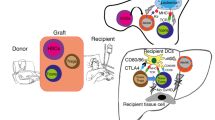
Summary:
Adoptive immunotherapy using natural killer (NK) cells may prove useful, especially in situations where infusion of T cells is impractical such as in recipients of haploidentical stem cell transplantation (HSCT) from haploidentical donors. NK cells may induce potent antileukemic and possibly antirejection activity and may even mitigate graft versus host disease (GvHD). Whether such effects are clinically important and whether they are mediated mainly or exclusively by KIR–HLA class I interactions remains to be determined. Recent advances in graft engineering provide for methods to isolate large numbers of purified NK cells. Several groups have shown that clinical grade NK cells up to a dose of 107/kg may be collected and purified for the purpose of infusion to patients. Early results, in a limited number of patients, show that these cell doses may be administered without adverse events and without inducing GvHD. Whether such infusions will be useful in preventing graft rejection, or exerting graft versus leukemia effects and hastening immune recovery requires further study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Robertson MJ, Ritz J . Biology and clinical relevance of human natural killer cells. Blood 1990; 76: 2421–2438.
Smyth MJ, Hayakawa Y, Takeda K, Yagita H . New aspects of natural-killer-cell surveillance and therapy of cancer. Nat Rev Cancer 2002; 2: 850–861.
French AR, Yokoyama WM . Natural killer cells and viral infections. Curr Opin Immunol 2003; 15: 45–51.
Moretta A, Bottino C, Mingari MC et al. What is a natural killer cell? Nat Immunol 2002; 3: 6–8.
Moretta L, Moretta A . Unravelling natural killer cell function: triggering and inhibitory human NK receptors. EMBO J 2004; 23: 255–259.
Vilches C, Parham P . KIR: diverse, rapidly evolving receptors of innate and adaptive immunity. Annu Rev Immunol 2002; 20: 217–251.
Farag SS, Fehniger TA, Ruggeri L et al. Natural killer cell receptors: new biology and insights into the graft-versus-leukemia effect. Blood 2002; 100: 1935–1947.
Borrego F, Kabat J, Kim DK et al. Structure and function of major histocompatibility complex (MHC) class I specific receptors expressed on human natural killer (NK) cells. Mol Immunol 2002; 38: 637–660.
Moretta A, Bottino C, Vitale M et al. Activating receptors and coreceptors involved in human natural killer cell-mediated cytolysis. Annu Rev Immunol 2001; 19: 197–223.
Parham P, McQueen KL . Alloreactive killer cells: hindrance and help for haematopoietic transplants. Nat Rev Immunol 2003; 3: 108–122.
Barao I, Murphy WJ . The immunobiology of natural killer cells and bone marrow allograft rejection. Biol Blood Marrow Transplant 2003; 9: 727–741.
Cooper MA, Fehniger TA, Turner SC et al. Human natural killer cells: a unique innate immunoregulatory role for the CD56(bright) subset. Blood 2001; 97: 3146–3151.
Robertson MJ, Soiffer RJ, Wolf SF et al. Response of human natural killer (NK) cells to NK cell stimulatory factor (NKSF): cytolytic activity and proliferation of NK cells are differentially regulated by NKSF. J Exp Med 1992; 175: 779–788.
Henslee-Downey PJ . Allogeneic transplantation across major HLA barriers. Best Pract Res Clin Haematol 2001; 14: 741–754.
Singhal S, Henslee-Downey PJ, Powles R et al. Haploidentical vs autologous hematopoietic stem cell transplantation in patients with acute leukemia beyond first remission. Bone Marrow Transplant 2003; 31: 889–895.
Aversa F, Tabilio A, Velardi A et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med 1998; 339: 1186–1193.
Martelli MF, Aversa F, Bachar-Lustig E et al. Transplants across human leukocyte antigen barriers. Semin Hematol 2002; 39: 48–56.
Handgretinger R, Klingebiel T, Lang P et al. Megadose transplantation of purified peripheral blood CD34(+) progenitor cells from HLA-mismatched parental donors in children. Bone Marrow Transplant 2001; 27: 777–783.
