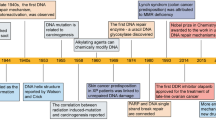
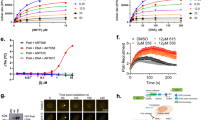
Abstract
Both DNA double- and single-strand break repair are highly coordinated processes utilizing signal transduction cascades and post-translational modifications such as phosphorylation, acetylation and ADP ribosylation. ‘Drugable’ targets within these networks have been identified that could potentially lead to novel therapeutic approaches within the oncology arena. Key regulators within these signalling cascades, such as DNA-dependent protein kinase, ataxia-telangiectasia mutated, checkpoint kinase 1 (CHK1), checkpoint kinase 2 (CHK2) and poly(ADP-ribose) polymerase, use either ATP or nicotinamide adenine dinucleotide for their enzymatic functions and are therefore readily accessible to small molecule inhibition at their catalytic sites. A range of highly potent and selective inhibitors of these DNA damage response pathways has now been identified through drug discovery efforts, with candidate molecules either approaching or already in clinical trials. This review will describe the small molecule inhibitors and drug discovery activities that focus on DNA break repair, along with the therapeutic rationale behind chemosensitization and the concept of synthetic lethality. We will also describe the emerging clinical data coming from this exciting new approach to targeted cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Abraham RT . (2001). Cell cycle checkpoint signaling through the ATM and ATR kinases. Genes Dev 15: 2177–2196.
Abraham RT . (2004). PI 3-kinase related kinases: ‘big’ players in stress-induced signaling pathways. DNA Repair (Amst) 3: 883–887.
Albertella MR, Green CM, Lehmann AR, O'Connor MJ . (2005a). A role for polymerase eta in the cellular tolerance to cisplatin-induced damage. Cancer Res 65: 9799–9806.
Albertella MR, Lau A, O'Connor MJ . (2005b). The overexpression of specialized DNA polymerases in cancer. DNA Repair (Amst) 4: 583–593.
Arienti KL, Brunmark A, Axe FU, McClure K, Lee A, Blevitt J et al. (2005). Checkpoint kinase inhibitors: SAR and radioprotective properties of a series of 2-arylbenzimidazoles. J Med Chem 48: 1873–1885.
Austen B, Powell JE, Alvi A, Edwards I, Hooper L, Starczynski J et al. (2005). Mutations in the ATM gene lead to impaired overall and treatment-free survival that is independent of IGVH mutation status in patients with B-CLL. Blood 106: 3175–3182.
Bartek J, Lukas J . (2003). Chk1 and Chk2 kinases in checkpoint control and cancer. Cancer Cell 3: 421–429.
Bartkova J, Bakkenist CJ, Rajpert-De Meyts E, Skakkebaek NE, Sehested M, Lukas J et al. (2005a). ATM activation in normal human tissues and testicular cancer. Cell Cycle 4: 838–845.
Bartkova J, Horejsí Z, Koed K, Krämer A, Tort F, Zieger K et al. (2005b). DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434: 864–870.
Bonicalzi ME, Haince JF, Droit A, Poirier GG . (2005). Regulation of poly(ADP-ribose) metabolism by poly(ADP-ribose) glycohydrolase: where and when? Cell Mol Life Sci 62: 739–750.
Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E et al. (2005). Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434: 913–917.
Calabrese CR, Almassy R, Barton S, Batey MA, Calvert AH, Canan-Koch S et al. (2004). Anticancer chemosensitization and radiosensitization by the novel poly(ADP-ribose) polymerase-1 inhibitor AG14361. J Natl Cancer Inst 96: 56–67.
Carlessi L, Buscemi G, Larson G, Hong Z, Wu JZ, Delia D . (2007). Biochemical and cellular characterization of VRX0466617, a novel and selective inhibitor for the checkpoint kinase Chk2. Mol Cancer Ther 6: 935–944.
