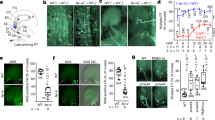
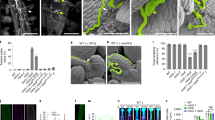
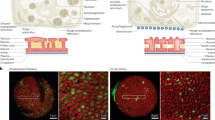
Abstract
Successful pollination and fertilization are absolute requirements for sexual reproduction in higher plants. Pollen hydration, germination and penetration of the stigma by pollen tubes are influenced by the exudate on wet stigmas1 and by the pollen coat in species with dry stigmas2,3,4,5. The exudate allows pollen tubes to grow directly into the stigma, whereas the pollen coat establishes the contact with the stigma. Pollen tubes then grow into the papillae, which are covered by a cuticle. The components of the exudate or pollen coat that are responsible for pollen tube penetration are not known. To discover the role of the exudate, we tested selected compounds for their ability to act as functional substitutes for exudate in the initial stages of pollen-tube growth on transgenic stigmaless tobacco plants1 that did not produce exudate. Here we show that lipids are the essential factor needed for pollen tubes to penetrate the stigma, and that, in the presence of these lipids, pollen tubes will also penetrate leaves. We propose that lipids direct pollen-tube growth by controlling the flow of water to pollen in species with dry and wet stigmas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Goldman, M. H. S., Goldberg, R. B. & Mariani, C. Female sterile tobacco plants are produced by stigma specific cell ablation. EMBO J. 13, 2976–2984 (1994).
Preuss, D., Lemieux, B., Yen, G. & Davis, R. W. Aconditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signalling during fertilization. Genes Dev. 7, 974–985 (1993).
Dickinson, H. G. Pollen dressed for success. Nature 364, 573–574 (1993).
Dickinson, H. G. Self-pollination: simply a social disease? Nature 367, 517–518 (1994).
Taylor, L. P. & Hepler, P. K. Pollen germination and tube growth. Annu. Rev. Plant Physiol. Plant Mol. Biol. 48, 461–491 (1997).
Mariani, C., De Beuckeleer, M., Truettner, J., Leemans, J. & Goldberg, R. B. Induction of male sterility in plants by a chimaeric ribonuclease gene. Nature 347, 737–741 (1990).
Konar, R. N. & Linskens, H. F. Physiology and biochemistry of the stigmatic fluid of Petunia hybrida. Planta 71, 378–387 (1966).
Labarca, C., Kroh, M. & Loewus, F. The composition of stigmatic exudate from Lilium longiflorum. Labelling studies with myo-inositol, d-glucose, and l-proline. Plant Physiol. 46, 150–156 (1970).
Cresti, M., Keijzer, C. J., Tiezzi, A., Ciampolini, F. & Focardi, S. Stigma of Nicotiana: ultrastructural and biochemical studies. Am. J. Bot. 73, 1713–1722 (1986).
Dumas, C. Lipochemistry of the progamic stage of a self-incompatible species: neutral lipids and fatty acids of the secretory stigma during its glandular activity, and the solid style, the ovary and the anther in Forsythia intermedia Zab. (heterostylic species). Planta 137, 177–184 (1977).
Elleman, C. J., Franklin-Tong, V. & Dickinson, H. G. Pollination in species with dry stigma: the nature of the early stigmatic response and the pathway taken by the pollen tubes. New Phytol. 121, 413–424 (1992).
Kandasamy, M. K., Nasrallah, J. B. & Nasrallah, M. E. Pollen pistil interactions and developmental regulation of pollen tube growth in Arabidopsis. Development 120, 3405–3418 (1994).
Evans, D. E., Sang, J. P., Cominos, X., Rothnie, N. E. & Knox, R. B. Astudy of phospholipids and galactolipids in pollen of two lines of Brassica napus L. (rapeseed) with different ratios of linoleic to linolenic acid. Plant Physiol. 92, 418–424 (1990).
Piffanelli, P., Ross, J. H. E. & Murphy, D. J. Intra and extracellular lipid composition and associated gene expression patterns during pollen development in Brassica napus. Plant J. 11, 549–562 (1997).
Dickinson, H. G. Dry stigmas, water and self-incompatability in Brassica. Sex. Plant Reprod. 8, 1–10 (1995).
