Invest. 101, 515–520 (1998).
Holst, J. J. Glucagon-like peptide-1 (GLP-1) — an intestinal hormone, signaling nutritional abundance, with an unusual therapeutic potential. Trends Endocrinol. Metab. 10, 229–235 ( 1999).
Gutzwiller, J. P. et al. Glucagon-like peptide-1 promotes satiety and reduces food intake in patients with diabetes mellitus type 2. Am. J. Physiol. 276 (5 Pt 2), R1541–R1544 (1999).
Erlanson-Albertsson, C. & York, D. Enterostatin – a peptide regulating fat intake. Obes. Res. 5, 360–372 (1997).
Smeets, M., Geiselman, P., Bray, G. A. & York, D. A. The effect of oral enterostatin on hunger and food intake in human volunteers . FASEB J. 13, A871 ( 1999).
Wetherford, S. C. et al. Intraventricular administration of enterostatin decreases food intake in baboons. Appetite 19, 225 (1992).
Morley, J. E., Flood, J. F., Horowitz, M., Morley, P. M. & Walter, M. J. Modulation of food intake by peripherally administered amylin. Am. J. Physiol. 276 (1 Pt 2), R178–R184 (1994 ).
Nagase, H., Bray, G. A. & York, D. A. Effects of pyruvate and lactate on food intake in rat strains sensitive and resistant to dietary obesity. Physiol. Behav. 59, 555–560 ( 1996).
Scharrer, E. Control of food intake by fatty acid oxidation and ketogenesis. Nutrition 15, 704–714 ( 1999).
Sullivan, A. C., Triscari, J., Hamilton, J. G. & Miller, O. N. Effect of (–)-hydroxycitrate upon the accumulation of lipid in the rat. II. Appetite. Lipids 9, 129– 134 (1974).
Heymsfield, S. B. et al. Garcinia cambogia (hydroxycitric acid) as a potential antiobesity agent: a randomized controlled trial. J. Am. Med. Assoc. 280, 1596–1600 (1998).
Gietzen, D. W., Erecius, L. F. & Rogers, Q. R. Neurochemical changes after imbalanced diets suggest a brain circuit mediating anorectic responses to amino acid deficiency in rats. J. Nutr. 128, 771– 781 (1998).
Tso, P., Liu, M., & Kalogeris, T. J. The role of apolipoprotein A-IV in food intake regulation . J. Nutr. 8,1503–1506 (1999).
Mantzoros, C. S. The role of leptin in human obesity and disease — a review of current evidence. Ann. Intern. Med. 130, 671– 680 (1999).
Cone, R. D. The central melanocortin system and energy homeostasis. Trends Endocrinol. Metab. 10, 211–216 (1999).
Wilson, B. D., Ollmann, M. M. & Barsh, G. S. The role of agouti-related protein in regulating body-weight. Mol. Med. Today 5, 250– 256 (1999).
Kristensen, P. et al. Hypothalamic CART is a new anorectic peptide regulated by leptin. Nature 393, 72– 76 (1998).
Heymsfield, S. B. et al. Recombinant leptin for weight loss in obese and lean adults: a randomized, controlled, dose-escalation study. J. Am. Med. Assoc. 282, 1568–1575.
Farooqi, I. S. et al. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N. Engl. J. Med. 341, 879–884 (1999).
Ogawa, Y. et al. Increased glucose metabolism and insulin sensitivity in transgenic skinny mice overexpressing leptin. Diabetes 48, 1822–1829 (1999).
Bjorbaek, C. et al. Activation of SOCS-3 messenger ribonucleic acid in the hypothalamus by ciliary neurotrophic factor. Endocrinology 140, 2035–2043 (1999).
Zimanyi, I. A., Fathi, Z. & Poindexter, G. S. Central control of feeding behavior by neuropeptide Y. Curr. Pharm. Des. 4, 349– 366 (1998).
