Abstract
The serotonin 5-hydroxytryptamine-1A (5-HT1A) receptor subtype is of central interest in research, particularly in the area of pathophysiology and pharmacological treatment of psychiatric disorders. Robalzotan (generic name for NAD-299) is a new putative drug that binds with high selectivity and affinity to 5-HT1A-receptors in the rodent brain in vitro and in vivo. The aim of this positron emission tomography study was to determine 5-HT1A receptor occupancy in the cynomolgus monkey brain in vivo after IV injection of robalzotan. Two healthy monkeys were examined with Positron Emission Tomography (PET) and the radioligand [carbonyl-11C]WAY-100635, the first after IV administration of 2 μg/kg and 20 μg/kg, and the second after 10 μg/kg and 100 μg/kg IV. 5-HT1A receptor occupancy was calculated using an equilibrium-ratio analysis. Robalzotan occupied 5-HT1A receptors in a dose-dependent and saturable manner. The highest 5-HT1A receptor occupancy (70–80%) was attained after 100 μg/kg. The relationship between robalzotan drug concentration and 5-HT1A receptor occupancy could be described by a hyperbolic function, which can be used to guide the selection of appropriate doses for the initial studies in man. The study further corroborates that quantitative neuroimaging of receptor binding has potentials for the evaluation and dose finding of new CNS drugs.
Similar content being viewed by others
Main
Serotonergic neurotransmission includes distinct 5-hydroxytryptamine (5-HT) receptor subtypes, classified within at least seven separate families (Hoyer et al. 1994; Peroutka and Howell 1994). The 5-HT1A receptor belongs to the family of G-protein coupled 5-HT1-receptors and is of central interest as a target for the treatment of psychiatric disorders (Saxena 1995).
Presynaptic 5-HT1A receptors mediate inhibition of 5-HT release and are highly concentrated on the cell bodies in the raphe nuclei (Romero et al. 1994). This receptor has been given a key role in current hypotheses on the treatment of anxiety and depression (Artigas et al. 1994; Baldwin and Rudge 1995; Berendsen 1995). An important observation is that (−)pindolol, a compound with affinity for β-adreno-and 5-HT1A receptors, antagonizes 5-HT1A-mediated response in the raphe nuclei (Hjorth and Carlsson 1986; Oksenberg and Peroutka 1988). Interestingly, pindolol may facilitate improvement of depressive patients by reducing the latency and enhancing the response rate to certain selective serotonin reuptake inhibitors (SSRI) (Artigas et al. 1994; Blier and Bergeron 1995). An augmenting or accelerating effect of pindolol has recently been confirmed in some placebo-controlled clinical studies (Perez et al. 1997; Tome et al. 1997; Zanardi et al. 1997), although negative results have also been reported (Berman et al. 1997; McAskill et al. 1998; Moreno et al. 1997; Perez et al. 1999). In another PET study, we have recently reported that pindolol binds specifically to 5-HT1A receptors in the living human brain (Andrée et al. 1999). This lends further support for a specific action and possible clinical implications of pindolol treatment.
Postsynaptic 5-HT1A receptors have been found in high densities in the hippocampus and in the superficial layers of the neocortex in man (Hall et al. 1997; Marazziti et al. 1994; Pazos et al. 1987). The high density of postsynaptic 5-HT1A receptors in the neocortex is of interest in schizophrenia research, since several studies have reported elevated 5-HT receptor binding in the neocortex and the hippocampus of post-mortal schizophrenic brains (Burnet et al. 1996; Gurevich and Joyce 1997; Hashimoto et al. 1991,1993; Joyce et al. 1993; Sumiyoshi et al. 1996). However, the antipsychotic potential of selective 5-HT1A receptor antagonists or agonists has not been explored in man.
Robalzotan with the substance name NAD-299, (R)-3-N,N-dicyclobutylamino-8-fluoro-3,4,dihydro-2H-1-benzopyran-5-carboxamide hydrogen (2R,3R)-tartate monohydrate), is a novel compound with high selectivity and affinity (0.6 nM) to 5-HT1A-receptors in the rodent brain in vitro and in vivo (Larsson et al. 1998; Stenfors et al. 1998). Functional and behavioral studies with robalzotan indicate antagonistic activity at the 5-HT1A-receptor (Johansson et al. 1997). The safety and tolerability of robalzotan is currently examined in healthy subjects.
