Abstract
Objective:
To determine the gastrointestinal (GI) tolerance of NUTRIOSE®FB in men.
Design:
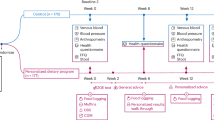
A randomized, placebo-controlled, parallel, double-blind study.
Setting:
The metabolic ward of TNO Quality of Life.
Subjects:
Forty-eight subjects started the study: 16 men participated in one of the three treatments. Subjects consumed either 22.5 g of pure maltodextrin (Glucidex®6), or 30 or 45 g of the dextrin NUTRIOSE®FB daily for 4–5 weeks. Forty-three subjects completed the study (age: 34.7±8.2 years; BMI 24.9±3.3 kg m2).
Measurements:
Tolerance of NUTRIOSE®FB was examined with a GI complaints questionnaire; effectiveness on colonic flora was examined by faecal analysis; fermentation by breath hydrogen excretion measurement. Furthermore, the effect on body weight (BW), energy intake and blood parameters were examined in the study.
Results:
Both doses of NUTRIOSE®FB were very well tolerated and GI complaints hardly differed from the placebo treatment. No diarrhoea was reported due to NUTRIOSE®FB supplementation. In the course of the study, some habituation and adaptation of GI symptoms were found. Fermentation and faecal characteristics (pH and enzyme activity) were significantly positively affected with NUTRIOSE®FB treatment. Body weight in both NUTRIOSE®FB groups remained stable over time, although the placebo-treated group showed a small increase in BW (Δday35−1 0.8±1.0 kg) (P=0.07). However, total food intake and macronutrient composition of the diet remained the same throughout the study. No significant differences were found between the three treatment groups in hunger and satiety scores and food preferences.
Conclusions:
Long-term supplementation of 30 or 45 g of the dextrin NUTRIOSE®FB per day was well tolerated, and may act as a pre-biotic supplement.
Sponsorship:
TNO Quality of Life was assigned by Roquette Frères to perform the study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Albrink MJ (1978). Dietary fibre, plasma insulin, and obesity. Am J Clin Nutr 31, S277–S279.
Alfieri MAH, Pomerleau J, Grace DM, Anderson L (1995). Fibre intake of normal weight, moderately obese and severely obese subjects. Obes Res 3, 541–547.
Birketvedt GS, Aaseth J, Florholmen JR, Ryttig K (2000). Long-term effect of fibre supplement and reduced energy intake on body weight and blood lipids in overweight subjects. Acta Med 43, 129–132.
Coudray C, Bellanger J, Vermorel M, Sinaud S, Wils D, Feillet-Coudray C et al. (2003). Two polyol, low digestible carbohydrates improve the apparent absorption of magnesium but not of calcium in healthy young men. J Nutr 133, 90–93.
Cybulski KA, Lachaussee J, Kissileff HR (1992). The threshold for satiating effectiveness of psyllium in a nutrient base. Physiol Behav 51, 89–93.
DeLargy HJ, Burley VJ, O'Sullivan KR, Fletcher RJ, Blundell JE (1995). Effects of different soluble:insoluble fibre ratios at breakfast at 24-h pattern of dietary intake and satiety. Eur J Clin Nutr 49, 754–766.
Evans E, Miller DS (1975). Bulking agents in the treatment of obesity. Nutr Metab 18, 199–203.
Flood MT, Auerbach MH, Craig SAS (2004). A review of the clinical toleration studies of polydextrose in food. Food Chem Toxicol 42, 1531–1542.
Friedewald WT, Levy RI, Fredrickson DS (1972). Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18, 499–502.
Gibson GR, Roberfroid MB (1995). Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr 125, 1401–1412.
Hamilton CC, Anderson JW (1992). Fibre and weight maintenance. J Florida MA 79, 379–381.
Hulshof T (1994). Fat and non-absorbable fat and the regulation of food intake Thesis, Wageningen.
Jorge JMN, Wexner SD, Ehrenpreis ED (1994). The lactulose hydrogen breath test as a measure of orocaecal transit time. Eur J Surg 160, 409–416.
Jouany JP (1982). Volatile fatty acids and alcohols determination in digestive contents, silage juice, bacterial culture and anaerobic fermentor contents. Sci Aliments 2, 131–144.
Kritchevsky D (1988). Dietary fibre. Annu Rev Nutr 8, 301–328.
Levine AS, Tallman JR, Grace MK, Parker SA, Billington CJ, Levitt MD (1989). Effect of breakfast cereals on short-term food intake. Am J Clin Nutr 50, 1303–1307.
Lovejoy J, DiGirolamo M (1992). Habitual dietary intake and insulin sensitivity in lean and obese adults. Am J Clin Nutr 55, 1174–1179.
Ma Z, Gingerich RL, Santiago JV, Klein S, Smith CH, Landt M (1996). Radioimmunoassay of leptin in human plasma. Clin Chem 42, 942–946.
