Abstract
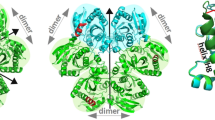
The trp repressor is a small dimeric regulatory protein which controls the expression of three operons in Escherichia coli1–3. The inactive aporepressor protein must bind two molecules of L-tryptophan to form the active repressor4. If desamino analogues of L-tryptophan such as indole propionate (IPA) are substituted for L-tryptophan, an inactive pseudorepressor is formed1,5,6. Because the desamino analogues thus cause derepression of operons under control of the trp repressor, they appear to be 'inducers'. We have determined the crystal structure of the pseudorepressor and refined it to 1.65 Å. The molecular structure was compared to that of the nearly isomorphous orthorhombic form of the repressor. Surprisingly, the indole ring of IPA is in the same position as the indole ring of L-tryptophan in the repressor, but is 'flipped over'. As a result, the carboxyl group of IPA is oriented toward the DNA-binding surface of the protein and is in a position where it sterically and electrostatically repels the phosphate backbone of both operator and non-operator DNA. This explains why IPA acts as an apparent trp inducer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rose, J. K., Squires, C. L., Yanofsky, C., Yang, H.-L. & Zubay, G. Nature new Biol. 245, 133–137 (1973).
Zurawski, G., Gunsalus, R. P., Brown, K. D. & Yanofsky, C. J. molec. Biol. 145, 47–73 (1981).
Kelley, R. L. & Yanofsky, C. Proc. natn. Acad. Sci. U.S.A. 79, 3120–3124 (1982).
Joachimiak, A., Kelley, R. L., Gunsalus, R. P., Yanofsky, C. & Sigler, P. B. Proc. natn. Acad. Sci. U.S.A. 80, 668–672 (1983).
McGeoch, D., McGeoch, J. & Morse, D. Nature new Biol. 245, 137–140 (1973).
Pauley, R. J., Fredericks, W. W. & Smith, O. H. J. Bacterial. 136, 219–226 (1978).
Marmorstein, R. Q., Joachimiak, A., Sprinzl, M. & Sigler, P. B. J. biol. Chem. 262, 4922–4927 (1987).
Joachimiak, A., Schevitz, R. W., Kelley, R. L., Yanofsky, C. & Sigler, P. B. J. biol. Chem. 258, 12641–12643 (1983).
Lane, A. N. Eur. J. Biochem. 157, 405–413 (1986).
Marmorstein, R. Q. & Sigler, P. B. J. biol. Chem. (submitted).
Zhang, R.-G. et al. Nature 327, 591–597 (1987).
Lawson, C. L. et al. Proteins 3, 18–31 (1988).
Jones, T. A. Meth. Enzym. 115, 157–171 (1985).
Finzel, B. C. J. appl. Cryslallogr. 20, 53–55 (1987).
Bradford, M. M. Analyt. Biochem. 72, 248–251 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lawson, C., Siglert, P. The structure of trp pseudorepressor at 1.65Å shows why indole propionate acts as a trp 'inducer'. Nature 333, 869–871 (1988). https://doi.org/10.1038/333869a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/333869a0
This article is cited by
-
An atomic view of the L-tryptophan binding site of trp repressor
Nature Structural Biology (1996)
-
Study ofl-tryptophan corepressor binding to mutatedE. coli tryptophan repressor proteins by optically detected triplet-state magnetic resonance
Journal of Fluorescence (1994)
-
Crystal structure of trp represser/operator complex at atomic resolution
Nature (1988)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.