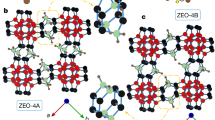
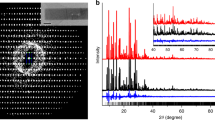
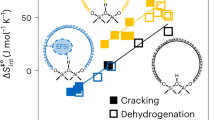
Abstract
THE structure of silicalite, a new polymorph of silica which was first prepared by Flanigen et al.1, is so similar to that of the silica-rich aluminosilicate zeolite ZSM-5 (ref. 2) that we suggest that it should be regarded as the aluminium-free end member of this zeolite series. Therefore it should be possible to prepare silica analogues of other silica-rich aluminosilicate zeolites such as ZSM-11 (ref. 3), ZSM-12 (ref. 4), ZSM-21 (ref. 5) and ZSM-34 (ref. 6). We report here the preparation of what we believe is the analogue of ZSM-11, which we call silicalite-2, adopting the terminology silicalite-1 for the form prepared by Flanigen et al.1 The silicalite-1, ZSM-5, ZSM-11 and, by inference, silicalite-2 frameworks contain 4-, 5- and 6-membered rings linked to form a system of channels with 10-membered ring openings1–3. In silicalite-1 and ZSM-5 the channels are a combination of linear and zig-zag1,2, while in ZSM-113 and silicalite-2 all the channels are linear. The lack of substitutional aluminium results in silicalites having no significant catalytic or cation exchange properties compared with the zeolites. However, silicalites are unusual in that they are the only known hydrophobic forms of silica and are capable of absorbing organic molecules up to ∼6 Å kinetic diameter. Thermogravimetric studies of silicalites-1 and -2 containing absorbed long straight-chain hydrocarbon compounds (C4–C16) show that degassing occurs in two stages, which we consider to be due to self-blocking of the channels at intersection points.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Flanigen, E. M. et al. Nature 271, 512 (1978).
Kokotailo, G. T., Lawton, S. L., Olson, D. H. & Meier, W. M. Nature 272, 437 (1978).
Kokotailo, G. T., Chu, P., Lawton, S. L. & Meier, W. M. Nature 275, 119 (1978).
US Pat. No. 3832449 (1974).
US Pat. No. 4046859 (1977).
US Pat. No. 4079096 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BIBBY, D., MILESTONE, N. & ALDRIDGE, L. Silicalite-2, a silica analogue of the aluminosilicate zeolite ZSM-11. Nature 280, 664–665 (1979). https://doi.org/10.1038/280664a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/280664a0
This article is cited by
-
Synthesis of Pure Silica Zeolites
Chemical Research in Chinese Universities (2022)
-
Calorimetric study of adsorption of alcohols on silicas
Adsorption (2016)
-
Controllable polymerization of poly-DVB–VBC–g–AO resin via surface-initiated atom transfer radical polymerization for uranium removal
Journal of Radioanalytical and Nuclear Chemistry (2015)
-
Characterizations and Catalytic Properties of Chromium Silicalite-2 Prepared by Direct Hydrothermal Synthesis and Impregnation
Catalysis Letters (2010)
-
Vapour phase beckmann rearrangement of cyclohexanone oxime over MEL type zeolites
Catalysis Letters (1993)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.