Abstract
Sensitivity to chemicals in humans has been proposed to be an acquired disorder in which individuals become increasingly sensitive to chemicals in the environment. A possible link between the manifestation of psychiatric symptoms in individuals claiming sensitivity to chemicals was investigated based on a leading hypothesis put forth by Bell and co-workers (1992) to explain the amplification of symptoms after chemical exposure. The hypothesis is that chemical sensitivities may be akin to sensitization observed in rodents after repeated psychostimulants. Repeated exposure to psychostimulants enhances behavioral activity and the underlying neurochemical responses in specific limbic pathways; a similar sensitization of limbic pathways has been proposed to occur in individuals who become sensitive to chemicals. To test this hypothesis, female Sprague-Dawley rats were exposed to either air or formaldehyde (Form) for 1 h/day for 7 days or 20 days (5 days/week × 4 weeks). Two to 4 days after the last exposure, rats were given a cocaine challenge (= early withdrawal) followed by an additional cocaine challenge 4–6 weeks later (= late withdrawal). No differences in cocaine-induced locomotor activity were noted between groups after 7 days of exposure. However, after 20 days of exposure to Form, vertical activity was significantly elevated at both early and late withdrawal times. These studies demonstrate that behavioral sensitization occurs after long-term, but not short-term, low-level exposure to Form, and lends support to the limbic system sensitization hypothesis of sensitivity to chemicals in humans.
Similar content being viewed by others
Main
Chemical sensitivity in humans is an ill-defined disorder occurring in a subset of the population and has been attributed to exposure to chemicals, usually volatile organic compounds. Individuals who develop sensitivity to chemicals report an array of symptoms involving the central nervous system (CNS) and other organ systems; among the symptoms are fatigue, depression, headaches, gastrointestinal problems, muscle and joint pain, irritability, memory and concentration difficulties, and many others (Ashford and Miller 1991; Miller 1994). Although a case definition for chemical sensitivity has not been agreed upon, the common feature of chemical sensitivity or intolerance is found in individuals experiencing symptoms as a result of various exposure settings, such as Gulf War veterans (Gulf War Syndrome), industrial workers, and those living near hazardous waste sites. Onset of chemical sensitivity is purported to occur after single, high-dose exposure (such as in a chemical spill) or repeated low-level exposure to chemicals (Ashford and Miller 1991). There is much controversy regarding the existence of chemical sensitivity in humans, which stems largely from (1) the inability to rigorously identify true sensitivities to chemicals due to the unreliability of self-reports linking illness to chemical exposure, (2) the diversity of symptoms and their overlap with other illness, such as somatoform disorder, chronic fatigue syndrome, fibromyalgia, panic disorder, and posttraumatic stress disorder (PTSD), and (3) possible misattribution of symptoms from other illness to chemical exposure (Black et al. 1990; Dager et al. 1987; Schottenfeld and Cullen 1985; Simon et al. 1990; Staudenmayer and Selner 1987, 1990; Terr 1986). Typically, those with chemical sensitivity complain of ill effects from chemicals that are present in very low concentrations in the environment, suggesting that an amplification has occurred in the processes involved with either the perception of illness from chemicals or the effects of these compounds on the system.
We sought to gain insight on the possible link between manifestation of psychiatric symptoms and chemical exposure based on one of the leading hypotheses put forth by Bell et al. (1992) to explain the amplification process in human chemical sensitivities. They have noted that chemical sensitivity strongly resembles the phenomenon of sensitization in rodents observed after repeated exposure to psychostimulants or stress. They hypothesize that amplification of responses in chemically sensitive individuals develops via a CNS sensitization process, with particular emphasis on limbic system components due to their relatively high sensitivity to various perturbations (such as electrical and chemical kindling), and widespread involvement in cognitive and affective dysfunctions, as are observed in individuals with chemical sensitivity (Ashford and Miller 1991; Dager et al 1987; Levy 1988). Sensitization is defined as the progressive and enduring enhancement in behavioral and neurochemical measures after either a single exposure or repeated, intermittent exposure to a variety of stimuli, the most common of which are psychostimulants and environmental stressors (Antelman 1988; Antelman et al. 1992; Caggiula et al. 1989; Robinson and Becker 1986). Several features of sensitization appear parallel to those of chemical sensitivity, as previously discussed (Antelman 1994; Bell et al. 1992; Sorg et al. 1994). The similarities include: the progressive increase in sensitivity to drugs/chemicals (Bell 1994; Bell et al. 1997; Post and Weiss 1988); the apparent permanence of sensitivity (Ashford and Miller 1991; Robinson and Becker 1986); the lack of symptoms/sensitization in the absence of chemical/drug and the onset of symptoms/sensitization upon chemical challenge (Ashford and Miller 1991; Miller 1994; Robinson and Becker 1986); the greater sensitivity of females versus males (Camp and Robinson 1988; Miller 1994); the spreading of sensitization in response to stimuli other than the initial stimulus used to induce sensitization (as with cross-sensitization between psychostimulants and stress) (Antelman et al. 1980; Miller 1994); and the apparent time-dependent nature of sensitization, wherein responses increase with the passage of time (Antelman et al. 1992; Bell et al. 1992; Caggiula et al. 1989; Paulson and Robinson 1995).