Pasman WJ (1998). Obesity Treatment and Weight Maintenance Thesis, Maastricht University, Maastricht.
Pasman WJ, Wauters MAJ, Westerterp-Plantenga MS, Saris WHM (1997). Effect of one week of fibre supplementation on hunger and satiety or energy intake. Appetite 29, 77–87.
Porikos K, Hagamen S (1986). Is fibre satiating? Effects of a high fibre preload on subsequent food intake of normal-weight and obese young men. Appetite 7, 153–162.
Reddy BS, Engle A, Simi B, Goldman M (1992). Effect of dietary fibre on colonic bacterial enzymes and bile acids in relation to colon cancer. Gastroenterology 102, 1475–1482.
Roediger W (1991). Cellular metabolism of short-chain fatty acids in colonic epithelial cells. In: Roche AF (ed). Short-Chain Fatty Acids: Metabolism and Clinical Importance. Report of the Tenth Ross Conference on Medical Research. Ross Laboratories: Columbus, Ohio. pp 67–71.
Rössner S (1992). Dietary fibre in the prevention and treatment of obesity. In: Schweizer TF, Edwards CA (ed). Dietary fibre – A Component of Food Chapter 15. Springer-Verlag: London, pp 265–277.
Rumessen JJ, Gudmand-Høyer E (1998). Fructans of chicory: intestinal transport and fermentation of different chain lengths and relation to fructose and sorbitol malabsorption. Am J Clin Nutr 68, 357–364.
Sas Institute Inc. (1989) SAS/STAT® User's Guide Version 6.12, 4th edn. SAS Institute Inc.: Cary, NC, USA.
Spiller GA (1993). Suggestions for a basis on which to determine a desirable intake of dietary fibre. In: Spiller GA (ed). CRC Handbook of Dietary Fibre in Human Nutrition 2nd edn. pp CRC Press Boca Raton: Florida, pp 351–354.
van den Heuvel EGHM, Wils D, Pasman WJ, Bakker M, Saniez M-H, Kardinaal AFM (2004). Short-term digestive tolerance of different doses of NUTRIOSE®FB, a food dextrin, in adult men. Eur J Clin Nutr 58, 1046–1055.
van den Heuvel EGHM, Wils D, Pasman WJ, Saniez M-H, Kardinaal AFM (2005). Dietary supplementation of different doses of NUTRIOSE®FB, a fermentable dextrin, alters the activity of faecal enzymes in healthy men. Eur J Nutr 44, 445–451.
Van Nuenen MHMC, Meyer PD, Venema K (2003). The effect of various inulins and Clostridium difficile on the metabolic activity of the human colonic microbiota in vitro. Micriob Ecol Health Dis 15, 137–144.
Venema K, Van Nuenen M, Smeets-Peeters M, Minekus M, Havenaar R (2000). TNO's in vitro large intestinal model: an excellent screening tool for functional food and pharmaceutical research. Ernährung 24, 558–564.
Venema K, Vermunt SHF, Brink EJ (2005). D-tagatose increases butyrate production by the colonic microbiota in healthy men and women. Microb Ecol Health Dis 17, 47–57.
Wynder EL, Stellman SD, Zang EA (1996). High fibre intake. Indicator of a healthy lifestyle (editorial) JAMA 275, 486–487.
Acknowledgements
We thank the volunteers who participated in the study. We thank all involved in the conduct of the study and laboratory (blood and faecal) analyses and statistics. Koen Venema and Gertruud Bakker are gratefully acknowledged for their useful discussion of the results.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guarantor: W Pasman.
Contributors: WP was involved in the design of the study, conduct, data analysis and wrote the manuscript. DW conceived the original idea, advised on study design and critically appraised the manuscript. M-HS and AK supervised the study and critically evaluated the manuscript.
Rights and permissions
About this article
Cite this article
Pasman, W., Wils, D., Saniez, MH. et al. Long-term gastrointestinal tolerance of NUTRIOSE®FB in healthy men. Eur J Clin Nutr 60, 1024–1034 (2006). https://doi.org/10.1038/sj.ejcn.1602418
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602418
Keywords
This article is cited by
-
Validity of food additive maltodextrin as placebo and effects on human gut physiology: systematic review of placebo-controlled clinical trials
European Journal of Nutrition (2022)
-
Optimization of wheat dextrin yogurt formulation using response surface methodology
Journal of Food Science and Technology (2021)
-
Resistant dextrin improves high-fat-high-fructose diet induced insulin resistance
Nutrition & Metabolism (2020)
-
From waste to health: sustainable exploitation of grape pomace seed extract to manufacture antioxidant, regenerative and prebiotic nanovesicles within circular economy
Scientific Reports (2020)
-
Prebiotic supplementation modulates advanced glycation end-products (AGEs), soluble receptor for AGEs (sRAGE), and cardiometabolic risk factors through improving metabolic endotoxemia: a randomized-controlled clinical trial
European Journal of Nutrition (2020)