Elleman, C. J. & Dickinson, H. G. Identification of pollen components regulating pollination-specific responses in the stigmatic papillae of Brassica oleracea. New Phytol. 133, 197–205 (1996).
Wolters-Arts, M., Derksen, J., Kooijman, J. W. & Mariani, C. Stigma development in Nicotiana tabacum. Cell death in transgenic plants as a marker to follow cell fate at high resolution. Sex. Plant Rep. 9, 243–254 (1996).
Hülskamp, M., Kopczak, S. D., Horejsr, T. F., Kihl, B. K. & Pruitt, R. E. Identification of genes required for pollen-stigma recognition in Arabidopsis thaliana. Plant J. 815, 703–714 (1995).
Cheung, A. Y., Zhan, X., Wang, H. & Wu, H. Organ-specific and AGAMOUS-related expression and glycosylation of a pollen tube growth-promoting protein. Proc. Natl Acad. Sci. USA 93, 3853–3858 (1996).
Lolle, S. J. & Cheung, A. Y. Promiscuous germination and growth of wild type pollen from Arabidopsis and related species on the shoot of the Arabidopsis mutant, fiddlehead. Dev. Biol. 155, 250–258 (1993).
Lolle, S. J. et al. Developmental regulation of cell interactions in the Arabidopsis fiddlehead-1 mutant: a role for the epidermal cell wall and cuticle. Dev. Biol. 189, 311–321 (1997).
Boden, N. in Micelles, Membranes, Microemulsions, and Monolayers (eds Gelbart, W. M., Ben-Shaul, A. & Roux, D.) 153–211 (Springer, New York, (1994).
Ikeda, S., Nasrallah, J. B., Dixit, R., Preiss S. & Nasrallah, M. E. An aquaporin like gene required for the Brassica self-incompatability response. Science 276, 1564–1566 (1997).
Pruitt, R. E. Molecular mechanics of smart stigmas. Trends Plant Sci. 2, 328–4329 (1997).
Baier, H. & Bonhoeffer, F. Axon guidance by gradients of a target-derived component. Science 255, 472–475 (1992).
Maret, G. & Dransfeld, K. Biomolecules and polymers in high steady magnetic fields. Top. Appl. Phys. 57, 143–204 (1985).
Malho, R., Feijo, J. A. & Pais, M. S. S. Effect of electrical fields and external ionic currents on pollen tube orientation. Sex. Plant Reprod. 5, 57–63 (1992).
Reger, B. J., Chaubal, R. & Pressey, R. Chemotropic responses by pearl millet pollen tubes. Sex. Plant Reprod. 5, 47–56 (1992).
Robbertse, P. J., Lock, J. J., Stoffberg, E. & Coetzer, L. A. Effect of boron on directionality of pollen tube growth in Petunia and Agapanthus. South African J. Bot. 56, 487–492 (1990).
Hyde, G. J. & Heath, I. B. Ca-2+-dependent polarization of axis establishment in the tip-growing organism, Saprolegnia ferax, by gradients of the ionophore A23187. Eur. J. Cell Biol. 67, 356–362 (1995).
Acknowledgements
We thank A. Pereira for the gift of the cer and popmutants, J. Derksen for reading the manuscript and M.B. van Veen for artwork.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wolters-Arts, M., Lush, W. & Mariani, C. Lipids are required for directional pollen-tube growth. Nature 392, 818–821 (1998). https://doi.org/10.1038/33929
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/33929
This article is cited by
-
The phosphorylated pathway of serine biosynthesis affects sperm, embryo, and sporophyte development, and metabolism in Marchantia polymorpha
Communications Biology (2024)
-
Nitrogen rate effects reproductive development and grain set of summer maize by influencing fatty acid metabolism
Plant and Soil (2023)
-
Identifying key contributing pollen and stigma biochemical traits driving genetic diversity and sexual compatibility in kola [Cola nitida (Vent.) Schott & Endl.]
Euphytica (2023)
-
Biotechnological approaches to overcome hybridization barriers and use of micropropagation tool for further improvement in Heliconia: a review
Plant Cell, Tissue and Organ Culture (PCTOC) (2022)
-
Breeding of ornamentals: success and technological status
The Nucleus (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.