Abstract
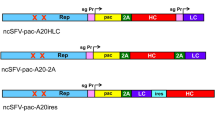
The most advanced oncolytic Newcastle disease virus (NDV) strains that are used in clinical trials for the treatment of cancer are wild-type mesogenic strains. These virus strains have an inherent, nongenetically engineered, oncolytic activity and selectively replicate in tumor cells but not in normal human cells. To date no investigations have been performed with genetically engineered mesogenic NDV regarding the oncolytic activity. We describe here the generation of recombinant viruses of the mesogenic naturally oncolytic NDV strain MTH68. We show that not only one, but also two additional transgenes coding for amino-acid chains with a molecular weight of 25 and 50 kDa can be inserted into the viral genome without affecting viral growth, oncolytic potency or tumor-selective replication of the virus. Transgenic expression of the heavy and light chains of a monoclonal antibody, as separate additional transcriptional cassettes, leads to the expression of full immunoglobulin G (IgG) monoclonal antibody by recombinant NDV. Infection of tumor cells with antibody-transgenic viruses results in the efficient production and secretion of a functional full size IgG antibody by the tumor cells, that specifically binds to its target-antigen in tumor tissue. This approach will allow to combine the advantages of oncolytic RNA viruses and monoclonal antibodies in a single powerful anticancer agent with improved or even new therapeutic properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Cassel WA, Garrett RE . Newcastle disease virus as an antineoplastic agent. Cancer 1965; 18: 863–868.
Cassel WA, Garrett RE . Tumor immunity after viral oncolysis. J Bacteriol 1966; 92: 792.
Csatary LK . Viruses in the treatment of cancer. Lancet 1971; 2: 825.
Sinkovics JG, Horvath JC . Newcastle disease virus (NDV): brief history of its oncolytic strains. J Clin Virol 2000; 16: 1–15.
Reichard KW, Lorence RM, Cascino CJ, Peeples ME, Walter RJ, Fernando MB et al. Newcastle disease virus selectively kills human tumor cells. J Surg Res 1992; 52: 448–453.
Lorence RM, Katubig BB, Reichard KW, Reyes HM, Phuangsab A, Sassetti MD et al. Complete regression of human fibrosarcoma xenografts after local Newcastle disease virus therapy. Cancer Res 1994; 54: 6017–6021.
Fabian Z, Csatary CM, Szeberenyi J, Csatary LK . p53-independent endoplasmic reticulum stress-mediated cytotoxicity of a Newcastle disease virus strain in tumor cell lines. J Virol 2007; 81: 2817–2830.
Alexander DJ . Newcastle Disease. Kluwer Academic Publishers: MA, 1988.
Csatary LK, Eckhardt S, Bukosza I, Czegledi F, Fenyvesi C, Gergely P et al. Attenuated veterinary virus vaccine for the treatment of cancer. Cancer Detect Prev 1993; 17: 619–627.
Csatary LK, Bakacs T . Use of Newcastle disease virus vaccine (MTH-68/H) in a patient with high-grade glioblastoma. JAMA 1999; 281: 1588–1589.
Csatary LK, Moss RW, Beuth J, Torocsik B, Szeberenyi J, Bakacs T . Beneficial treatment of patients with advanced cancer using a Newcastle disease virus vaccine (MTH-68/H). Anticancer Res 1999; 19: 635–638.
Csatary LK, Gosztonyi G, Szeberenyi J, Fabian Z, Liszka V, Bodey B et al. MTH-68/H oncolytic viral treatment in human high-grade gliomas. J Neurooncol 2004; 67: 83–93.
Pecora AL, Rizvi N, Cohen GI, Meropol NJ, Sterman D, Marshall JL et al. Phase I trial of intravenous administration of PV701, an oncolytic virus, in patients with advanced solid cancers. J Clin Oncol 2002; 20: 2251–2266.
Laurie SA, Bell JC, Atkins HL, Roach J, Bamat MK, O'Neil JD et al. A phase 1 clinical study of intravenous administration of PV701, an oncolytic virus, using two-step desensitization. Clin Cancer Res 2006; 12: 2555–2562.