[carbonyl-11C]WAY-100635 is a suitable radioligand for quantitative determination of 5-HT1A receptors in the monkey and human brain in vivo using positron emission tomography (PET) (Farde et al. 1997, 1998; Pike et al. 1996). This suitability has been supported by autoradiography studies, in which the radioligand [3H]WAY-100635 has been used for the characterization of the 5-HT1A receptor distribution in vitro (Hall et al. 1997; Pazos et al. 1987) showing a consistent result for the regional 5-HT1A receptor distribution as that shown in the living human brain by PET.
The aims of this PET study were to determine 5-HT1A receptor occupancy in the cynomolgus monkey brain in vivo after IV injection of robalzotan, and to establish the hyperbolic function between plasma concentration and receptor occupancy. Two healthy monkeys were examined with PET and the radioligand [carbonyl-11C]WAY-100635, the first after administration of 2 μg/kg and 20 μg/kg and the second after 10 μg/kg and 100 μg/kg IV. 5-HT1A receptor occupancy in the neocortex and the raphe nuclei were calculated using an equilibrium-ratio analysis with the cerebellum as reference region.
MATERIALS AND METHODS
This study was performed at the Department of Clinical Neuroscience at the Karolinska Hospital after approval by the Animal Ethics Committee of Northern Stockholm. Principles of laboratory animal care (NIH publication No. 85–23, revised 1985) were followed).
Compound
Robalzotan (NAD-299) was obtained from AstraZeneca AB, Södertälje, Sweden. The solutions of NAD-299 were prepared as an intravenous stock-solution (0.4 μmol/ml) batch 2230–4–1 according to GLP routines.
Radiochemistry
[carbonyl-11C]WAY-100635 was prepared by 11C acylation of the precursor WAY-100634 with [carbonyl-11C]-cyclobexanecarbonyl chloride, as previously described in detail (Hall et al. 1997; Pike et al. 1996). The specific radioactivity was about 2000 Ci/mmol (74 GBq/μmol) at EOS (end of synthesis). The radiochemical purity was higher than 99%.
Robalzotan (NAD-299) Concentration in Plasma
Venous blood samples (1–2 ml) were drawn from a femoral vein, before drug administration (0 minutes), at start of the PET measurement (20 minutes) and at about 40, 60, 80, and, after the time for the highest doses, 100 minutes. The exact timing was dependent on success with the venopuncture at each sampling. The mean plasma concentration values (Cpl.mean), determined for the time-interval 40–60 minutes after drug administration, were calculated and used for the determination of the hyperbolic function between plasma concentrations and 5-HT1A receptor occupancy. NAD-299 was isolated from monkey plasma samples by liquid-liquid extraction and analyzed by reversed phase liquid chromatography (LC) combined with atmospheric pressure chemical ionization (APCI) mass spectrometry (MS). Standard samples, prepared in monkey plasma, were used for the calibration curves. An automated LC-MS-MS system allowing unattended over-night operation was used. NAD-299 could be reliably measured down to 0.2 nM. See Figure 1.
The PET System
The PET system used (i.e., Siemens ECAT Exact HR47), has a spatial resolution in the imaging plane of about 3.8 mm FWHM (Full Width at Half Maximum) (Wienhard et al. 1994). The system was used in the three-dimensional mode and images were displayed in 47 horizontal sections. Radioactivity in the brain was measured continuously according to a pre-programmed sequence of frames for up to 93 minutes after injection of [carbonyl-11C]WAY-100635. See Figure 2.
PET Studies on Monkeys
Two cynomolgus monkeys with a weight of 3–4 kg were obtained from the National Laboratory of Bacteriology, Solna, Sweden. Anesthesia was induced by repeated IM injection of ketamine (Ketalar® 5–10 mg kg−1 h−1). Blood pressure and pulse rate were recorded every 20 minutes. Temperature was kept at about 37°C using a heating pad that was regulated by the rectal temperature. A head fixation system secured a fixed position of the monkeys' head in a plane parallel to that defined by the canto-meatal line (Karlsson et al. 1993).
