Abstract
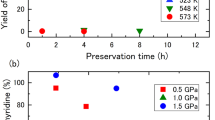
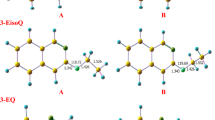
THE slow decomposition of ethylene oxide vapour has received the attention of a number of investigators1–3. In extension of their work, we have studied the limiting conditions of temperature and pressure under which the reaction becomes explosive in a 550 c.c. quartz bulb.
Similar content being viewed by others
Article PDF
References
Heckert, W. W., and Mack, E., J. Amer. Chem. Soc., 51, 2706 (1929).
Thompson, H. W., and Meissner, M., Trans. Farad. Soc., 32, 1451 (1936).
Seddon, R. V., and Travers, M. W., Proc. Roy. Soc., A, 156, 234 (1936).
Cf. Burgoyne, J. H., and Burden, E. A., Nature, 162, 181 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BURGOYNE, J., BURDEN, F. Explosive Decomposition of Ethylene Oxide. Nature 163, 723 (1949). https://doi.org/10.1038/163723a0
Issue Date:
DOI: https://doi.org/10.1038/163723a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.