Sensitization by psychostimulants and stress has been proposed as a model for psychiatric disorders including paranoid psychosis, panic disorder, and PTSD (Antelman 1988; Post and Weiss 1988). The model is based on several similar characteristics with regard to the amplification and persistence of a response to outside stimuli, exacerbation by stressors, and the ability to condition to stimuli, among others (Friedman 1994). Based on this information and the sensitization hypothesis of chemical sensitivity, this laboratory has recently attempted to design an animal model for chemical sensitivity by testing whether repeated chemical exposure produces sensitization of the CNS. The study investigated whether rats repeatedly exposed to formaldehyde (Form) vapors would cross-sensitize to cocaine's effects on locomotor activity. Formaldehyde is among the most ubiquitous volatile organic compounds found in indoor air, present in hundreds of common products such as paper, insulation, wood products and resins, and appears to produce illness in many humans with sensitivity to chemicals (Ashford and Miller 1991). Repeated Form inhalation produced a cross-sensitization to subsequent cocaine-induced locomotion similar in magnitude to sensitization induced by repeated cocaine, suggesting that specific CNS components mediating the motor-stimulant effects of cocaine were upregulated (Sorg et al. 1996). Although this was a first step toward supporting the sensitization hypothesis of chemical sensitivity, the magnitude of Form levels administered (approximately 11 ppm) exceeded the normal range of human exposures (approximately 1 ppm). Irritant levels in humans are reported to occur at approximately 0.1–1.0 ppm (see Kane and Alarie 1977). Typical occupational and home exposure levels are less than 0.2 ppm, but can reach 1–2 ppm (Clary 1983; Kane and Alarie 1977; Spengler 1991). The goal of the present work was to determine if repeated low-level exposure to Form in the range of human exposures would also produce a cross-sensitivity to cocaine's effects. A second aim was to determine if cocaine cross-sensitivity to Form exposure was long lasting. Two additional behavioral tests were chosen based on observations of increased anxiety and increased somatization in individuals with chemical sensitivity (Ashford and Miller 1991). An anxiety test using the elevated plus-maze and a hotplate test for measuring nociceptive levels were used to examine animals repeatedly exposed to Form.
METHODS
Animals and Apparatus
Female Sprague-Dawley rats obtained from Simonsen Laboratories (Gilroy, CA) were group-housed four per cage in a temperature- and humidity-controlled room with food and water available ad libitum except for the time during which they were placed into the exposure chambers or the behavioral testing apparatus. The room was maintained on a 12-h light/dark cycle, with lights on at 0700 h. All protocols were carried out in accordance with the Guide for the Care and Use of Laboratory Animals as adopted by the National Institutes of Health.
Form gas was generated by the depolymerization of 1 g paraformaldehyde placed into a glass test tube measuring 2 × 12 cm. The test tube was placed inside a permeation tube holder, which was immersed in a water bath heated to 70°C. Air was pumped over the Form at a rate of 2.5 L/min past the exit port of each of four sealed Plexiglas chambers measuring 26 cm × 29 cm containing a wire mesh floor. Formaldehyde levels present in the vapor were determined by passing the sample air over a cartridge containing dinitrophenylhydrazine (DNPH). After 5-min collections, the cartridges were stored at 5°C until analysis. The sample and blank cartridges were eluted with acetonitrile. Hydrazone concentrations in the eluent were determined by reverse-phase high performance liquid chromatography as previously described (Sirju and Shepson 1995). Locomotor activity measurements and the elevated plus-maze and hotplate tests were conducted as previously described (Sorg et al. 1996).
Experimental Design
Rats were placed in groups of four into the Plexiglas chambers according to the grouping in their home cages. Two separate experiments were performed. In the first experiment, rats were exposed to air (control condition) or Form vapors for 1 h/day for 7 days. In the second experiment, a separate group of rats was exposed for 1 h/day for 20 days (5 days/week × 4 weeks; no exposure on weekends). All exposures were carried out during the first half of the light cycle. For the first experiment, the same 1 g of paraformaldehyde was used for each of the seven daily exposures. For the second experiment, the same 1 g of paraformaldehyde was used for the first 10 days of exposure, and replaced with a fresh 1 g of paraformaldehyde for the second 10-day exposure. Formaldehyde levels measured were as follows (mean ± SEM): day 1: 1.43 ± 0.141 ppm; day 7: 0.723 ± 0.044 ppm, and day 10: 0.633 ± 0.012 ppm. Thus, a gradual decline in Form levels occurred with daily exposures in experiment 1, whereas in experiment 2, levels declined until 10 days, at which time the procedure using 1 g fresh paraformaldehyde was repeated for the second 10-day period.