Lorence RM, Roberts MS, O'Neil JD, Groene WS, Miller JA, Mueller SN et al. Phase 1 clinical experience using intravenous administration of PV701, an oncolytic Newcastle disease virus. Curr Cancer Drug Targets 2007; 7: 157–167.
Peeters BP, de Leeuw OS, Koch G, Gielkens AL . Rescue of Newcastle disease virus from cloned cDNA: evidence that cleavability of the fusion protein is a major determinant for virulence. J Virol 1999; 73: 5001–5009.
Romer-Oberdorfer A, Mundt E, Mebatsion T, Buchholz UJ, Mettenleiter TC . Generation of recombinant lentogenic Newcastle disease virus from cDNA. J Gen Virol 1999; 80 (Part 11): 2987–2995.
Nakaya T, Cros J, Park MS, Nakaya Y, Zheng H, Sagrera A et al. Recombinant Newcastle disease virus as a vaccine vector. J Virol 2001; 75: 11868–11873.
Engel-Herbert I, Werner O, Teifke JP, Mebatsion T, Mettenleiter TC, Romer-Oberdorfer A . Characterization of a recombinant Newcastle disease virus expressing the green fluorescent protein. J Virol Methods 2003; 108: 19–28.
Zhao H, Peeters BP . Recombinant Newcastle disease virus as a viral vector: effect of genomic location of foreign gene on gene expression and virus replication. J Gen Virol 2003; 84: 781–788.
Janke M, Peeters B, de Leeuw O, Moorman R, Arnold A, Fournier P et al. Recombinant Newcastle disease virus (NDV) with inserted gene coding for GM-CSF as a new vector for cancer immunogene therapy. Gene Therapy 2007; 14: 1639–1649.
Krishnamurthy S, Huang Z, Samal SK . Recovery of a virulent strain of Newcastle disease virus from cloned cDNA: expression of a foreign gene results in growth retardation and attenuation. Virology 2000; 278: 168–182.
de Leeuw OS, Koch G, Hartog L, Ravenshorst N, Peeters BP . Virulence of Newcastle disease virus is determined by the cleavage site of the fusion protein and by both the stem region and globular head of the haemagglutinin-neuraminidase protein. J Gen Virol 2005; 86: 1759–1769.
Lamb RA, Kolakofsky D . Paramyxoviridae: the viruses and their replication. In: Fields BN (ed). Virology. Lippincott Williams & Wilkins: PA, 2001, pp 1305–1485.
Parato KA, Senger D, Forsyth PA, Bell JC . Recent progress in the battle between oncolytic viruses and tumours. Nat Rev Cancer 2005; 5: 965–976.
Dingli D, Peng KW, Harvey ME, Greipp PR, O'Connor MK, Cattaneo R et al. Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood 2004; 103: 1641–1646.
Hasegawa K, Pham L, O'Connor MK, Federspiel MJ, Russell SJ, Peng KW . Dual therapy of ovarian cancer using measles viruses expressing carcinoembryonic antigen and sodium iodide symporter. Clin Cancer Res 2006; 12: 1868–1875.
Grote D, Cattaneo R, Fielding AK . Neutrophils contribute to the measles virus-induced antitumor effect: enhancement by granulocyte macrophage colony-stimulating factor expression. Cancer Res 2003; 63: 6463–6468.
Fernandez M, Porosnicu M, Markovic D, Barber GN . Genetically engineered vesicular stomatitis virus in gene therapy: application for treatment of malignant disease. J Virol 2002; 76: 895–904.
Shin EJ, Wanna GB, Choi B, Aguila III D, Ebert O, Genden EM et al. Interleukin-12 expression enhances vesicular stomatitis virus oncolytic therapy in murine squamous cell carcinoma. Laryngoscope 2007; 117: 210–214.
Bian H, Fournier P, Moormann R, Peeters B, Schirrmacher V . Selective gene transfer in vitro to tumor cells via recombinant Newcastle disease virus. Cancer Gene Ther 2005; 12: 295–303.
Vigil A, Park MS, Martinez O, Chua MA, Xiao S, Cros JF et al. Use of reverse genetics to enhance the oncolytic properties of Newcastle disease virus. Cancer Res 2007; 67: 8285–8292.