Passweg JR, Kuhne T, Gregor M et al. Increased stem cell dose, as obtained using currently available technology, may not be sufficient for engraftment of haploidentical stem cell transplants. Bone Marrow Transplant 2000; 26: 1033–1036.
Rowe JM, Lazarus HM . Genetically haploidentical stem cell transplantation for acute leukemia. Bone Marrow Transplant 2001; 27: 669–676.
Ruggeri L, Capanni M, Casucci M et al. Role of natural killer cell alloreactivity in HLA-mismatched hematopoietic stem cell transplantation. Blood 1999; 94: 333–339.
Eyrich M, Lang P, Lal S et al. A prospective analysis of the pattern of immune reconstitution in a paediatric cohort following transplantation of positively selected human leucocyte antigen-disparate haematopoietic stem cells from parental donors. Br J Haematol 2001; 114: 422–432.
Ruggeri L, Capanni M, Tosti A et al. Innate immunity against hematological malignancies. Cytotherapy 2002; 4: 343–346.
Ruggeri L, Capanni M, Urbani E et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science 2002; 295: 2097–2100.
Ruggeri L, Capanni M, Martelli MF, Velardi A . Cellular therapy: exploiting NK cell alloreactivity in transplantation. Curr Opin Hematol 2001; 8: 355–359.
Shlomchik WD, Couzens MS, Tang CB et al. Prevention of graft versus host disease by inactivation of host antigen-presenting cells. Science 1999; 285: 412–415.
Davies SM, Ruggieri L, DeFor T et al. Evaluation of KIR ligand incompatibility in mismatched unrelated donor hematopoietic transplants. Killer immunoglobulin-like receptor. Blood 2002; 100: 3825–3827.
Giebel S, Locatelli F, Lamparelli T et al. Survival advantage with KIR ligand incompatibility in hematopoietic stem cell transplantation from unrelated donors. Blood 2003; 102: 814–819.
Gagne K, Brizard G, Gueglio B et al. Relevance of KIR gene polymorphisms in bone marrow transplantation outcome. Hum Immunol 2002; 63: 271–280.
Hsu KC, Taylor CA, O'Reilly RJ et al. AML patients lacking KIR ligand for donor KIR exhibit increased overall survival in HLA-identical sibling transplants. Biol Blood Marrow Transplant 2004; 10 (Suppl. 1): 7.
Bishara A, De Santis D, Witt CC et al. The beneficial role of inhibitory KIR genes of HLA class I NK epitopes in haploidentically mismatched stem cell allografts may be masked by residual donor-alloreactive T cells causing GVHD. Tissue Antigens 2004; 63: 204–211.
Lowe EJ, Turner V, Handgretinger R et al. T-cell alloreactivity dominates natural killer cell alloreactivity in minimally T-cell-depleted HLA-non-identical paediatric bone marrow transplantation. Br J Haematol 2003; 123: 323–326.
Cook MA, Milligan DW, Fegan CD et al. The impact of donor KIR and patient HLA-C genotypes on outcome following HLA-identical sibling hematopoietic stem cell transplantation for myeloid leukemia. Blood 2004; 103: 1521–1526.
Leung W, Iyengar R, Turner V et al. Determinants of antileukemia effects of allogeneic NK cells. J Immunol 2004; 172: 644–650.
Kolb HJ, Mittermuller J, Clemm C et al. Donor leukocyte transfusions for treatment of recurrent chronic myelogenous leukemia in marrow transplant patients. Blood 1990; 76: 2462–2465.
Dazzi F, Szydlo RM, Craddock C et al. Comparison of single-dose and escalating-dose regimens of donor lymphocyte infusion for relapse after allografting for chronic myeloid leukemia. Blood 2000; 95: 67–71.
Guglielmi C, Arcese W, Dazzi F et al. Donor lymphocyte infusion for relapsed chronic myelogenous leukemia: prognostic relevance of the initial cell dose. Blood 2002; 100: 397–405.
Soiffer RJ, Alyea EP, Hochberg E et al. Randomized trial of CD8+ T-cell depletion in the prevention of graft-versus-host disease associated with donor lymphocyte infusion. Biol Blood Marrow Transplant 2002; 8: 625–632.