Two to 4 days after the last air or Form exposure, rats were placed into photocell cages for monitoring horizontal and vertical activity. Rats were placed into the photocell apparatus and allowed to habituate for 1 h after which time saline (1 mL/kg IP) was administered, and activity was monitored for an additional 1-h period. The following day, the animals’ response to cocaine hydrochloride (15 mg/kg IP; gift from the National Institute on Drug Abuse) was monitored in the same manner as for saline except that postinjection activity was measured for 2 h. For experiment 1, rats were given an additional cocaine challenge as above 4 weeks after discontinuing daily air or Form exposure. For experiment 2, rats were given both an additional saline and cocaine challenge (1–6 days after saline) 4–6 weeks after discontinuing daily air or Form exposure.
For both experiments, within 2 weeks after discontinuing daily air or Form treatment, two additional behavioral tests were conducted (during the time between early and late cocaine challenges). For the elevated plus-maze test, rats were removed from their home cages and allowed to habituate to the test room for a minimum of 30 min. Rats were placed into the center of the maze (neutral zone), and the amount of time spent in the open and closed arms as well as the number of open and closed arm entries was recorded over a 5-min period in the presence of an experimenter who was unaware of the treatment groups. Animals were considered to be in open or closed arms only when all four limbs crossed out of the neutral zone. Assessment of changes in nociception between treatment groups was done using a hotplate test. Animals were removed from their home cage one at a time, placed onto the hotplate apparatus (52°C), and the latency to lick the hind paw was measured. Thereafter, each rat was immediately returned to its home cage.
Data Analysis
All cumulative photocell counts over the 1- or 2-h period as well as the plus-maze and hotplate results were analyzed with a one-way analysis of variance (ANOVA) followed by a Scheffe's F-test in the case of a significant difference (p < .05). For the time course data, a two-way ANOVA with repeated measures over time was conducted followed by a least significant difference (LSD) test in the case of a significant interaction (p < .05).
RESULTS
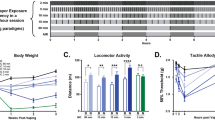
Body weights measured at early and late withdrawal times revealed no clear pattern of effects by Form. In rats exposed for 7 days, mean ± SEM body weights (in gs) at early withdrawal were 289 ± 6 (control), 304 ± 6 (Form) (p = .087), and at late withdrawal were 305 ± 8 (control), 335 ± 21 (Form) (p = .0034). In rats exposed for 20 days, mean ± SEM body weights at early withdrawal were 259 ± 2 (control), 252 ± 2 (Form) (p = .024), and at late withdrawal were 267 ± 2 (control), 265 ± 2 (Form) (p = .452).
Response to 7 Days Form
The data summarized in Figures 1A and C , left panels, demonstrate that after 7 days of Form exposure, neither vertical nor horizontal activities in response to a saline injection were different between treatment groups 2–4 days after discontinuing daily exposure. Whereas a cocaine challenge elicited an increase in vertical and horizontal activity, no differences were present between Form- or air-exposed animals at either early (Figures 1A and C, right panels) or late (Figures 1B and D) withdrawal times. At late withdrawal (4 weeks after the last Form exposure), cocaine-induced behavioral activity was significantly decreased compared to that from the early withdrawal period (p < .0001, paired t-test, two tailed).
Saline- and cocaine-induced activity in 7-day air and Form pretreated rats. Values are mean ± SEM of photocell counts obtained over a 1-h period on the saline day or for a 2-h period on the cocaine day. n = 15 for air-exposed controls (Con); n = 16 for Form group. (A and C): Early withdrawal, saline and cocaine challenge were administered within 2–4 days after discontinuing daily exposure. (B and D): Late withdrawal, cocaine challenge was administered 4 weeks after discontinuing daily exposure.
Response to 20 Days Form
In the second experiment in which rats were exposed to daily Form for 20 days (5 days/week × 4 weeks), a saline challenge did not produce differences in activity between groups at either withdrawal period (Figures 2A, B, C, D , left panels). However, at early withdrawal, cocaine produced hyperactivity in controls, which was significantly enhanced in Form-exposed animals for vertical activity (Figure 2A, right panel). This augmentation was maintained for up to 4–6 weeks when rats were given a second cocaine challenge (Figure 2B, right panel). A paired t-test revealed a significant increase in cocaine-induced vertical activity from air-exposed controls when comparing early to late withdrawal times (p = .0126). Unlike for vertical activity, cocaine-induced horizontal movements were not significantly different between treatment groups at either early or late withdrawal periods (Figures 2C and D, right panels). Figures 3A and B show the time course for cocaine-induced vertical activity obtained from the cumulative photocell counts at early and late withdrawal (from Figures 2A and B, right panels). At early withdrawal, Form-pretreated rats demonstrated cocaine-induced vertical activity that was significantly enhanced over much of the time course after cocaine injection; this effect was also maintained at the late withdrawal time.
Saline- and cocaine-induced activity in 7-day air and Form pretreated rats. Values are mean ± SEM of photocell counts obtained over a 1-h period on the saline day or for a 2-h period on the cocaine day. n = 20–24/group (in some cases, malfunction of photocell equipment occurred). (A and C): Early withdrawal, saline and cocaine challenge were administered within 2–4 days after discontinuing daily exposure. (B and D): Late withdrawal, saline and cocaine challenge were administered 4–6 weeks after discontinuing daily exposure. *p < .05, as determined by a one-way analysis of variance followed by a Scheffe's F-test. For early withdrawal, F1,46 = 29.11, p < .0001; for late withdrawal, F1,40 = 9.02, p = .0046).
