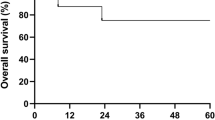
Summary:
An X-linked severe combined immunodeficient (SCID) patient received a nonirradiated erythrocyte transfusion and developed transfusion-associated graft-versus-host disease (TAGVHD), which was controllable with high-dose corticosteroids. Haplo-identical SCT was performed, after a myeloablative conditioning regimen. At day +26, he developed GVHD. Chimerism studies revealed DNA of the erythrocyte transfusion donor (ETD) and recipient only. Because of early nonengraftment and the presence of alloreactive T cells of ETD origin, the patient was treated with an immunosuppressive conditioning regimen followed by a second SCT from the same donor. While tapering immunosuppression, he again developed mild GVHD, and DNA of ETD and bone marrow donor origin were both present. On cyclosporin, the ETD-DNA signal finally disappeared. High-resolution HLA typing revealed haplo-identity between BMD, ETD and the patient, which might have contributed to the relative mild course of the TAGVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Schroeder ML . Transfusion-associated graft-versus-host disease. Br J Haematol 2002; 117: 275–287.
Orlin JB, Ellis MH . Transfusion-associated graft-versus-host disease. Curr Opin Hematol 1997; 4: 442–448.
Friedman DF, Kwittken P, Cizman B et al. DNA-based HLA typing of nonhematopoietic tissue used to select the marrow transplant donor for successful treatment of transfusion-associated graft-versus-host disease. Clin Diagn Lab Immunol 1994; 1: 590–596.
Petz LD, Calhoun L, Yam P et al. Transfusion-associated graft-versus-host disease in immunocompetent patients: report of a fatal case associated with transfusion of blood from a second-degree relative, and survey of predisposing factors. Transfusion 1993; 33: 742–750.
Fischer A . Primary immunodeficiency diseases: an experimental model for molecular medicine. Lancet 2001; 357: 1863–1869.
van Blokland WTM, Ignatiadis MG, Bosboom EC et al. An optimal microsatellite analysis for donor and recipient cells after bone marrow tranplantation. In: Charron D (ed.). Genetic Diversity of HLA, Functional and Medical Implication. EDK Press: Paris, France, 1997, pp 586–588.
Knobloch C, Goldman SF, Lattke H et al. Stable engraftment of allogeneic T lymphocytes in a patient with severe combined immunodeficiency following a transfusion with unirradiated blood. Transplantation 1991; 51: 818–824.
Knobloch C, Dennert G . Loss of F1 hybrid resistance to bone marrow grafts after rejection of parental lymphocytes: demonstration of parental anti-F1 T killer cells and general immunosuppression in the host. Transplantation 1988; 45: 175–183.
Roelen DL, Stobbe I, Young NT et al. Permissible and immunogenic HLA-A mismatches: cytotoxic T-cell precursor frequencies reflect graft survival data. Hum Immunol 2001; 62: 661–667.
Acknowledgements
The authors would like to thank Joyce van Loon, Petra van der Weide and Anette van Dijk for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Royen-Kerkhof, A., Wulffraat, N., Kamphuis, S. et al. Nonlethal transfusion associated graft-versus-host disease in a severe combined immunodeficient patient. Bone Marrow Transplant 32, 1027–1030 (2003). https://doi.org/10.1038/sj.bmt.1704266
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704266