Chalmers A, Johnston P, Woodcock M, Joiner M, Marples B . (2004). PARP-1, PARP-2, and the cellular response to low doses of ionizing radiation. Int J Radiat Oncol Biol Phys 58: 410–419.
Chappell C, Hanakahi LA, Karimi-Busheri F, Weinfeld M, West SC . (2002). Involvement of human polynucleotide kinase in double-strand break repair by non-homologous end joining. EMBO J 21: 2827–2832.
Curtin NJ . (2005). PARP inhibitors for cancer therapy. Expert Rev Mol Med 7: 1–20.
D'Amours D, Desnoyers S, D'Silva I, Poirier GG . (1999). Poly(ADP-ribosyl)ation reactions in the regulation of nuclear functions. Biochem J 342: 249–268.
De Murcia JM, Niedergang C, Trucco C, Ricoul M, Dutrillaux B, Mark M et al. (1997). Requirement of poly(ADP-ribose) polymerase in recovery from DNA damage in mice and in cells. Proc Natl Acad Sci USA 94: 7303–7307.
De Soto JA, Wang X, Tominaga Y, Wang RH, Cao L, Qiao W et al. (2006). The inhibition and treatment of breast cancer with poly (ADP-ribose) polymerase (PARP-1) inhibitors. Int J Biol Sci 2: 179–185.
Durkacz BW, Omidiji O, Gray DA, Shall S . (1980). (ADP-ribose)n participates in DNA excision repair. Nature 283: 593–596.
Evans AR, Limp-Foster M, Kelley MR . (2000). Going APE over ref-1. Mutat Res 461: 83–108.
Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, Richardson TB et al. (2005). Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434: 917–921.
Fedier A, Fink D . (2004). Mutations in DNA mismatch repair genes: implications for DNA damage signaling and drug sensitivity (review). Int J Oncol 24: 1039–1047.
Fong PC, Spicer J, Reade S, Reid A, Vidal L, Schellens JH et al. (2006). Phase I pharmacokinetic (PK) and pharmacodynamic (PD) evaluation of a small molecule inhibitor of poly ADP-ribose polymerase (PARP), KU-0059436 (Ku) in patients (p) with advanced tumours. J Clin Oncol ASCO Annual Meeting Proceedings Part I 24: A3022.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. (2005). Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434: 907–913.
Hardcastle IR, Cockcroft X, Curtin NJ, El-Murr MD, Leahy JJ, Stockley M et al. (2005). Discovery of potent chromen-4-one inhibitors of the DNA-dependent protein kinase (DNA-PK) using a small-molecule library approach. J Med Chem 48: 7829–7846.
Hartman IV JL, Garvik B, Hartwell L . (2001). Principles for the buffering of genetic variation. Science 291: 1001–1004.
Hartwell LH, Weinert TA . (1989). Checkpoints: controls that ensure the order of cell cycle events. Science 246: 629–634.
Hay T, Jenkins H, Sansom OJ, Martin NM, Smith GC, Clarke AR . (2005). Efficient deletion of normal Brca2-deficient intestinal epithelium by poly(ADP-ribose) polymerase inhibition models potential prophylactic therapy. Cancer Res 65: 10145–10148.
Hickson I, Zhao Y, Richardson CJ, Green SJ, Martin NM et al. (2004). Identification and characterization of a novel and specific inhibitor of the ataxia-telangiectasia mutated kinase ATM. Cancer Res 64: 9152–9159.
Hoeijmakers JH . (2001). Genome maintenance mechanisms for preventing cancer. Nature 411: 366–374.
Hollick JJ, Rigoreau LJ, Cano-Soumillac C, Cockcroft X, Curtin NJ, Frigerio M et al. (2007). Pyranone, thiopyranone, and pyridone inhibitors of phosphatidylinositol 3-kinase related kinases. Structure–activity relationships for DNA-dependent protein kinase inhibition, and identification of the first potent and selective inhibitor of the ataxia telangiectasia mutated kinase. J Med Chem 50: 1958–1972.