Time course of cocaine-induced vertical activity in 20-day air and Form pretreated rats. Data are taken from cumulative photocell counts shown in Figure 2A and Figure 2B, right panels. Values are mean ± SEM of photocell counts obtained over each 15-min collection period. See Figure 2 legend for number of observations. Results from a two-way ANOVA for (A): preinjection: treatment F1,45 = 4.11, p = .0485, time F3,138 = 166.18, p < .0001, interaction F3,138 = 1.02, p = .385; postinjection: treatment F1,46 = 29.11, p < .0001, time F7,322 = 22.93, p < .0001, interaction F7,322 = 8.12, p < .0001. For (B): preinjection: treatment F1,40 = 6.33, p = .016, time F3,120 = 144.81, p < .0001, interaction F3,120 = 0.63, p = .600; postinjection: treatment F1,40 = 9.02, p = .0046, time F7,280 = 32.27, p < .0001, interaction F7,280 = 3.97, p = .0004. *p < .05, as determined by a two-way ANOVA followed by an LSD test.
Anxiety and Nociception
Table 1 shows the results from the elevated plus-maze test for anxiety and the hotplate test for the nociceptive response. Regardless of whether the daily exposure treatment was presented for 7 or 20 days, no significant differences in performance on the plus-maze or hotplate were detected among groups. However, there was a strong trend for rats exposed to Form for 7 days to demonstrate a decrease in the percent of time on the open arms of the plus-maze (p = .0614).
DISCUSSION
The main findings of this study are: (1) long-term Form exposure (approximately 1 ppm, 1 h/day × 5 days/week × 4 weeks) produced long-lasting cross-sensitization to the motor-stimulant effects of cocaine, whereas short-term exposure to Form inhalation (1 h/day × 7 days) did not produce cross-sensitization to cocaine-induced activity; (2) neither anxiety performance as measured on an elevated plus-maze nor nociception as measured on a hotplate was significantly altered by any of the treatments when rats were tested under nonchallenge conditions (i.e., in a nondrug or chemical state).
Rodents sensitized to psychostimulants such as cocaine or amphetamine or to stressors demonstrate an augmentation in locomotor activity in response to a subsequent challenge of psychostimulant (for review, see Kalivas and Stewart 1991). One major mechanism contributing to the enhanced locomotor activity in sensitized animals involves the mesolimbic dopamine system. Extracellular dopamine levels in the ventral striatum (nucleus accumbens) are augmented in animals sensitized to psychostimulants or stress, and this enhancement is believed to partially contribute to the sensitized behavioral response (Kalivas and Duffy 1990; Parsons and Justice 1993; Robinson et al. 1988; Sorg and Kalivas 1991). Thus, cross-sensitization with cocaine-induced activity suggests that the function of specific limbic regions such as the mesolimbic dopamine system may be altered by long-term exposure to Form inhalation. Future experiments will address this issue. Previous work has reported enhanced apomorphine-induced locomotor activity (von Euler et al. 1991) and an increase in dopamine D2 receptor binding in the striatum (von Euler et al. 1993) after long-term, low-level toluene exposure. Thus, either increased dopaminergic neurotransmission and/or increased postsynaptic responsiveness to dopamine in the nucleus accumbens may in part account for these as well as the present findings.
This study provides evidence that the potential for altered limbic system function is present after repeated exposure to a chemical present in low levels. Sensitization of neural substrates hypothesized to underlie psychiatric disorders such as panic disorder and PTSD may thus explain some of the symptoms observed in chemically sensitive individuals (Friedman and Schnurr 1995; Levy 1988). It should be emphasized that amplification of symptoms in chemical sensitivity may originate via sensitization of multiple limbic pathways, including the amygdala, prefrontal cortex and hippocampus, which send excitatory amino acid projections to the nucleus accumbens (Christie et al. 1987; DeFrance et al. 1980; Mogenson et al. 1993). These structures are critical for inducing behavioral sensitization to psychostimulants and stress (Eichler and Antelman 1979; Kalivas and Alesdatter 1993; Wolf et al. 1995; Yoshikawa et al. 1993). Thus, the amplification process may involve multiple limbic system components, impacting many affective and cognitive behaviors found to be altered after chemical exposures in sensitive human populations. Recent data from this laboratory support the idea that alterations may also occur in the amygdala. Rats exposed to Form for 20 days as in the present study demonstrated a greater conditioned fear response (freezing) to a conditioned odor stimulus in a paradigm in which footshock was paired with odor (unpublished results).
