Abstract
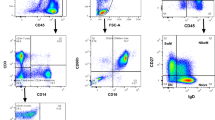
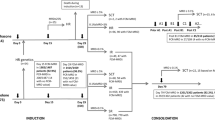
Assessment of minimal residual disease (MRD) by flow cytometry is considered to be based on the reproducibility of the leukemic immunophenotype detected at diagnosis. However, we previously noticed modulation of surface antigen expression in acute lymphoblastic leukemia (ALL) during the early treatment. Hence, we investigated this in 30 children with B-cell precursor ALL consecutively enrolled in the AIEOP-BFM ALL 2000 protocol. Quantitative expression of seven antigens useful in MRD monitoring was studied at diagnosis and compared to that measured at different time points of remission induction therapy. Downmodulation in the expression of CD10 and CD34 occurred at follow-up. By contrast, upmodulation of CD19, CD20, CD45RA, and CD11a was observed, while the expression of CD58 remained stable. Despite this, we could unambiguously discriminate leukemic cells from normal residual B cells. This holds true when bone marrow (BM) samples from similarly treated T-ALL patients, but not from healthy donors, were used as reference. Our results indicate that immunophenotypic modulation occurs in ALL during the early phases of BFM-type protocols. However, the accuracy of MRD detection by flow cytometry seems not negatively affected if adequate analysis protocols are employed. Investigators should take this phenomenon into account in order to avoid pitfalls in flow cytometric MRD studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Campana D, Coustan-Smith E . Detection of minimal residual disease in acute leukemia by flow cytometry. Cytometry 1999; 38: 139–152.
Szczepanski T, Orfao A, van der Velden VH, San Miguel JF, van Dongen JJ . Minimal residual disease in leukaemia patients. Lancet Oncol 2001; 2: 409–417.
van Dongen JJ, Seriu T, Panzer-Grumayer ER, Biondi A, Pongers-Willemse MJ, Corral L et al. Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood. Lancet 1998; 352: 1731–1738.
Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia. European Organization for Research and Treatment of Cancer: Childhood Leukemia Cooperative Group. N Engl J Med 1998; 339: 591–598.
Coustan-Smith E, Behm FG, Sanchez J, Boyett JM, Hancock ML, Raimondi SC et al. Immunological detection of minimal residual disease in children with acute lymphoblastic leukaemia. Lancet 1998; 351: 550–554.
Dworzak MN, Froschl G, Printz D, Mann G, Potschger U, Muhlegger N et al. Prognostic significance and modalities of flow cytometric minimal residual disease detection in childhood acute lymphoblastic leukemia. Blood 2002; 99: 1952–1958.
Björklund E, Mazur J, Soderhall S, Porwit-MacDonald A . Flow cytometric follow-up of minimal residual disease in bone marrow gives prognostic information in children with acute lymphoblastic leukemia. Leukemia 2003; 17: 138–148.
Vidriales MB, Perez JJ, Lopez-Berges MC, Gutierrez N, Ciudad J, Lucio P et al. Minimal residual disease in adolescent (older than 14 years) and adult acute lymphoblastic leukemias: early immunophenotypic evaluation has high clinical value. Blood 2003; 101: 4695–4700.
San Miguel JF, Ciudad J, Vidriales MB, Orfao A, Lucio P, Porwit-Mac Donald A et al. Immunophenotypical detection of minimal residual disease in acute leukemia. Crit Rev Oncol/Hematol 1999; 32: 175–185.
Campana D . Determination of minimal residual disease in leukaemia patients. Br J Haematol 2003; 121: 823–828.
Borella L, Casper JT, Lauer SJ . Shifts in expression of cell membrane phenotypes in childhood lymphoid malignancies at relapse. Blood 1979; 54: 64–71.
Greaves M, Paxton A, Janossy G, Pain C, Johnson S, Lister TA . Acute lymphoblastic leukaemia associated antigen. III Alterations in expression during treatment and in relapse. Leuk Res 1980; 4: 1–14.
Lauer S, Piaskowski V, Camitta B, Casper J . Bone marrow and extramedullary variations of cell membrane antigen expression in childhood lymphoid neoplasias at relapse. Leuk Res 1982; 6: 769–774.
Pui CH, Raimondi SC, Head DR, Schell MJ, Rivera GK, Mirro JJ et al. Characterization of childhood acute leukemia with multiple myeloid and lymphoid markers at diagnosis and at relapse. Blood 1991; 78: 1327–1337.
Raghavachar A, Thiel E, Bartram CR . Analyses of phenotype and genotype in acute lymphoblastic leukemias at first presentation and in relapse. Blood 1987; 70: 1079–1083.
Abshire TC, Buchanan GR, Jackson JF, Shuster JJ, Brock B, Head D et al. Morphologic, immunologic and cytogenetic studies in children with acute lymphoblastic leukaemia at diagnosis and relapse: a Pediatric Oncology Group study. Leukemia 1992; 6: 357–362.
van Wering ER, Beishuizen A, Roeffen ET, van der Linden-Schrever BE, Verhoeven MA, Hahlen K et al. Immunophenotypic changes between diagnosis and relapse in childhood acute lymphoblastic leukemia. Leukemia 1995; 9: 1523–1533.