Palmiter, R. D. et al. in Pennington Center Nutrition Series: Nutrition, Genetics, and Obesity 269–286 (Louisiana State Univ. Press, Baton Rouge, 1999).
Yaswen, L., Diehl, N., Brennan, M. B. & Hochgeschwender, U. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nature Med. 5, 1066–1070 (1999).
Chambers, J. et al. Melanin-concentrating hormone is the cognate ligand for the orphan G-protein-coupled receptor SLC-1. Nature 400 , 261–269 (1999).
Saito, Y. et al. Molecular characterization of the melanin-concentrating-hormone receptor. Nature 400, 265– 269 (1999).
Shimada, M. et al. Mice lacking melanin-concentrating hormone are hypophagic and lean. Nature 396, 670– 673 (1998).
Lembo, P. M. et al. The receptor for the orexigenic peptide melanin-concentrating hormone is a G-protein-coupled receptor. Nature Cell Biol. 5, 267–271 (1999).
Barton, C., York, D. A. & Bray, G. A. Opioid receptor subtype control of galinin-induced feeding . Peptides 17, 237–240 (1996).
Rokaeus, A., Jiang, K., Spyrou, G. & Waschek, J. A. Transcriptional control of the galanin gene. Tissue-specific expression and induction by NGF, protein kinase C, and estrogen. Ann. NY Acad. Sci. 863, 1–13 (1998).
Sakurai, T. et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92, 573–585 ( 1998).
Lin, L. et al. The sleep disorder canine narcolepsy is caused by a mutation in the hypocretin (Orexin) Receptor 2 gene. Cell 98, 365–376 (1999).
Chemelli, R. M. et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 98, 437– 451 (1999).
Kalra, S. P. et al. Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr. Rev. 1, 68–100 (1999).
Karolyi, I. J. et al. Altered anxiety and weight gain in corticotropin-releasing-hormone-binding protein-deficient mice. Proc. Natl Acad. Sci. USA 96 , 11595–11600 (1999).
Vickers, S. P., Clifton, P. G., Dourish, C. T. & Tecott, L. H. Reduced satiating effect of d-fenfluramine in serotonin 5-HT (2C) receptor mutant mice. Psychopharmacology 143, 309 –314 (1999).
Smith, B. K., York, D. A. & Bray, G. A. Activation of hypothalamic serotonin receptors reduced intake of dietary fat and protein but not carbohydrate. Am. J. Physiol. 277, R802–R811 (1999).
Connolly, H. M. et al. Valvular heart disease associated with fenfluramine-phentermine . N. Engl. J. Med. 337, 581– 588 (1997).
Ryan, D. H. et al. Serial echocardiographic and clinical evaluation of valvular regurgitation before, during, and after treatment with fenfluramine or dexfenfluramine and mazindol or phentermine. Obes. Res. 7, 313–322 (1999).
Rothman, R. B., Ayestas, M. A., Dersch, C. M., & Baumann, M. H. Aminorex, fenfluramine, and chlorphentermine are serotonin transporter substrates. Implications for primary pulmonary hypertension. Circulation 100, 869–875 (1999).
Heinonen, P. et al. Identification of a three-amino acid deletion in the alpha2B-adrenergic receptor that is associated with reduced basal metabolic rate in obese subjects . J. Clin. Endocrinol. Metab. 84, 2429– 2433 (1999).
Terry, P., Gilbert, D. B. & Cooper, S. J. Dopamine receptor subtype agonists and feeding behavior . Obes. Res. 3, 515S ( 1995).
Sakata, S., Yoshimatsu, H. & Kurokawa, M. Hypothalamic neuronal histamine: implications of its homeostatic control of energy metabolism. Nutrition 13, 403–411 (1997).
Lovenberg, T. W. et al. Cloning and functional expression of the human histamine H3 receptor. Mol. Pharmacol. 55, 1101– 1107 (1999).
Kurose, Y. & Terashima, Y. Histamine regulates food intake through modulating noradrenaline release in the para-ventricular nucleus. Brain Res. 15, 115–118 ( 1999).