Hu HY, Horton JK, Gryk MR, Prasad R, Naron JM, Sun DA et al. (2004). Identification of small molecule synthetic inhibitors of DNA polymerase beta by NMR chemical shift mapping. J Biol Chem 279: 39736–39744.
Ismail IH, Martensson S, Moshinsky D, Rice A, Tang C, Howlett A et al. (2004). SU11752 inhibits the DNA-dependent protein kinase and DNA double-strand break repair resulting in ionizing radiation sensitization. Oncogene 23: 873–882.
Izzard RA, Jackson SP, Smith GC . (1999). Competitive and noncompetitive inhibition of the DNA-dependent protein kinase. Cancer Res 59: 2581–2586.
Jackson SP . (2001). Detecting, signalling and repairing DNA double-strand breaks. Biochem Soc Trans 29: 655–661.
Janetka JW, Ashwell S, Zabludoff S, Lyne P . (2007). Inhibitors of checkpoint kinases: from discovery to the clinic. Curr Opin Drug Discov Devel 10: 473–486.
Jazayeri A, Falck J, Lukas C, Bartek J, Smith GC, Lukas J et al. (2006). ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nat Cell Biol 8: 37–45.
Jeggo PA . (1998). Identification of genes involved in repair of DNA double-strand breaks in mammalian cells. Radiat Res 150: S80–S91.
Kaelin Jr WG . (2005). The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer 5: 689–698.
Karimi-Busheri F, Rasouli-Nia A, Allalunis-Turner J, Weinfeld M . (2007). Human polynucleotide kinase participates in repair of DNA double-strand breaks by nonhomologous end joining but not homologous recombination. Cancer Res 67: 6619–6625.
Kawabe T . (2004). G2 checkpoint abrogators as anticancer drugs. Mol Cancer Ther 3: 513–519.
Kennedy RD, Chen CC, Stuckert P, Archila EM, De la Vega MA, Moreau LA et al. (2007). Fanconi anemia pathway-deficient tumor cells are hypersensitive to inhibition of ataxia telangiectasia mutated. J Clin Invest 117: 1440–1449.
Kennedy RD, D'Andrea AD . (2006). DNA repair pathways in clinical practice: lessons from pediatric cancer susceptibility syndromes. J Clin Oncol 24: 3799–3808.
Koberle B, Masters JR, Hartley JA, Wood RD . (1999). Defective repair of cisplatin-induced DNA damage caused by reduced XPA protein in testicular germ cell tumours. Curr Biol 9: 273–276.
Krokan EH, Slupphaug G (eds). (2007). Repair of small base lesions in DNA – from molecular biology to phenotype. DNA Repair 6: 397–560.
Kurz EU, Lees-Miller SP . (2004). DNA damage-induced activation of ATM and ATM-dependent signaling pathways. DNA Repair (Amst) 3: 889–900.
Lau A, Swinbank KM, Ahmed PS, Taylor DL, Jackson SP, Smith GC et al. (2005). Suppression of HIV-1 infection by a small molecule inhibitor of the ATM kinase. Nat Cell Biol 7: 493–500.
Lavin MF, Kozlov S . (2007). ATM activation and DNA damage response. Cell Cycle 6: 931–942.
Lindahl T . (1993). Instability and decay of the primary structure of DNA. Nature 362: 709–715.
Liu L, Gerson ST . (2004). Therapeutic impact of methoxyamine: blocking repair of abasic sites in the base excision repair pathway. Curr Opin Investig Drugs 5: 623–627.
Lucchesi JC . (1968). Synthetic lethality and semi-lethality among functionally related mutants of Drosophila melanogaster. Genetics 59: 37–44.
Lynch HT, de la Chapelle A . (1999). Genetic susceptibility to non-polyposis colorectal cancer. J Med Genet 36: 801–818.
Madhusudan S, Smart F, Shrimpton P, Parsons JL, Gardiner L, Houlbrook S et al. (2005). Isolation of a small molecule inhibitor of DNA base excision repair. Nucleic Acids Res 33: 4711–4724.