Lewalle P, Triffet A, Delforge A et al. Donor lymphocyte infusions in adult haploidentical transplant: a dose finding study. Bone Marrow Transplant 2003; 31: 39–44.
Hester J . A clinical role for peripheral blood lymphocyte infusions and perspectives on collection. Ther Apher Dial 2003; 7: 305–311.
Iyengar R, Handgretinger R, Babarin-Dorner A et al. Purification of human natural killer cells using a clinical-scale immunomagnetic method. Cytotherapy 2003; 5: 479–484.
Uharek L, Hilbers U, Hartung G et al. Cellular immunotherapy with alloreactive NK cells: promising results of a phase I/II study investigating the use of ex-vivo activated donor CD3−CD56+ NK cells after transplantation of highly purified HLA-haplotype mismatched stem cell grafts in patients. Onkologie 2003; 26 S5: 38.
Koehl U, Soerensen J, Esser R et al. IL-2 activated NK cell immunotherapy of three children after haploidentical stem cell transplantation. Blood Cells, Mol Dis 2004 (in press).
Lang P, Pfeiffer M, Handgretinger R et al. Clinical scale isolation of T cell-depleted CD56+ donor lymphocytes in children. Bone Marrow Transplant 2002; 29: 497–502.
Uharek L, Hilbers U, Hartung G et al. Graft rejection and leukaemia relapse after haploidentical stem cell transplantation in adult patients with high risk haematological disease. Bone Marrow Transplantation 2004; 33 (Suppl. 1): S19.
Passweg JR, Tichelli A, Meyer-Monard S et al. Purified donor NK-lymphocyte infusion to consolidate engraftment after haploidentical stem cell transplantation. Leukemia 2004; 18: 1835–1838.
Sato H, Shiobara S, Yasue S et al. Lymphocyte collection for donor leucocyte infusion from normal donors: estimation of the minimum processed blood volume and safety of the procedure. Vox Sang 2001; 81: 124–127.
Slavin S, Morecki S, Shapira M et al. Immunotherapy using rIL-2 activated mismatched donor lymphocytes positively selected for CD56+ for the treatment of resistant haematologic malignancies after stem cell transplantation. Bone Marrow Transplant. 2004; 33 (Suppl. 1): S174.
Igarashi T, Wynberg J, Srinivasan R et al. Enhanced cytotoxicity of allogeneic NK cells with killer immunoglobulin-like receptor ligand incompatibility against melanoma and renal cell carcinoma cells. Blood 2004; 104: 170–177.
Uharek L, Zeis M, Glass B et al. High lytic activity against human leukemia cells after activation of allogeneic NK cells by IL-12 and IL-2. Leukemia 1996; 10: 1758–1764.
Nagao F, Suzui M, Takeda K et al. Mobilization of NK cells by exercise: downmodulation of adhesion molecules on NK cells by catecholamines. Am J Physiol Regul Integr Comp Physiol 2000; 279: R1251–R1256.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Passweg, J., Stern, M., Koehl, U. et al. Use of natural killer cells in hematopoetic stem cell transplantation. Bone Marrow Transplant 35, 637–643 (2005). https://doi.org/10.1038/sj.bmt.1704810
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704810
Keywords
This article is cited by
-
Intestinal barrier loss as a critical pathogenic link between inflammatory bowel disease and graft-versus-host disease
Mucosal Immunology (2015)
-
Interleukin-2-stimulated natural killer cells are less susceptible to mycophenolate mofetil than non-activated NK cells: possible consequences for immunotherapy
Cancer Immunology, Immunotherapy (2014)
-
Diagnosis and evaluation of intestinal graft-versus-host disease after allogeneic hematopoietic stem cell transplantation following reduced-intensity and myeloablative conditioning regimens
International Journal of Hematology (2013)
-
Natural killer cell immune escape in acute myeloid leukemia
Leukemia (2012)
-
Disease stage stratified effects of cell dose in unrelated BMT for hematological malignancies: a report from Japan marrow donor program
Bone Marrow Transplantation (2011)