Stahl, A. et al. Identification of the major intestinal fatty acid transport protein . Mol. Cell 4, 299–308 (1999).
Bray, G. A. The Obese Patient (Saunders, Philadelphia, 1976).
Danforth, E. Jr & Himms-Hagen, J. H. Obesity and diabetes and the beta-3 adrenergic receptor. Eur. J. Endocrinol. 136, 362–365 ( 1997).
Klingenberg, M. & Huang, S. G. Structure and function of the uncoupling protein from brown adipose tissue. Biochim. Biophys. Acta 8, 271–296 (1999).
Fleury, C. et al. Uncoupling protein-2: a novel gene linked to obesity and hyperinsulinemia . Nature Genet. 15, 269– 272 (1997).
Gimeno, R. E. et al. Cloning and characterization of an uncoupling protein homolog: a potential molecular mediator of human thermogenesis. Diabetes 46, 900–906 ( 1997).
Vidal-Puig, A., Solanes, G., Grujic, D., Flier, J. S. & Lowell, B. B. UCP3: an uncoupling protein homologue expressed preferentially and abundantly in skeletal muscle and brown adipose tissue. Biochem. Biophys. Res. Commun. 235, 79–82 (1997).
Boss, O. et al. Uncoupling protein-3: a new member of the mitochondrial carrier family with tissue-specific expression. FEBS Lett. 408, 39–42 (1997).
Gong, D. W., He, Y., Karas, M. & Reitman, M. Uncoupling protein-3 is a mediator of thermogenesis regulated by thyroid hormone, beta3-adrenergic agonists, and leptin. J. Biol. Chem. 39, 24129–24132 (1997).
Elchebly, M. et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 283, 1544–1548 (1999).
Cases, S. et al. Identification of a gene encoding an acyl CoA: diacylglycerol acyltransferase, a key enzyme in triacyglycerol synthesis. Proc. Natl Acad. Sci. USA 95, 13018–13023 (1998).
Marin, P. et al. Androgen treatment of abdominally obese men. Obes. Res. 1, 245–251 ( 1993).
Marin, P. et al. The effects of testosterone on body composition and metabolism in middle-aged obese men. Int. J. Obes. Relat. Metab. Disord. 16, 991–997 (1992).
Lovejoy, J. C. et al. Oral anabolic steroid treatment, but not parenteral androgen treatment, decreases abdominal fat in obese, older men. Int. J. Obes. Relat. Metab. Disord. 19, 614– 624 (1995).
Kin, K. R. et al. Low-dose growth hormone treatment with diet restriction accelerates body fat loss, exerts anabolic effect and improves growth hormone secretory dysfunction in obese adults. Hormone Res. 51, 78–84 (1999).
Bujalska, I. J., Kumar, S., Hewison, M. & Stewart, P. M. Differentiation of adipose stromal cells: the roles of glucocorticoids and 11 beta-hydroxysteroid dehydrogenase. Endocrinology 140, 3188– 3196 (1999).
Greenway, F. L. & Bray, G. A. Topical fat reduction . Obes. Res. 3 (Suppl. 4), 561S –568S (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bray, G., Tartaglia, L. Medicinal strategies in the treatment of obesity. Nature 404, 672–677 (2000). https://doi.org/10.1038/35007544
Issue Date:
DOI: https://doi.org/10.1038/35007544
This article is cited by
-
Effective weight control via an implanted self-powered vagus nerve stimulation device
Nature Communications (2018)
-
Les traitements médicamenteux de l’obésité
Acta Endoscopica (2017)
-
Osterix represses adipogenesis by negatively regulating PPARγ transcriptional activity
Scientific Reports (2016)
-
Preventive obesity agent montmorillonite adsorbs dietary lipids and enhances lipid excretion from the digestive tract
Scientific Reports (2016)
-
miR-146a-mediated suppression of the inflammatory response in human adipocytes
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.