Each of the two monkeys participated in three PET-measurements that were performed on the same day. The first was performed at baseline conditions, the second after a low dose of robalzotan that was administered IV, and the third after a 10-times higher dose (20, 100 μg/kg). The 5-HT1A receptor occupancy values in the first monkey were used to select suitable doses that were administered to the second monkey. The strategy was to select doses that should cover a wide occupancy interval.
In each PET-measurement, a sterile physiological phosphate buffer (pH 7.4) solution containing about 1.3–1.5 mCi (48–56 MBq) of [carbonyl-11C]WAY-100635 was injected as a bolus into a sural vein during 2 seconds. The specific radioactivity (SR) was 867–2039 Ci/mmol (32–75 GBq/μmol) for the measurements in the first monkey, and 192–905 Ci/mmol (7.1–33 GBq/μmol) for the second, at the time of IV administration. Each PET-measurement was performed at expected robalzotan peak plasma levels.
Regions of Interest
For analysis of regional brain radioactivity, regions of interest (ROIs) were drawn on the reconstructed PET images. ROIs were drawn for the whole brain, the neocortex, the raphe nuclei, and the cerebellum. The anatomical delineation of regions was guided by an atlas showing a series of horizontal sections of a cynomolgus monkey brain in situ obtained by serial cryomicrotomy (Karlsson et al. 1993).
Calculation of 5-HT1A Receptor Occupancy by the Ratio Method
Regional radioactivity was determined for each ROI measured for each sequential time-frame, corrected for 11C-decay and plotted versus time. The 5-HT1A receptor occupancy induced by pretreatment was calculated according to a procedure that has been described previously (Farde et al. 1992, 1997). Assuming negligible specific binding, the cerebellum can be used as reference region for free and non-specific binding (Cf) in brain. Specific [carbonyl-11C]WAY-100635 binding (Cb) to 5-HT1A receptors was calculated as the difference between total binding (Ct) in a ROI and cf. Receptor occupancy (%) was defined as the percent reduction of Cb/Cf. The ratio Cb/Cf was calculated for the 20–40 minutes time interval of each PET-measurement according to Equation 1: 
Following an IV bolus injection, the relation between receptor binding and the concentration of ligand is given by a hyperbolic (curvilinear) function, derived from the law of mass action (Farde et al. 1986, 1989; Ito et al. 1998), according to Equation 2:  where B is the concentration of ligand (i.e., robalzotan), bound to receptors, Bmax is the total number of available receptors, KD is the apparent equilibrium dissociation constant, and F is the concentration of free (unbound) ligand. Assuming that the relationship between robalzotan in brain and plasma and the free fraction are not concentration dependent, F may be substituted with the robalzotan plasma concentration (Farde et al. 1989), that, thus, represents an estimate of the free concentrate in brain (Karlsson et al. 1995b). All receptors (Bmax) are occupied at 100% occupancy. Replacing F with robalzotanpl. and Bmax with 100%, Equation 1 can be rewritten as Equation 3:
where B is the concentration of ligand (i.e., robalzotan), bound to receptors, Bmax is the total number of available receptors, KD is the apparent equilibrium dissociation constant, and F is the concentration of free (unbound) ligand. Assuming that the relationship between robalzotan in brain and plasma and the free fraction are not concentration dependent, F may be substituted with the robalzotan plasma concentration (Farde et al. 1989), that, thus, represents an estimate of the free concentrate in brain (Karlsson et al. 1995b). All receptors (Bmax) are occupied at 100% occupancy. Replacing F with robalzotanpl. and Bmax with 100%, Equation 1 can be rewritten as Equation 3: 
5-HT1A receptor occupancy was plotted versus the mean plasma concentrations of robalzotan 40–60 minutes after robalzotan administration. This time corresponds to 20–40 minutes after injection of the radioligand [carbonyl-11C]WAY-100635. Each data point was treated as an independent observation, and Equation 1 was fitted in a least square sense through an iterative procedure to the experimental data points, using Kaleidagraph® 3.0 (Abelbeck Software, USA).