Menrad A, Menssen HD . ED-B fibronectin as a target for antibody-based cancer treatments. Expert Opin Ther Targets 2005; 9: 491–500.
Schnell MJ, Mebatsion T, Conzelmann KK . Infectious rabies viruses from cloned cDNA. EMBO J 1994; 13: 4195–4203.
Peeters BP, Gruijthuijsen YK, de Leeuw OS, Gielkens AL . Genome replication of Newcastle disease virus: involvement of the rule-of-six. Arch Virol 2000; 145: 1829–1845.
Tarli L, Balza E, Viti F, Borsi L, Castellani P, Berndorff D et al. A high-affinity human antibody that targets tumoral blood vessels. Blood 1999; 94: 192–198.
Skiadopoulos MH, Surman SR, Riggs JM, Orvell C, Collins PL, Murphy BR . Evaluation of the replication and immunogenicity of recombinant human parainfluenza virus type 3 vectors expressing up to three foreign glycoproteins. Virology 2002; 297: 136–152.
Stojdl DF, Lichty B, Knowles S, Marius R, Atkins H, Sonenberg N et al. Exploiting tumor-specific defects in the interferon pathway with a previously unknown oncolytic virus. Nat Med 2000; 6: 821–825.
Hashiro G, Loh PC, Yau JT . The preferential cytotoxicity of reovirus for certain transformed cell lines. Arch Virol 1977; 54: 307–315.
Everts B, van der Poel HG . Replication-selective oncolytic viruses in the treatment of cancer. Cancer Gene Ther 2005; 12: 141–161.
Fiola C, Peeters B, Fournier P, Arnold A, Bucur M, Schirrmacher V . Tumor selective replication of Newcastle disease virus: association with defects of tumor cells in antiviral defence. Int J Cancer 2006; 119: 328–338.
Zardi L, Carnemolla B, Siri A, Petersen TE, Paolella G, Sebastio G et al. Transformed human cells produce a new fibronectin isoform by preferential alternative splicing of a previously unobserved exon. EMBO J 1987; 6: 2337–2342.
Menrad A, Cao YJ, Redlitz A . ED-B Fibronectin as a Target for Antibody Based Therapeutics and the Evaluation of Phage Angiomics for Target Identification and Validation. Strategic Research Institute Conference on Vascular Targeting Agents 2004, 08–09 November, p 1.
Jain RK . Transport of molecules across tumor vasculature. Cancer Metastasis Rev 1987; 6: 559–593.
Jain RK . Delivery of molecular and cellular medicine to solid tumors. Adv Drug Deliv Rev 1997; 26: 71–90.
Kontermann RE . Intrabodies as therapeutic agents. Methods 2004; 34: 163–170.
Acknowledgements
We thank Karl-Klaus Conzelmann for the cell line BHK-T7 and the plasmid pX8δT, Petra Boukamp for the cell line HaCaT and Andreas Menrad for recombinant ED-B and helpful discussions. We acknowledge Katja Köckritz and Steve Baethge for excellent technical support and Klaus Bosslet for critical discussions and support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Supplementary information
Rights and permissions
About this article
Cite this article
Pühler, F., Willuda, J., Puhlmann, J. et al. Generation of a recombinant oncolytic Newcastle disease virus and expression of a full IgG antibody from two transgenes. Gene Ther 15, 371–383 (2008). https://doi.org/10.1038/sj.gt.3303095
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3303095
Keywords
This article is cited by
-
Oncolytic Newcastle disease virus expressing chimeric antibody enhanced anti-tumor efficacy in orthotopic hepatoma-bearing mice
Journal of Experimental & Clinical Cancer Research (2015)
-
Isolation of more potent oncolytic paramyxovirus by bioselection
Gene Therapy (2013)
-
Construction of a minigenome rescue system for Newcastle disease virus strain Italien
Archives of Virology (2011)
-
Rac1 is required for oncolytic NDV replication in human cancer cells and establishes a link between tumorigenesis and sensitivity to oncolytic virus
Oncogene (2010)
-
Overexpression of Newcastle disease virus (NDV) V protein enhances NDV production kinetics in chicken embryo fibroblasts
Applied Microbiology and Biotechnology (2010)