Abstract
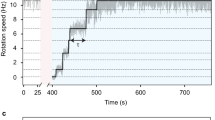
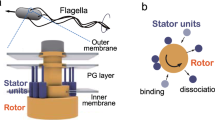
Rotation of the bacterial flagellar motor is driven by an ensemble of torque-generating units containing the proteins MotA and MotB1,2,3. Here, by inducing expression of MotA in motA- cells under conditions of low viscous load, we show that the limiting speed of the motor is independent of the number of units: at vanishing load, one unit turns the motor as rapidly as many. This result indicates that each unit may remain attached to the rotor for most of its mechanochemical cycle, that is, that it has a high duty ratio4. Thus, torque generators behave more like kinesin, the protein that moves vesicles along microtubules, than myosin, the protein that powers muscle. However, their translation rates, stepping frequencies and power outputs are much higher, being greater than 30 µm s-1, 12 kHz and 1.5 × 105 pN nm s-1, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Block, S. M. & Berg, H. C. Successive incorporation of force-generating units in the bacterial rotary motor. Nature 309, 470–473 (1984).
Blair, D. F. & Berg, H. C. Restoration of torque in defective flagellar motors. Science 242, 1678– 1681 (1988).
Samuel, A. D. T. & Berg, H. C. Torque-generating units of the bacterial flagellar motor step independently. Biophys. J. 71, 918–923 ( 1996).
Leibler, S. & Huse, D. A. Porters versus rowers: a unified stochastic model of motor proteins. J. Cell Biol. 121 , 1357–1368 (1993).
Macnab, R. M. in Escherichia coli and Salmonella: Cellular and Molecular Biology Vol. 1 (eds Neidhardt, F. C. et al.) 123– 145 (American Society for Microbiology, Washington, 1996).
Berry, R. M. & Armitage, J. P. The bacterial flagellar motor. Adv. Microb. Physiol. 41, 291– 337 (1999).
Silverman, M. & Simon, M. Flagellar rotation and the mechanism of bacterial motility. Nature 249, 73– 74 (1974).
Gittes, F. & Schmidt, C. F. Interference model for back-focal-plane displacement detection in optical tweezers. Opt. Lett. 23, 7–9 (1998).
Berg, H. C. & Turner, L. Torque generated by the flagellar motor of Escherichia coli. Biophys. J. 65, 2201–2216 (1993).
Chen, X. & Berg, H. C. Torque-speed relationship of the flagellar rotary motor of Escherichia coli. Biophys. J. (in the press).
Block, S. M., Goldstein, L. S. B. & Schnapp, B. J. Bead movement by single kinesin molecules studied with optical tweezers. Nature 348, 348– 352 (1990).
Uyeda, T., Abramson, P. D. & Spudich, J. A. The neck region of the myosin motor domain acts as a lever arm to generate movement. Proc. Natl Acad. Sci. USA 93, 4459–4464 (1996).
Howard, J. Molecular motors: structural adaptations to cellular functions. Nature 389, 561–567 ( 1997).
Macnab, R. M. Bacterial flagella rotating in bundles: a study in helical geometry. Proc. Natl Acad. Sci. USA 74, 221– 225 (1997).
DeRosier, D. J. The turn of the screw: the bacterial flagellar motor. Cell 93, 17–20 (1998).
Muramoto, K. et al. High-speed rotation and speed stability of the sodium-driven flagellar motor in Vibrio alginolyticus. J. Mol. Biol. 251, 50–58 (1995).
Visscher, K., Schnitzer, M. J. & Block, S. M. Single kinesin molecules studied with a molecular force clamp. Nature 400, 184– 189 (1999).
Toyoshima, Y. Y. et al. Myosin subfragment-1 is sufficient to move actin filaments in vitro. Nature 328, 536– 539 (1987).
Yasuda, R., Noji, H., Kinosita, K. J. & Yoshida, M. F1-ATPase is a highly efficient molecular motor that rotates with discrete 120° steps. Cell 93, 1117–1124 (1998).
Silverman, M. & Simon, M. Operon controlling motility and chemotaxis in E. coli. Nature 264, 577– 580 (1976).
Armstrong, J. B. & Adler, J. Genetics of motility in Escherichia coli: complementation of paralyzed mutants. Genetics 56, 363–373 ( 1967).
Fung, D. C. Y. K. Powering the Escherichia coli Flagellar Motor with an External Voltage Source. Thesis, Harvard Univ. (1994).
Maurer, L. & Orndorff, P. E. Identification and characterization of genes determining receptor binding and pilus length of Escherichia coli type 1 pili. J. Bacteriol. 169, 640 –645 (1987).
Kuwajima, G. Flagellin domain that affects H antigenicity of Escherichia coli K-12. J. Bacteriol. 170, 485– 488 (1988).
Scharf, B. E., Fahrner, K. A., Turner, L. & Berg, H. C. Control of direction of flagellar rotation in bacterial chemotaxis. Proc. Natl Acad. Sci. USA 95, 201–206 ( 1998).
Berry, R. M. & Berg, H. C. Absence of a barrier to backwards rotation of the bacterial flagellar motor demonstrated with optical tweezers. Proc. Natl Acad. Sci. USA 94, 14433– 14437 (1997).
Happel, J. & Brenner, H. Low Reynolds Number Hydrodynamics (Kluwer Academic, Drodrecht, 1991).
Berry, R. M. & Berg, H. C. Torque generated by the flagellar motor of Escherichia coli while driven backward. Biophys. J. 76, 580–587 ( 1999).
Block, S. M., Blair, D. F. & Berg, H. C. Compliance of bacterial flagella measured with optical tweezers. Nature 338, 514– 517 (1989).
Block, S. M., Blair, D. F. & Berg, H. C. Compliance of bacterial polyhooks measured with optical tweezers. Cytometry 12, 492– 496 (1991).
Acknowledgements
We thank K. Fahrner and A. Samuel for plasmid and strain constructions, and K. Fahrner, A. Samuel and L. Turner for comments on the manuscript. This work was supported by the Wellcome Trust, the Rowland Institute for Science and by the US NIH.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ryu, W., Berry, R. & Berg, H. Torque-generating units of the flagellar motor of Escherichia coli have a high duty ratio. Nature 403, 444–447 (2000). https://doi.org/10.1038/35000233
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35000233
This article is cited by
-
Probing nanomotion of single bacteria with graphene drums
Nature Nanotechnology (2022)
-
The ecological roles of bacterial chemotaxis
Nature Reviews Microbiology (2022)
-
A multi-state dynamic process confers mechano-adaptation to a biological nanomachine
Nature Communications (2022)
-
Single-proton torque generation of the bacterial flagellar motor
Journal of the Korean Physical Society (2022)
-
Cooperative stator assembly of bacterial flagellar motor mediated by rotation
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.