Abstract
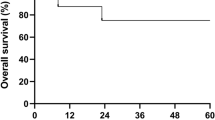
A 27-year-old man with aplastic anemia and renal insufficiency requiring dialysis underwent allogeneic PBSCT. The preparative regimen consisted of melphalan, ATG and TLI. GVHD prophylaxis consisted of cyclosporine and prednisolone. He was dialyzed prior to administration of melphalan and at 24 and 72 h after it. Otherwise, the dialysis schedule was unchanged, at three times a week. Engraftment was rapid. Regimen-related toxicity was minimal. Pharmacokinetic parameters of melphalan were not significantly altered with its plasma half-life 1.5 h. Patients with renal failure can receive allogeneic HSCT, and a combination of melphalan, ATG and TLI may serve as an alternative to CY and ATG.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Storb R, Blume KG, O'Donnell MR et al. Cyclophosphamide and antithymocyte globulin to condition patients with aplastic anemia for allogeneic marrow transplantations: the experience in four centers Biol Blood Marrow Transplant 2001 7: 39 44
Horowitz MM . Current status of allogeneic bone marrow transplantation in acquired aplastic anemia Semin Hematol 2000 37: 30 42
Bearman SI, Appelbaum FR, Buckner CD et al. Regimen-related toxicity in patients undergoing bone marrow transplantation J Clin Oncol 1988 6: 1562 1568
Samuels BL, Bitran JD . High-dose intravenous melphalan: a review J Clin Oncol 1995 13: 1786 1799
Pinguet F, Joulia JM, Martel P et al. High-performance liquid chromatographic assay for melphalan in human plasma. Application to pharmacokinetic studies J Chromatogr B Biomed Appl 1996 686: 43 49
Bramwell V, Calvert RT, Edwards G et al. The disposition of cyclophosphamide in a group of myeloma patients Cancer Chemother Pharmacol 1979 3: 253 259
Grochow LB, Colvin M . Clinical pharmacokinetics of cyclophosphamide Clin Pharmacokinet 1979 4: 380 394
Moore MJ . Clinical pharmacokinetics of cyclophosphamide Clin Pharmacokinet 1991 20: 194 208
Yamamoto R, Kanda Y, Matsuyama T et al. Myopericarditis caused by cyclophosphamide used to mobilize peripheral blood stem cells in a myeloma patient with renal failure Bone Marrow Transplant 2000 26: 685 688
Bunn D, Lea CK, Bevan DJ et al. The pharmacokinetics of anti-thymocyte globulin (ATG) following intravenous infusion in man Clin Nephrol 1996 45: 29 32
Slavin S, Strober S, Fuks Z, Kaplan HS . Induction of specific tissue transplantation tolerance using fractionated total lymphoid irradiation in adult mice: long-term survival of allogeneic bone marrow and skin grafts J Exp Med 1977 146: 34 48
Strober S, Modry DL, Hoppe RT et al. Induction of specific unresponsiveness to heart allografts in mongrel dogs treated with total lymphoid irradiation and antithymocyte globulin J Immunol 1984 132: 1013 1018
Chan KW, Li CK, Worth LL et al. A fludarabine-based conditioning regimen for severe aplastic anemia Bone Marrow Transplant 2001 27: 125 128
Helenglass G, Powles RL, McElwain TJ et al. Melphalan and total body irradiation (TBI) vs cyclophosphamide and TBI as conditioning for allogeneic matched sibling bone marrow transplants for acute myeloblastic leukaemia in first remission Bone Marrow Transplant 1988 3: 21 29
Kottaridis PD, Milligan DW, Chopra R et al. In vivo CAMPATH-1H prevents graft-versus-host disease following nonmyeloablative stem cell transplantation Blood 2000 96: 2419 2425
Giralt S, Thall PF, Khouri I et al. Melphalan and purine analog-containing preparative regimens: reduced-intensity conditioning for patients with hematologic malignancies undergoing allogeneic progenitor cell transplantation Blood 2001 97: 631 637
Gera S, Musch E, Osterheld HK, Loos U . Relevance of the hydrolysis and protein binding of melphalan to the treatment of multiple myeloma Cancer Chemother Pharmacol 1989 23: 76 80
Bosanquet AG, Gilby ED . Pharmacokinetics of oral andintravenous melphalan during routine treatment of multiplemyeloma Eur J Cancer Clin Oncol 1982 18: 355 362
Pecherstorfer M, Zimmer-Roth I, Weidinger S et al. High-dose intravenous melphalan in a patient with multiple myeloma and oliguric renal failure Clin Invest 1994 72: 522 525
Tricot G, Alberts DS, Johnson C et al. Safety of autotransplants with high-dose melphalan in renal failure: a pharmacokinetic and toxicity study Clin Cancer RES 1996 2: 947 952
Kergueris MF, Milpied N, Moreau P et al. Pharmacokinetics of high-dose melphalan in adults: influence of renal function Anticancer Res 1994 14: 2379 2382
Acknowledgements
We thank Dr I Khouri, MD Anderson Cancer Center, USA, Dr J Hansen, Fred Hutchinson Cancer Research Center, USA, Dr R Gale, University of California Los Angeles, USA, and Dr J Martinez, University Hospital La Fe, Spain for good advice in planning the transplantation protocol, Miss Nakae for data collection, and the dialysis nurses and technicians for caring for this patient.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hamaki, T., Katori, H., Kami, M. et al. Successful allogeneic blood stem cell transplantation for aplastic anemia in a patient with renal insufficiency requiring dialysis. Bone Marrow Transplant 30, 195–198 (2002). https://doi.org/10.1038/sj.bmt.1703584
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703584
Keywords
This article is cited by
-
Successful allo-SCT in a Fanconi anemia patient with renal impairment using reduced doses of CY and fludarabine
Bone Marrow Transplantation (2010)
-
Successful bone marrow transplantation in a girl with Fanconi anemia and preterminal renal failure
Bone Marrow Transplantation (2008)
-
Marrow transplantation for severe aplastic anemia with significant renal impairment
Bone Marrow Transplantation (2007)