Abstract
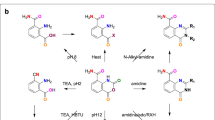
IT has recently been shown1 that amino derivatives of acridine combine high antiseptic potency with low toxicity when the orientation of the amino group(s) is other than the 1- (that is, 9-) position. Unfortunately, the general synthetic methods available for preparing specific amino -acridines involve many stages and are usually wasteful of both time and material2, so that a general reaction for converting simple aromatic compounds to amino-acridines in a single operation would give valuable assistance in exploring further the chemotherapy of these compounds.
Similar content being viewed by others
Article PDF
References
Albert, Francis, Garrod and Linnell, Brit. J. Exper. Path., 19, 41 (1938); Albert, Dyer and Linnell, Quart. J. Pharm., 10, 649 (1937).
Cf. Albert and Linnell, J. Chem. Soc., 88, 1614 (1936); 22 (1938).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ALBERT, A., LARGE, D. Preparation of 2: 8-Diamino-Acridine. Nature 142, 435 (1938). https://doi.org/10.1038/142435a0
Issue Date:
DOI: https://doi.org/10.1038/142435a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.