The present results are in agreement with a previous study demonstrating enhanced physiological and behavioral sensitivity in mice after repeated exposure to Form (Kane and Alarie 1977; Wood and Coleman 1995). Although Form does not enter the brain, alterations in electroencephalographic activity in cortical and limbic structures have been described after Form and other chemical exposures (Bokina et al. 1976). One possibility for the observation that long-term Form exposure cross-sensitizes to cocaine's effects is that the repeated inescapable Form exposure may provide a stressful stimulus due to its irritant properties on the upper airway. Although the degree of irritation by Form was not quantified in the present study, no signs of this were present, such as face-wiping behavior that was commonly observed during the first few minutes of high levels (11 ppm) of Form exposure (Sorg et al. 1996). Nevertheless, the results may be explained as the occurrence of cross-sensitization between psychostimulants and stress, a widely documented phenomenon (Antelman et al. 1980; Robinson et al. 1985; Sorg and Kalivas 1991). It remains to be tested whether Form-induced effects are acting via those pathways utilized by stress-induced sensitization, or are producing effects either independently or in concert with stress. Whether the effects of Form exposure occur through stress-induced (i.e., via activation of the hypothalamic-pituitary-adrenal axis) versus other chemical-induced pathways may have implications for prevention and/or treatment strategies in chemically sensitive individuals.
When examining the effects of sensitization to single or repeated stimuli, the passage of time is a critical factor. Previous studies examining early and late withdrawal times suggest that different mechanisms are utilized in mediating behavioral sensitization. In some studies, a psychostimulant challenge in behaviorally sensitized rats does not produce an augmentation of extracellular dopamine levels at one to seven days of withdrawal (Segal and Kuczenski 1992a,b). Indeed, it has been suggested that the appearance of sensitized behavior at an early withdrawal time may be mediated by nondopaminergic mechanisms. In contrast, studies demonstrate that at later withdrawal times, the levels of extracellular dopamine in the nucleus accumbens are augmented in response to a psychostimulant challenge (Kalivas and Duffy 1990, 1993; Pettit et al. 1990; Robinson et al. 1988). Conversely, somatodendritic dopamine release and basal firing rate of ventral tegmental area neurons is enhanced early after withdrawal, and both measures are depressed or unaltered after 10 or more days of withdrawal from cocaine (Ackerman and White 1992; see Kalivas et al. 1993 for review). Additional changes occur in dopamine circuitry that do not endure after longer withdrawal from psychostimulants (Kalivas et al. 1993), suggesting that behavioral sensitization at early and late withdrawal times is mediated by different mechanisms.
Time-dependent sensitization has been found after only a single stimulus application (Antelman et al. 1992; Caggiula et al. 1989), and may help to explain our results which demonstrate an increased vertical activity in the air-exposed control group after the passage of time (compare Figures 2A and B, right panels). This increase implicates either daily handling and/or the single cocaine injection given at early withdrawal in producing this enhanced activity. The absence of a sensitized response after the shorter duration of Form exposure (7-day) as compared with the 20-day exposure is likely not due to differences in the passage of time, because even after 4 weeks of withdrawal from daily Form, rats did not demonstrate an augmentation in cocaine-induced activity. Thus, if sensitization was induced by a 7-day exposure to Form, time-dependent increases may be expected to occur (Caggiula et al. 1989). However, the response after 4 weeks of withdrawal was lower than that produced at the early withdrawal time in this group. Although a single stimulus can produce sensitization, in our hands, daily handling and removal of animals from their home cage for 1 h/day for 7 days appears to have produced a transient increase in the sensitivity to cocaine's effects. Such a transient increase may be similar to that reported after short-term cocaine treatment, whose behavioral sensitization effects do not persist beyond 1 week (Post and Weiss 1988).
A few caveats that should be pointed out from the present study concern the treatment schedule for the 7-day and 20-day exposures. First, all 7-day exposures (control and Form groups) were tested followed by all 20-day exposure groups. Thus, it is possible that incidental stressors occurring during the 20-day treatment but not during the 7-day treatment produced the observed sensitization. Because control groups from each experimental paradigm would also be subjected to the same environment, it does not appear to be a likely explanation for the effects; however, future studies would address this issue by performing the various exposures simultaneously. Second, the duration and/or schedule of Form treatment may both be determinants of whether sensitization will occur. Intermittent exposure to Form for 7 consecutive days at a relatively high dose (11 ppm) was previously shown to produce sensitized locomotor responding to a subsequent cocaine injection (Sorg et al. 1996). In the present study, animals exposed to a lower Form dose using the same treatment schedule did not demonstrate sensitization, suggesting that Form effects on cross-sensitivity to cocaine are dependent on dose. When this same lower dose was administered using a different treatment schedule (5 days/week × 4 weeks) with a 2-day interval between each week of exposure, cross-sensitization to cocaine was again observed. Therefore, it may be the duration of Form exposure and/or the particular schedule chosen that produces sensitization. Future studies should be directed toward examining the mechanism for sensitization and whether similar or different mechanisms underlie sensitization produced by high dose versus low dose Form.