Malkin D, Li FP, Strong LC, Fraumeni Jr JF, Nelson CE, Kim DH et al. (1990). Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science 250: 1233–1238.
Masutani M, Nozaki T, Nakamoto K, Nakagama H, Suzuki H, Kusuoka O et al. (2000). The response of Parp knockout mice against DNA damaging agents. Mutat Res 462: 159–166.
Matsuoka S, Ballif BA, Smogorzewska A, McDonald III ER, Hurov KE, Luo J et al. (2007). ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 316: 1160–1166.
McCabe N, Lord CJ, Tutt AN, Martin NM, Smith GC, Ashworth A . (2005). BRCA2-deficient CAPAN-1 cells are extremely sensitive to the inhibition of poly (ADP-ribose) polymerase: an issue of potency. Cancer Biol Ther 4: 934–936.
McCabe N, Turner NC, Lord CJ, Kluzek K, Bialkowska A, Swift S et al. (2006). Deficiency in the repair of DNA damage by homologous recombination and sensitivity to poly(ADP-ribose) polymerase inhibition. Cancer Res 66: 8109–8115.
Miknyoczki SJ, Jones-Bolin S, Pritchard S, Hunter K, Zhao H, Wan W et al. (2003). Chemopotentiation of temozolomide, irinotecan, and cisplatin activity by CEP-6800, a poly(ADP-ribose) polymerase inhibitor. Mol Cancer Ther 2: 371–382.
Muller C, Christodoulopoulos G, Salles B, Panasci L . (1998). DNA-dependent protein kinase activity correlates with clinical and in vitro sensitivity of chronic lymphocytic leukemia lymphocytes to nitrogen mustards. Blood 92: 2213–2219.
Nghiem P, Park PK, Kim Y, Vaziri C, Schreiber SL . (2001). ATR inhibition selectively sensitizes G1 checkpoint-deficient cells to lethal premature chromatin condensation. Proc Natl Acad Sci USA 98: 9092–9097.
Plummer R . (2005). First in human phase I trial of the PARP inhibitor AG-014699 with temozolomide (TMZ) in patients (pts) with advanced solid tumors. J Clin Oncol ASCO Annual Meeting Proceedings 23: A3065.
Plummer R . (2006). First and final report of a phase II study of the poly(ADP-ribose) polymerase (PARP) inhibitor, AG014699, in combination with temozolomide (TMZ) in patients with metastatic malignant melanoma (MM). J Clin Oncol ASCO Annual Meeting Proceedings Part I 24: A8013.
Powell SN, DeFrank JS, Connell P, Eogan M, Preffer F, Dombkowski D et al. (1995). Differential sensitivity of p53(−) and p53(+) cells to caffeine-induced radiosensitization and override of G2 delay. Cancer Res 55: 1643–1648.
Ratnam K, Low JA . (2007). Current development of clinical inhibitors of poly(ADP-ribose) polymerase in oncology. Clin Cancer Res 13: 1383–1388.
Riballo E, Kuhne M, Rief N, Doherty A, Smith GC, Recio MJ et al. (2004). A pathway of double-strand break rejoining dependent upon ATM, Artemis, and proteins locating to gamma-H2AX foci. Mol Cell 16: 715–724.
Sarkaria JN, Busby EC, Tibbetts RS, Roos P, Taya Y, Karnitz LM et al. (1999). Inhibition of ATM and ATR kinase activities by the radiosensitizing agent, caffeine. Cancer Res 59: 4375–4382.
Sarkaria JN, Tibbetts RS, Busby EC, Kennedy AP, Hill DE, Abraham RT . (1998). Inhibition of phosphoinositide 3-kinase related kinases by the radiosensitizing agent wortmannin. Cancer Res 58: 4375–4382.
Shimizu N, Sugimoto K, Tang J, Nishi T, Sato I, Hiramoto M et al. (2000). High-performance affinity beads for identifying drug receptors. Nat Biotechnol 18: 877–881.