RESULTS
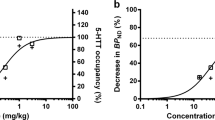
The plasma concentrations of robalzotan increased with dose and was maximal (Cmax) at time of start of the PET-measurements, 20 minutes after IV drug administration (Tmax) (Table 1 ). During the first PET-measurement, performed at baseline conditions, there was a high uptake of radioactivity in the neocortical regions and the raphe nuclei, whereas the radioactivity in the cerebellum was low (Figure 3). After pretreatment with robalzotan, the radioactivity in the neocortex and the raphe nuclei was reduced in a dose-dependent manner (Figure 3). The mean plasma concentration values (Cpl.mean), determined for the time-interval 40–60 minutes after drug administration, was 0.5, 4, 7.2, and 31.4 nmol/l, respectively after robalzotan administration of 2, 10, 20, and 100 μg/kg as an IV injection (Table 1). The highest occupancy (70–80%) was calculated for the measurement after the 100 μg/kg dose (Table 1).
The curvilinear function for a saturation hyperbola was fitted to the experimental data points (Karlsson et al. 1995a). The results indicate that 50% of the 5-HT1A receptors in the raphe nuclei are occupied at a robalzotan plasma concentration of 3.9 nmol/L. The corresponding value for the neocortex was 5.4 nmol/L (Figure 4).
DISCUSSION
After IV injection of robalzotan in the two cynomolgus monkeys, there was a binding of [carbonyl-11C]WAY-100635 to 5-HT1A receptors (Figure 3). The results indicate that robalzotan (generic name NAD-299) rapidly passes the blood-brain barrier and binds to central 5-HT1A receptors in the cynomolgus brain in vivo.
The relationship between robalzotan plasma concentration and 5-HT1A receptor occupancy after IV administration of robalzotan could be described by a hyperbolic function, both for the neocortex and the raphe nuclei (Figure 4), which can be used to guide the selection of appropriate doses for initial studies in man. The study demonstrates that quantitative neuroimaging of receptor binding has potential for the evaluation and dose finding of new CNS drugs.
The 5-HT1A receptor occupancy values were numerically higher in the raphe nuclei in all four measurements compared to the slightly lower receptor occupancy calculated for the neocortical region. However, it must be taken into account that this difference is based on pooled data from four measurements in two monkeys only, therefore, these values should be taken with caution. Considering the small volume of the monkey brain (65 ml) and the raphe nuclei in relation to the resolution of the PET-system (3.8 mm FWHM), it cannot be concluded that robalzotan binds preferentially to 5-HT1A receptors in the raphe nucleus.
The predominant metabolite of the robalzotan in vitro in all species tested including rat, dog, and man, is the mono-N-dealkylated derivative (Gagner Milchert et al. 1998). This metabolite has more than thousand fold less affinity for the 5-HT1A receptor than the parent compound (personal communications, Seth-Olov Thornberg, AstraZeneca). Hydroxylated robalzotan and its dealkylated derivative are other labeled metabolites which have been suggested in a PET-study with [11C]robalzotan in cynomolgus monkeys (Sandell et al. 1999a). The hydroxylated metabolites are polar and not likely to pass the blood-brain barrier and contribute to the 5-HT1A receptor occupancy.
The expected rapid metabolism was, nevertheless, a reason for starting the PET-measurements as early as about 20 minutes after robalozotan IV administration. According to a PET study with [11C]robalzotan in cynomolgus monkey, this time is sufficient to allow robalzotan to be distributed to brain and to reach binding equilibrium (Sandell et al. 1999a). The plasma curves confirm that the PET-measurements started at peak plasma levels (Cmax) in all four measurements (Figure 3A, B, C, D).
Recently, we have labeled robalzotan (NAD-299) with carbon-11 for visualization of the 5-HT1A receptor (Sandell et al. 1999b). Autoradiographic examination of [11C]NAD-299 binding in the post mortem human brain demonstrated high specific binding in the hippocampus, the raphe nuclei and the neocortex. The results indicate that 11C-labeled NAD-299 has potential as radioligand for PET examination of 5-HT1A receptors in vivo.