There was a significant augmentation of activity in vertical, but not horizontal, activity in response to cocaine. A previous study in this laboratory demonstrated a significant and robust augmentation in horizontal activity after early withdrawal from a 7-day exposure to a high dose (11 ppm) of Form inhalation (Sorg et al. 1996), and although not reported in that study, vertical activity (rearing) was also significantly elevated at later withdrawal times (unpublished observations). Reports of an augmentation in vertical but not horizontal activity in cocaine- or stress-sensitized rats has previously been observed (Kalivas and Duffy 1989; Kalivas et al. 1988), and blockade of vertical but not horizontal activity by the NMDA receptor antagonist, MK-801, has been reported (Kalivas and Alesdatter 1993). Moreover, blockade of opioid receptors by naloxone prevented the vertical but not horizontal response to acute and repeated amphetamine (Balcells-Olivero and Vezina 1996). It remains unclear if specific circuitry within the mesolimbic pathway is altered to produce enhanced vertical as compared with horizontal activity, but the differences may also be a function of the relative degree of sensitization (see Le Moal 1995).
An additional aim of the study was to determine if behavior such as anxiety and pain threshold, parameters that appear to be altered in humans with chemical sensitivity, were modified by repeated Form exposure. Therefore, further behavioral testing on the elevated plus-maze and hotplate was conducted. The elevated plus-maze results indicated no significant changes in anxiety levels between Form and control rats from experiments 1 or 2, although a strong trend for increased anxiety in 7-day Form-pretreated rats was observed. These results and those from the hotplate test are in general agreement with our previous study examining 7-day, high dose Form (11 ppm) pretreated rats, in that no differences were found for these two measures. The absence of clear differences in anxiety or nociception may be due to measurement of animals under basal conditions rather than under a drug- or chemical-induced state. Future studies will need to address whether these measures are simply not altered after repeated chemical exposure or if changes may be found only under the appropriate challenge conditions.
In summary, repeated daily low-level Form exposure produced behavioral cross-sensitization to cocaine-induced activity. The behavioral activity data suggest that enhanced reactivity of specific limbic pathways, including the nucleus accumbens, may be present in daily Form-treated rats. The results indicate that Form exerts at least some of its effects via stimulation of circuitry known to modulate cocaine's effects on activity. No changes after saline challenge were noted, and no significant alterations in anxiety or nociception in the nonchallenged state were apparent in daily Form rats. Therefore, as with behavioral sensitization to psychostimulants and stress, activity changes were small or imperceptible under basal (nonchallenge) conditions. The results lend some support to a limbic system sensitization hypothesis as a potential mechanism contributing to the amplification process of chemical sensitivity in humans. The present study may help establish the development of a mechanistically based animal model, which will provide clues regarding alterations in specific brain pathways of individuals who develop sensitivity to chemicals. The findings offer a basis on which exposure to low-level formaldehyde and potentially other chemicals may be linked to certain psychiatric symptomatology in individuals who appear to be chemically sensitive.
References
Ackerman JM, White FJ . (1992): Decreased activity of rat A10 dopamine neurons following withdrawal from repeated cocaine. Eur J Pharmacol 218: 171–173
Antelman SM . (1988): Stressor-induced sensitization to subsequent stress: Implications for the development and treatment of clinical disorders. In Kalivas PW, Barnes CD (eds), Sensitization in the Nervous System. Caldwell, NJ, Telford Press, pp 227–256
Antelman SM . (1994): Time-dependent sensitization in animals: A possible model of multiple chemical sensitivity in humans. Toxicol Ind Health 10: 335–342
Antelman SM, Eichler AJ, Black CA, Kocan D . (1980): Interchangeability of stress and amphetamine in sensitization. Science 207: 329–331
Antelman SM, Kocan D, Knopf S, Edwards DJ, Caggiula AR . (1992): One brief exposure to a psychological stressor induces long-lasting, time-dependent sensitization of both the cataleptic and neurochemical responses to haloperidol. Life Sci 51: 261–266
Ashford NA, Miller CS . (1991): Chemical Exposures Low Levels and High Stakes. New York, Van Nostrand Reinhold
Balcells-Olivero M, Vezina P . (1996): Naltrexone attenuates acute amphetamine-induced rearing and blocks its sensitization by repeated amphetamine. Soc Neurosci Abst 22: 78
Bell IR . (1994): Neuropsychiatric aspects of sensitivity to low level chemicals: A neural sensitization model. Toxicol Ind Health 10: 277–312
