Abstract
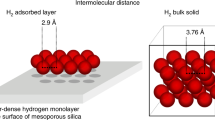
SOME years ago we showed1 that the melting points of molecular layers spread over solid surfaces are lower than the melting points of the bulk substances. The melting point may be lower by as much as 30° C and depends on the nature of the substance, the number of molecular layers spread over the solid surface and the nature of the solid support. Monolayers show maximum lowering, while after a coverage of four to ten layers the melting points reach their bulk values. This phenomenon is a general property of all substances.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Karagounis, G., Helv. Chim. Acta, 37, 805 (1954). Deronane, E. G., Ampere Colloquium (1968).
Karagounis, G., Helv. Chim. Acta, 36, 282 (1953).
Bloembergen, N., Purcell, E. M., and Pound, R. V., Phys. Rev., 73, 796 (1948).
Karagounis, G., and Gutbrod, M., Praktiha. Acad. Athens, (October, 1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KARAGOUNIS, G., PAPAYANNAKIS, E. & STASSINOPOULOS, C. Lowering of the Melting Point of Molecular Layers observed by Nuclear Magnetic Resonance. Nature 221, 655–656 (1969). https://doi.org/10.1038/221655a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/221655a0
This article is cited by
-
Fracture of ice in strong high-frequency electromagnetic fields
Soviet Mining Science (1971)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.