CONCLUSION
This PET-study confirms that robalzotan (NAD-299) passes the blood-brain barrier and binds to central 5-HT1A receptors in a saturable and dose-dependent manner, in the primate brain in vivo. The relationship between robalzotan drug concentrations and 5-HT1A receptor occupancy could be described by a hyperbolic function that can be used to guide the selection of appropriate doses for the initial studies in man.
References
Andrée B, Thorberg S-O, Halldin C, Farde L . (1999): Pindolol binding to 5-HT1A receptors in the human brain confirmed with Positron Emission Tomography. Psychopharmacology 144: 303–305
Artigas F, Perez V, Alvarez E . (1994): Pindolol induces a rapid improvement of depressed patients treated with serotonin reuptake inhibitors. Arch Gen Psychiatry 51: 248–251
Baldwin D, Rudge S . (1995): The role of serotonin in depression and anxiety. Intl Clin Psychopharmacol 9: 41–45
Berendsen HH . (1995): Interactions between 5-hydroxy-tryptamine receptor subtypes: Is a disturbed receptor balance contributing to the symptomatology of depression in humans? Pharmacol Therap 66: 17–37
Berman RM, Darnell AM, Miller HL, Anand A, Charney DS . (1997): Effect of pindolol in hastening response to fluoxetine in the treatment of major depression: A double-blind, placebo-controlled trial. Am J Psychiatry 154: 37–43
Blier P, Bergeron R . (1995): Effectiveness of pindolol with selected antidepressant drugs in the treatment of major depression. J Clinical Psychopharmacol 15: 217–222
Burnet PW, Eastwood SL, Harrison PK . (1996): 5-HT1A and 5-HT2A receptor mRNAs and binding site densities are differentially altered in schizophrenia. Neuropsychopharmacology 15: 442–455
Farde L, Eriksson L, Blomquist G, Halldin C . (1989): Kinetic analysis of central [11C]raclopride binding to D2-dopamine receptors studied by PET—a comparison to the equilibrium analysis. J Cereb Blood Flow Metab 9: 696–708
Farde L, Gionvart N, Ito H, Lundkvist C, Pike VW, McCarron JA, Halldin C . (1997): PET-characterization of [carbonyl-11C]WAY-100635 binding to 5-HT1A receptors in the primate brain. Psychopharmacology 133: 196–202
Farde L, Hall H, Ehrin E, Sedvall G . (1986): Quantitative analysis of D2 dopamine receptor binding in the living human brain by PET. Science 231: 258–261
Farde L, Ito H, Swahn C-G, Pike VW, Halldin C . (1998): Quantitative analysis of [carbonyl-11C]WAY-100635 binding to central HT1A receptors in man. J Nucl Med 39: 1965–1971
Farde L, Nordström AL, Wiesel F-A, Pauli S, Halldin C, Sedvall G . (1992): Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Arch Gen Psychiatry 49: 538–544
Gagner Milchert I, Gabrielson J, Lindhe J, Terelius Y . (1998): In vitro- in vivo extrapolation of robalzotan metabolic parameters: An interspecies comparison. Eur Neuropsychopharmacol 8: 184
Gurevich EV, Joyce JN . (1997): Alterations in the cortical serotonergic system in schizophrenia: A postmortem study. Biol Psychiatry 42: 529–545
Hall H, Lundkvist C, Halldin C, Farde L, Pike VW, McCarron JA, Fletcher A, Cliffe IA, Barf T, Wilkstrom H, Sedvall G . (1997): Autoradiographic localization of 5-HT1A receptors in the post-mortem human brain using [3H[WAY-100625 and [11C]way-100635. Brain Res 745: 96–108
Hashimoto T, Kitamura N, Kajimoto Y, Shirai Y, Shirakawa O, Mita T, Nishino N, Tanaka C . (1993): Differential changes in serotonin 5-HT1A and 5-HT2 receptor binding in patients with chronic schizophrenia. Psychopharmacology 112: S35–S39