Baer MR, Stewart CC, Dodge RK, Leget G, Sule N, Mrozek K et al. High frequency of immunophenotype changes in acute myeloid leukemia at relapse: implications for residual disease detection (Cancer and Leukemia Group B Study 8361). Blood 2001; 97: 3574–3580.
Coustan-Smith E, Sancho J, Hancock ML, Razzouk BI, Ribeiro RC, Rivera GK et al. Use of peripheral blood instead of bone marrow to monitor residual disease in children with acute lymphoblastic leukemia. Blood 2002; 100: 2399–2402.
Bennet JM, Catovsky D, Daniel MT, Flandrin G, Galton DAG, Gralnik HR, et al., The FAB cooperative group. The morphological classification of acute lymphoblastic leukemia: concordance among observers and clinical correlation. Br J Haematol 1981; 47: 553–561.
Jennings CD, Foon A . Recent advances in flow cytometry: application to the diagnosis of hematologic malignancy. Blood 1997; 90: 2863–2892.
Basso G, Buldini B, De Zen L, Orfao A . New methodologic approaches for immunophenotyping acute leukemias. Haematologica 2001; 86: 675–692.
Pongers-Willemse MJ, Seriu T, Stolz F, d’Aniello E, Gameiro P, Pisa P et al. Primers and protocols for standardized detection of minimal residual disease in acute lymphoblastic leukemia using immunoglobulin and T cell receptor gene rearrangements and TAL1 deletions as PCR targets: report of the BIOMED-1 CONCERTED ACTION: investigation of minimal residual disease in acute leukemia. Leukemia 1999; 13: 110–118.
Szczepanski T, Langerak AW, Wolvers-Tettero IL, Ossenkoppele GJ, Verhoef G, Stul M et al. Immunoglobulin and T cell receptor gene rearrangement patterns in acute lymphoblastic leukemia are less mature in adults than in children: implications for selection of PCR targets for detection of minimal residual disease. Leukemia 1998; 12: 1081–1088.
van der Velden VHJ, Hochhaus A, Cazzaniga G, Szczepanski T, Gabert J, van Dongen JJM . Detection of minimal residual disease in hematologic malignancies by real-time quantitative PCR: principles, approaches, and laboratory aspects. Leukemia 2003; 17: 1013–1034.
Verhagen O, Willemse M, Breunis W, Wijkhuis A, Jacobs D, Joosten S et al. Application of germline IGH probes in real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia. Leukemia 2000; 14: 1426–1435.
Veltroni O, De Zen O, Sanzari MC, Maglia O, Dworzak MN, Ratei R, et al., on behalf of the I-BFM-ALL-FCM-MRD-Study Group. Expression of CD58 in normal, regenerating and leukemic bone marrow B cells: implications for the detection of minimal residual disease in acute lymphocytic leukemia. Haematologica 2003; 11: 1245–1252.
Dworzak MN, Panzer-Grumayer ER . Flow cytometric detection of minimal residual disease in acute lymphoblastic leukemia. Leuk Lymphoma 2003; 44: 1445–1455.
Chorvath B, Sedlak J, Duraj J, Pleskova I, Dubovsky P, Augustinova Doxiadis I et al. Modulation of CALLA (CD10) antigen on cultured ALL (REH) cells: effect of various modulators. Neoplasma 1990; 37: 609–618.
Al-Katib A, Mohammad RM, Khan K, Dan ME, Pettit GR, Sensenbrenner LL . Bryostatin 1-induced modulation of the acute lymphoblastic leukaemia cell line Reh. J Immunother 1993; 14: 33–42.
Stahnke K, Eckhoff S, Mohr A, Meyer LH, Debatin KM . Apoptosis induction in peripheral leukemia cells by remission induction treatment in vivo: selective depletion and apoptosis in a CD34+ subpopulation of leukemia cells. Leukemia 2003; 17: 2130–2139.
Acknowledgements
We thank Giovanni Giudici for his excellent technical assistance with cytogenetic studies, the MRD core-group for the PCR-based MRD data, and the physician and nurses of the Monza's Pediatric Department for their continued support. This work was supported by the Fondazione M Tettamanti and by the Associazione Italiana per la Ricerca sul Cancro (AIRC), Fondazione Città della Speranza, Fondazione Cariplo. The study was promoted by the International BFM-Study Group under the direction of G Masera, H Gadner, and M Schrappe.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Gaipa, G., Basso, G., Maglia, O. et al. Drug-induced immunophenotypic modulation in childhood ALL: implications for minimal residual disease detection. Leukemia 19, 49–56 (2005). https://doi.org/10.1038/sj.leu.2403559
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403559
Keywords
This article is cited by
-
Critical evaluation of the utility of pre- and post-therapy immunophenotypes in assessment of measurable residual disease in B-ALL
Annals of Hematology (2021)
-
Flow Cytometric Minimal Residual Disease Analysis in Acute Leukemia: Current Status
Indian Journal of Hematology and Blood Transfusion (2020)
-
Novel Therapies in Acute Lymphoblastic Leukemia
Current Hematologic Malignancy Reports (2018)
-
Flow cytometry in the diagnosis and monitoring of acute leukemia in children
Journal of Hematopathology (2015)
-
Minimal residual disease in acute lymphoblastic leukemia: optimal methods and clinical relevance, pitfalls and recent approaches
Medical Oncology (2014)