Abstract
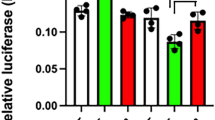
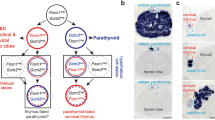
THE pit-1 gene is a member of a large family of genes that encode proteins which are involved in development and which contain a highly homologous region, referred to as the POU domain1–9. Pit-1, a pituitary-specific transcription factor, can activate the transcription of the growth hormone and prolactin promoters1,10. It is expressed in mature thyrotroph, somatotroph and lactotroph cell types11,12 of the anterior pituitary which arise sequentially during development; somatotrophs and lactotrophs, which secrete growth hormone and prolactin, respectively, are the last to arise13. Intriguingly, during ontogeny, pit-1 transcripts are observed in the rat neural tube and neural plate (embryonic day 10–11) and disappear thereafter (day 13), only to reappear exclusively in the anterior lobe of the pituitary gland (day 15) just before activation of prolactin and growth hormone12,14,15. This biphasic pattern suggests a complex mechanism of initial activation of pit-1 gene expression. Transcription and transfection analyses in vitro using wild-type and mutated promoters indicate that Pit-1 can positively autoregulate the expression of the pit-1 promoter as a consequence of binding to two Pit-1-binding elements. Mutation of the 5' Pit-1-binding site abolished positive autoregulation, whereas mutation of the element located immediately 3' of the cap site markedly increased expression of the pit-1 promoter. These data are consistent with a positive, attenuated autoregulatory loop that seems to function in maintaining pit-1 gene expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ingraham, H. A. et al. Cell 55, 519–529 (1988).
Bodner, M. et al. Cell 55, 505–518 (1988).
Ko, H-S., Fast, P., McBride, W. & Staudt, L. M. Cell 55, 135–144 (1988).
Müller, M. M., Rupert, S., Schaffner, W. & Matthias, P. Nature 236, 544–551 (1988).
Clerc, R. G., Corcoran, L. M., LeBowitz, J. H., Baltimore, D. & Sharp, P. A. Genes Dev. 2, 1570–1581 (1988).
Scheidereit, C. et al. Nature 336, 552–557 (1988).
Sturm, R. A., Das, G. & Herr, W. Genes Dev. 2, 1582–1599 (1988).
Finney, M., Ruvkun, G. & Horvitz, H. R. Cell 55, 757–769 (1988).
Herr, W. et al. Genes Dev. 2, 1513–1515 (1988).
Mangalam, H. J. et al. Genes Dev. 3, 946–958 (1989).
Crenshaw III, E. B., Kalla, K., Simmons, D. M., Swanson, L. W. & Rosenfeld, M. G. Genes Dev. 3, 959 (1989).
Simmons, D. W. et al. Genes Dev. 4, 962–979 (1990).
Cooke, N. E., Brook, F. R. & Frawlay, L. L. Endocrinology 117, 187 (1985).
He, X. et al. Nature 340, 35–42 (1989).
Dollé, P. Cell 60, 809–820 (1990).
Nelson, C., Albert, V., Elsholtz, H. P., Lu, L. E.-W. & Rosenfeld, M. G. Science 236, 14000–14405 (1988).
Elsholtz, H. P., Albert, V. R., Treacy, M. N. & Rosenfeld, M. G. Genes Dev. 4, 43–51 (1990).
Montminy, M. R. & Bilezikjian, L. M. Nature 328, 175–178 (1987).
Ingraham, H. A. et al. Cell 61, 1021–1033 (1990).
Krasnow, M. A., Saffman, E. E., Kornfeld, K. & Hogness, D. Cell 57, 1031–1043 (1989).
Biggin, M. D. & Tjian, R. Cell 53, 699–711 (1988).
Kuziora, M. A. & McGinnis Cell 55, 477–485 (1988).
Treisman, J. & Desplan, C. Nature 341, 335–337 (1989).
Thayer, M. J. et al. Cell 58, 241–248 (1989).
Ausubel, F. M. et al. Current Protocols in Molecular Biology, 641–644 (Wiley-Interscience, New York, 1987).
Sanger, F., Nicklen, S. & Carlson, A. Proc. natn. Acad Sci. U.S.A. 74, 5463–5467 (1977).
de Wet, J. R., Wodd, K. V., DeLuca, M., Helinski, D. R. & Subramani, S. Molec. cell. Biol. 7, 725–737 (1987).
Chen, C. & Okayama, H. Molec. Cell. Biol. 7, 2745–2752 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, R., Ingraham, H., Treacy, M. et al. Autoregulation of pit-1 gene expression mediated by two cis-active promoter elements. Nature 346, 583–586 (1990). https://doi.org/10.1038/346583a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/346583a0
This article is cited by
-
Multi-omic profiling of pituitary thyrotropic cells and progenitors
BMC Biology (2021)
-
Analysis of polymorphism within POU1F1 gene in relation to milk production traits in dairy Sarda sheep breed
Molecular Biology Reports (2012)
-
Tumor-associated CpG demethylation augments hypoxia-induced effects by positive autoregulation of HIF-1α
Oncogene (2011)
-
A novel recessive splicing mutation in the POU1F1 gene causing combined pituitary hormone deficiency
Journal of Endocrinological Investigation (2009)
-
Combined effect of mutations of the GH1 gene and its proximal promoter region in a child with growth hormone neurosecretory dysfunction (GHND)
Journal of Molecular Medicine (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.