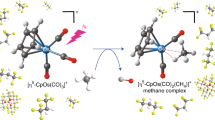
Abstract
BOTH methyl radicals and hydrogen atoms have been proposed as chain-carriers in the photo-chlorination of methane1,3; and, although the former are generally favoured, in neither case has direct evidence been produced4. However, the theoretical heat content change of the corresponding partial reaction would be approximately zero, if methyl radicals were involved, but endothermic by 21 kcal./mole, on the basis of hydrogen atom formation.
Similar content being viewed by others
Article PDF
References
Coehn and Cordes, Z. phys. Chem., B, 9, 1 (1930).
Tamura, Rev. Phys. Chem. Japan, 15, 86 (1941).
Steiner and Watson, Farad. Soc. Discussion, 2, 89 (1947).
Noyes and Leighton, “The Photochemistry of Gases”, 288 (Reinhold Publishing Corporation, 1941).
Ritchie and Norrish, Proc. Roy. Soc., A, 140, 112, 713 (1933).
Norrish, Trans. Farad. Soc., 27, 471 (1931).
Bricker and Johnson, Indust. and Eng. Chem., Anal. Edit., 17, 400 (1945).
Krauskopf and Rollefson, J. Amer. Chem. Soc., 56, 2542 (1934).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WINNING, W. Intermediates in the Chlorine-sensitized Photo-oxidation of Methane. Nature 167, 525 (1951). https://doi.org/10.1038/167525a0
Issue Date:
DOI: https://doi.org/10.1038/167525a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.