Hashimoto T, Nishino N, Nakai H, Tanaka C . (1991): Increase in serotonin 5-HT1A receptors in prefrontal and temporal cortices of brains from patients with chronic schizophrenia. Life Sci 48: 355–363
Hjorth S, Carlsson A . (1986): Is pindolol a mixed agonist-antagonist at central serotonin (5-HT) receptors? Eur J Pharmacol 129: 131–138
Hoyer D, Clarke DE, Fozard JR, Hartig PR, Martin GR, Mylecharane EJ, Saxena PR, Humphrey PP . (1994): International Union of Pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol Rev 46: 157–203
Ito H, Hietala J, Blomqvist G, Halldin C, Farde L . (1998): Comparison of the transient equilibrium and continuous infusion method for quantitative PET analysis of [11C]raclopride binding. J Cereb Blood Flow Metab 18: 941–950
Johansson L, Sohn D, Thornberg SO, Jackson DM, Kelder D, Larsson LG, Renyi L, Ross SB, Wallsten C, Eriksson H, Hu PS, Jerning E, Mohell N, Westlind-Danielson A . (1997): The pharmacological characterization of a novel selective 5-hydroxytryptamine1A receptor antagonist, NAD-299. J Pharmacol Exp Therap 283: 216–225
Joyce JN, Shane A, Lexow N, Winokur A, Casanova MF, Kleinman JE . (1993): Serotonin uptake sites and serotonin receptors are altered in the limbic system of schizophrenics. Neuropsychopharmacology 8: 315–336
Karlsson P, Farde L, Halldin C, Sedvall G, Skrumsager B, Foged C . (1993): [11C]NNC687 and [11C]NNC756 as radioligands for PET-determination of central D1-dopamine receptors. Psychopharmacology 113: 149–156
Karlsson P, Farde L, Halldin C, Sedvall G, Ynddal L, Sloth-Nielsen M . (1995a): Oral administration of NNC 756&—a placebo controlled PET study of D1-dopamine receptor occupancy and pharmacodynamics in man. Psychopharmacology 119: 1–8
Karlsson P, Farde L, Halldin C, Swahn C, Sedvall G . (1995b): Evaluation of [11C]SCH39166 as D1-dopamine receptor ligand for central D1 dopamine receptor binding and occupancy in man. Psychopharmacology 121: 309–316
Larsson LG, Stenfors C, Ross SB . (1998): Differential regional antagonism of 8-OH-DPAT-induced decrease in serotonin synthesis by two 5-HT1A receptor antagonists. Eur J Pharmacol 346: 209–215
Marazziti D, Marracci S, Palego L, Rotondo A, Mazzanti C, Nardi I, Ladinsky H, Giraldo E, Borsini F, Cassano GB . (1994): Localization and gene expression of serotonin 1A (5HT1A): Receptors in human brain postmortem. Brain Res 658: 55–59
McAskill R, Mir S, Taylor D . (1998): Pindolol augmentation of antidepressant therapy [published erratum appears in Br J Psychiatry 173:443, 1998]. Br J Psychiatry 173: 203–208
Moreno FA, Gelenberg AJ, Bachar K, Delgado PL . (1997): Pindolol augmentation of treatment-resistant depressed patients. J Clin Psychiatry 58: 437–439
Oksenberg D, Peroutka SJ . (1988): Antagonism of 5-hydroxytryptamine1A (5-HT1A): receptor-mediated modulation of adenylate cyclase activity by pindolol and propranolol isomers. Biochem Pharmacol 37: 3429–3433
Pazos A, Probst A, Palacios JM . (1987): Serotonin receptors in the human brain. III. Autoradiographic mapping of serotonin-1 receptors. Neuroscience 21: 97–122
Perez V, Gilaberte I, Faries D, Alvarez E, Artigas F . (1997): Randomised, double-blind, placebo-controlled trial of pindolol in combination with fluoxetine antidepressant treatment. Lancet 349: 1594–1597
Perez V, Soler J, Puigdemont D, Alvarez E, Artigas F . (1999): A double-blind, randomized, placebo-controlled trial of pindolol augmentation in depressive patients resistant to serotonin reuptake inhibitors. Grup de Recerca en Trastorms Afectius. Arch Gen Psychiatry 56: 375–379