Bell IR, Miller CS, Schwartz GE . (1992): An olfactory-limbic model of multiple chemical sensitivity syndrome: Possible relationships to kindling and affective spectrum disorders. Biol Psychiatry 32: 218–242
Bell IR, Schwartz GE, Bootzin RR, Wyatt JK . (1997): Time-dependent sensitization of heart rate and blood pressure over multiple laboratory sessions in elderly individuals with chemical intolerance. Arch Environ Health 52: 6–17
Black DW, Rathe A, Goldstein RB . (1990): Environmental illness. A controlled study of 26 subjects with “20 century disease.”. JAMA 264: 3166–3170
Bokina AI, Eksler ND, Semenenko AD, Merkur'yeva RV . (1976): Investigation of the mechanism of action of atmospheric pollutants on the central nervous system and comparative evaluation of methods of study. Environ Health Perspect 13: 37–42
Caggiula AR, Antelman SM, Aul E, Knopf S, Edwards DJ . (1989): Prior stress attenuates the analgesic response but sensitizes the corticosterone and cortical dopamine responses to stress 10 days later. Psychopharmacology 99: 233–237
Camp DM, Robinson TE . (1988): Susceptibility to sensitization. II. The influence of gonadal hormones on enduring changes in brain monoamines and behavior produced by the repeated administration of D-amphetamine or restraint stress. Behav Brain Res 30: 69–88
Christie MJ, Summers RJ, Stephenson JA, Cook CJ, Beart PM . (1987): Excitatory amino acid projections to the nucleus accumbens septi in the rat: A retrograde transport study utilizing D[3H]aspartate and [3H]GABA. Neuroscience 22: 425–439
Clary JJ . (1983): Risk assessment for exposure to formaldehyde. In Gibson JE (ed), Formaldehyde Toxicity. Washington, D.C. McGraw-Hill, pp 284–294
Dager SR, Holland JP, Cowley DS, Dunner DL . (1987): Panic disorder precipitated by exposure to organic solvents in the work place. Am J Psychiatry 144: 1056–1058
DeFrance JF, Marchand JE, Stanley JC, Sikes RW, Chronister RB . (1980): Convergence of excitatory amygdaloid and hippocampal input in the nucleus accumbens septi. Brain Res 185: 183–191
Eichler AJ, Antelman SM . (1979): Sensitization to amphetamine and stress may involve nucleus accumbens and medial frontal cortex. Brain Res 176: 412–416
Friedman MJ . (1994): Neurobiological sensitization models of post-traumatic stress disorder: Their possible relevance to multiple chemical sensitivity syndrome. Toxicol Ind Health 10: 449–462
Friedman MJ, Schnurr PP . (1995): The relationship between trauma, post-traumatic stress disorder, and physical health. In Friedman MJ, Charney DS, Deutch AY (eds), Neurobiological and Clinical Consequences of Stress: From Normal Adaptation to PTSD. Philadelphia, Lippincott-Raven Press, pp 507–524
Kalivas PW, Alesdatter JE . (1993): Involvement of N-methyl-D-aspartate receptor stimulation in the ventral tegmental area and amygdala in behavioral sensitization to cocaine. J Pharmacol Exp Ther 267: 486–495
Kalivas PW, Duffy P . (1989): Similar effects of daily cocaine and stress on mesocorticolimbic dopamine neurotransmission in the rat. Biol Psychiatry 25: 913–928
Kalivas PW, Duffy P . (1990): The effect of acute and daily cocaine treatment on extracellular dopamine in the nucleus accumbens. Synapse 5: 48–58
Kalivas PW, Duffy P . (1993): Time course of extracellular dopamine and behavioral sensitization to cocaine. I. Dopamine axon terminals. J Neurosci 13: 266–275
Kalivas PW, Stewart J . (1991): Dopamine transmission in the initiation and expression of drug- and stress-induced sensitization of motor activity. Brain Res Rev 16: 223–244
Kalivas PW, Duffy P, DuMars LA, Skinner C . (1988): Behavioral and neurochemical effects of acute and daily cocaine administration in rats. J Pharmacol Exp Ther 245: 485–492
Kalivas PW, Sorg BA, Hooks MS . (1993): The pharmacology and neural circuitry of sensitization to psychostimulants. Behav Pharmacol 4: 315–334
Kane LE, Alarie Y . (1977): Sensory irritation to formaldehyde and acrolein during single and repeated exposures in mice. Am Ind Hyg Assoc J 38: 509–522
Le Moal M . (1995): Mesocorticolimbic dopaminergic neurons. In Bloom FE, Kupfer DJ (eds), Psychopharmacology: The Fourth Generation of Progress. New York, Raven Press, pp 283–294
Levy CJ . (1988): Agent orange exposure and posttraumatic stress disorder. J Nerv Ment Dis 176: 242–245
Miller CS . (1994): Chemical sensitivity: History and phenomenology. Toxicol Ind Health 10: 253–276
Mogenson GJ, Brudzyunski SM, Wu M, Yang CR, Yim CCY . (1993): From motivation to action: A review of dopaminergic regulation of limbic-nucleus accumbens-pedunculopontine nucleus circuitries involved in limbic-motor integration. In Kalivas PW, Barnes CD (eds), Limbic Motor Circuits and Neuropsychiatry. Boca Raton, FL, CRC Press, pp 193–236
Parsons LH, Justice JB Jr . (1993): Serotonin and dopamine sensitization in the nucleus accumbens, ventral tegmental area and dorsal raphe nucleus following repeated cocaine administration. J Neurochem 61: 1611–1619