Shinohara ET, Geng L, Tan J, Chen H, Shir Y et al. (2005). DNA-dependent protein kinase is a molecular target for the development of noncytotoxic radiation-sensitizing drugs. Cancer Res 65: 4987–4992.
Shiobara M, Miyazaki M, Ito H, Togawa A, Nakajima N, Nomura F et al. (2001). Enhanced polyadenosine diphosphate-ribosylation in cirrhotic liver and carcinoma tissues in patients with hepatocellular carcinoma. J Gastroenterol Hepatol 16: 338–344.
Smith GC, Jackson SP . (1999). The DNA-dependent protein kinase. Genes Dev 13: 916–934.
Smith GCM, Jackson SP . (2003) In: Bradshaw RA, Dennis EA (eds). Handbook of Cell Signalling vol. 1 Elsevier Science: USA, pp 557–561.
Southan GJ, Szabo C . (2003). Poly(ADP-ribose) polymerase inhibitors. Curr Med Chem 10: 321–340.
Srivastava S, Zou ZQ, Pirollo K, Blattner W, Chang EH . (1990). Germ-line transmission of a mutated p53 gene in a cancer-prone family with Li–Fraumeni syndrome. Nature 348: 747–749.
Stiff T, Walker SA, Cerosaletti K, Goodarzi AA, Petermann E, Concannon P et al. (2006). ATR-dependent phosphorylation and activation of ATM in response to UV treatment or replication fork stalling. EMBO J 25: 5775–5782.
Tomoda T, Kurashige T, Moriki T, Yamamoto H, Fujimoto S, Taniguchi T . (1991). Enhanced expression of poly(ADP-ribose) synthetase gene in malignant lymphoma. Am J Hematol 37: 223–227.
Virag L, Szabo C . (2002). The therapeutic potential of poly(ADP-ribose) polymerase inhibitors. Pharmacol Rev 54: 375–429.
Won J, Kim M, Kim N, Ahn JH, Lee WG, Kim SS et al. (2006). Small molecule-based reversible reprogramming of cellular lifespan. Nat Chem Biol 2: 369–374.
Yang S, Irani K, Heffron SE, Jurnak F, Meyskens Jr FL . (2005). Alterations in the expression of the apurinic/apyrimidinic endonuclease-1/redox factor-1 (APE/Ref-1) in human melanoma and identification of the therapeutic potential of resveratrol as an APE/Ref-1 inhibitor. Mol Cancer Ther 4: 1923–1935.
Yap TA . (2007). First in human phase I pharmacokinetic (PK) and pharmacodynamic (PD) study of KU-0059436 (Ku), a small molecule inhibitor of poly ADP-ribose polymerase (PARP) in cancer patients (p), including BRCA1/2 mutation carriers. J Clin Oncol ASCO Annual Meeting Proceedings Part I 25: A3529.
Zhao Y, Thomas HD, Batey MA, Cowell IG, Richardson CJ, Griffin RJ et al. (2006). Preclinical evaluation of a potent novel DNA-dependent protein kinase inhibitor NU7441. Cancer Res 66: 5354–5362.
Zou L, Elledge SJ . (2003). Sensing DNA damage through ATRIP recognition of RPA–ssDNA complexes. Science 300: 1542–1548.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
This article is cited by
-
N-terminus-modified Hec1 suppresses tumour growth by interfering with kinetochore–microtubule dynamics
Oncogene (2015)
-
Methoxyamine sensitizes the resistant glioblastoma T98G cell line to the alkylating agent temozolomide
Clinical and Experimental Medicine (2013)
-
A phenanthrene derived PARP inhibitor is an extra-centrosomes de-clustering agent exclusively eradicating human cancer cells
BMC Cancer (2011)
-
Strategies to improve radiotherapy with targeted drugs
Nature Reviews Cancer (2011)
-
Harnessing the complexity of DNA-damage response pathways to improve cancer treatment outcomes
Oncogene (2010)