Peroutka SJ, Howell TA . (1994): The molecular evolution of G protein-coupled receptors: Focus on 5-hydroxytryptamine receptors. Neuropharmacology 33: 319–324
Pike VW, McCarron JA, Lammertsma AA, Osman S, Hume SP, Sargent PA, Bench CJ, Cliffe IA, Fletcher A, Grasby PM . (1996): Exquisite delineation of 5-HT1A receptors in human brain with PET and [carbonyl-11C]WAY-100635. Eur J Pharmacol 301: R5–R7
Romero L, Celada P, Artigas F . (1994): Reduction of in vivo striatal 5-hydroxytryptamine release by 8-OH-DPAT after inactivation of Gi/G(o) proteins in dorsal raphe nucleus. Eur J Pharmacol 265: 103–106
Sandell J, Halldin C, Chou Y-W, Thornberg S-O, Sohn D, Werner T, Farde L . (1999a): Metabolic studies measured by HPLC in monkey plasma of the new 5-HT1A radioligand [C-11]NAD-299. J Labell Comp Radiopharm 42(Suppl 1):675–677
Sandell J, Halldin C, Hall H, Thorberg S-O, Werner T, Sohn D, Sedvall G, Farde L . (1999b): Radiosynthesis of autoradiographic evaluation of [11C]NAD-299, a radioligand for visualization of the 5-HT1A receptor. Nucl Med Biol 26: 159–164
Saxena PR . (1995): Serotonin receptors: Subtypes, functional responses and therapeutic relevance. Pharmacol Therap 66: 339–368
Stenfors C, Werner T, Ross SB . (1998): In vivo labelling of the mouse brain 5-hydroxytryptamine 1A receptor with the novel selective antagonist 3H-NAD-299. Naunyn-Schmiedebergs Arch Pharmacol 357: 500–507
Sumiyoshi T, Stockmeier CA, Overholser JC, Dilley GE, Meltzer HY . (1996): Serotonin 1A receptors are increased in postmortem prefrontal cortex in schizophrenia. Brain Res 708: 209–214
Tome MB, Isaac MT, Harte R, Holland C . (1997): Paroxetin and pindolol: A randomized trial of serotonergic autoreceptor blockade in the reduction of antidepressant latency. Intl Clin Psychopharmacol 12: 81–89
Wienhard K, Dahlbom M, Eriksson L, Michel C, Bruckbauer T, Pietrzyk U, Heiss W . (1994): The ECAT EXACT HR: Performance of a new high resolution position scanner. J Comp Ass Tomogr 18: 110–118
Zanardi R, Artigas F, Franchini L, Sforzini L, Gasperini M, Smeraldi E, Perez J . (1997): How long should pindolol be associated with paroxetine to improve the antidepressant response? J Clin Psychopharmacol 17: 446–450
Acknowledgements
The members of the PET group at Karolinska Institutet and collaborators at AstraZeneca AB are gratefully acknowledged. The study was supported by a grant from AstraZeneca AB, Södertälje and the Human Frontier Science Program Organization (HFSPO). RG0235/1998-B.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Farde, L., Andrée, B., Ginovart, N. et al. PET-Determination of Robalzotan (NAD-299) Induced 5-HT1A Receptor Occupancy in the Monkey Brain. Neuropsychopharmacol 22, 422–429 (2000). https://doi.org/10.1016/S0893-133X(99)00125-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S0893-133X(99)00125-6
Keywords
This article is cited by
-
Modeling of PET data in CNS drug discovery and development
Journal of Pharmacokinetics and Pharmacodynamics (2013)
-
Occupancy of dopamine D2 and D3 and serotonin 5-HT1A receptors by the novel antipsychotic drug candidate, cariprazine (RGH-188), in monkey brain measured using positron emission tomography
Psychopharmacology (2011)
-
Positron emission tomographic analysis of dose-dependent NAD-299 binding to 5-hydroxytryptamine-1A receptors in the human brain
Psychopharmacology (2003)