Paulson PE, Robinson TE . (1995): Amphetamine-induced time-dependent sensitization of dopamine neurotransmission in the dorsal and ventral striatum: A microdialysis study in behaving rats. Synapse 19: 56–65
Pettit HO, Pan H-T, Parsons LH, Justice JB Jr . (1990): Extracellular concentrations of cocaine and dopamine are enhanced during chronic cocaine administration. J Neurochem 55: 798–804
Post RM, Weiss SRB . (1988): Sensitization and kindling: Implications for the evolution of psychiatric symptomatology. In Kalivas PW, Barnes CD (eds), Sensitization in the Nervous System. Caldwell, NJ, Telford Press, pp 257–292
Robinson TE, Becker JB . (1986): Enduring changes in brain and behavior produced by chronic amphetamine administration: A review and evaluation of animal models of amphetamine psychosis. Brain Res Rev 11: 157–198
Robinson TE, Angus AL, Becker JB . (1985): Sensitization to stress: The enduring effects of prior stress on amphetamine-induced rotational behavior. Life Sci 37: 1039–1042
Robinson TE, Jurson PA, Bennett JA, Bentgen KM . (1988): Persistent sensitization of dopamine nuerotransmission in ventral striatum (nucleus accumbens) produced by prior experience with (+)-amphetamine: A microdialysis study in freely moving rats. Brain Res 462: 211–222
Schottenfeld RS, Cullen MR . (1985): Occupation induced post-traumatic stress disorders. Am J Psychiatry 142: 198–202
Segal DS, Kuczenski R . (1992a): Repeated cocaine administration induces behavioral sensitization and corresponding decreased extracellular dopamine responses in caudate and accumbens. Brain Res 577: 351–355
Segal DS, Kuczenski R . (1992b): In vivo microdialysis reveals a diminished amphetamine-induced dopamine response corresponding to behavioral sensitization produced by repeated amphetamine pretreatment. Brain Res 521: 330–332
Simon GE, Katon WJ, Sparks PJ . (1990): Allergic to life: Psychological factors in environmental illness. Am J Psychiatry 147: 901–906
Sirju A-P, Shepson PB . (1995): Laboratory and field investigation of the DNPH cartridge technique for the measure of atmospheric carbonyl compounds. Environ Sci Technol 29: 385–392
Sorg BA, Kalivas PW . (1991): Effects of cocaine and footshock stress on extracellular dopamine levels in the ventral striatum. Brain Res 559: 29–36
Sorg BA, Hooks MS, Kalivas PW . (1994): Neuroanatomy and neurochemical mechanisms of time-dependent sensitization. Toxicol Ind Health 10: 369–386
Sorg BA, Willis JR, Nowatka TC, Ulibarri C, See RE, Westberg HH . (1996): A proposed animal neurosensitization model for multiple chemical sensitivity in studies with formalin. Toxicology 111: 135–145
Spengler JD . (1991): Sources and concentrations of indoor air pollution. In Samet JM, Spengler JD (eds), Indoor Air Pollution. Baltimore, MD, Johns Hopkins University Press, pp 33–67
Staudenmayer H, Selner JC . (1987): Post-traumatic stress syndrome (PTSS): Escape into the environment. J Clin Psychol 43: 156–157
Staudenmayer H, Selner JC . (1990): Neuropsychology during relaxation in generalized, universal “allergic“ reactivity to the environment: A comparison study. J Psychosom Res 34: 259–270
Terr AI . (1986): Environmental illness: A clinical review of 50 cases. Arch Intern Med 146: 145–149
von Euler G, Ogren S-O, Bondy SC, McKee M, Warner M, Gustafsson JA, Eneroth P, Fuxe K . (1991): Subacute exposure to low concentrations of toluene affects dopamine-mediated locomotor activity in the rat. Toxicology 67: 333–349
von Euler G, Ogren S-O, Li SM, Fuxe K, Gustafsson J-A . (1993): Persistent effects of subchronic toluene exposure on spatial learning and memory, dopamine-mediated locomotor activity and dopamine D2 agonist binding in the rat. Toxicology 77: 223–232
Wolf ME, Dahlin SL, Hu X-T, Xue CJ . (1995): Effects of lesions of prefrontal cortex, amygdala, or fornix on behavioral sensitization to amphetamine: Comparison with N-methyl-D-aspartate antagonists. Neuroscience 69: 417–439
Wood RW, Coleman JB . (1995): Behavioral evaluation of the irritant properties of formaldehyde. Toxicol Appl Pharmacol 130: 67–72
Yoshikawa T, Watanabe A, Shibuya H, Toru M . (1993): Involvement of the fimbria fornix in the initiation but not in the expression of methamphetamine-induced sensitization. Pharmacol Biochem Behav 45: 691–695
Acknowledgements
The authors thank Thomas C. Nowatka for excellent technical assistance. This work was supported by the Wallace Research Foundation (BAS).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sorg, B., Willis, J., See, R. et al. Repeated Low-Level Formaldehyde Exposure Produces Cross-Sensitization to Cocaine: Possible Relevance to Chemical Sensitivity in Humans. Neuropsychopharmacol 18, 385–394 (1998). https://doi.org/10.1016/S0893-133X(97)00179-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S0893-133X(97)00179-6
Keywords
This article is cited by
-
Formaldehyde Impairs Learning and Memory Involving the Disturbance of Hydrogen Sulfide Generation in the Hippocampus of Rats
Journal of Molecular Neuroscience (2013)