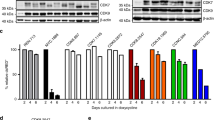
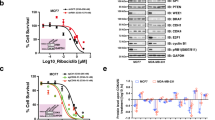
Abstract
Mutant Abl kinases (such as BCR-Abl) drive the development of leukemia; however little is known regarding whether Abl kinases contribute to the development or progression of solid tumors. We recently demonstrated that endogenous Abl kinases (c-Abl, Arg) are activated by deregulated ErbB receptors and Src kinases, and drive invasion of aggressive breast cancer cells. In this study, we examined whether activation of endogenous Abl kinases affects transformation, proliferation and survival, which are major contributors to breast cancer development and metastatic progression. Using a pharmacological inhibitor and RNAi, we demonstrate that activation of endogenous Abl kinases dramatically promotes breast cancer cell proliferation and anchorage-independent growth in serum, as well as survival following nutrient deprivation. Activation of Abl kinases mediates phosphorylation of STAT3, and promotes proliferation by accelerating G1 → S progression. Moreover, we identify IGF-1R as a novel upstream activator of endogenous Abl kinases, and demonstrate that Abl kinase activation is required for IGF-1-stimulated cell cycle progression in breast cancer cells. Since activation of Abl kinases affects multiple steps of breast cancer development and progression, Abl kinase inhibitors are likely to be effective agents for the treatment of breast cancers containing highly active Abl kinases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Buettner R, Mora LB, Jove R . (2002). Activated STAT signaling in human tumors provides novel molecular targets for therapeutic intervention. Clin Cancer Res 8: 945–954.
Burkle A . (2005). Poly(ADP-ribose). The most elaborate metabolite of NAD+. FEBS J 272: 4576–4589.
Camirand A, Zakikhani M, Young F, Pollak M . (2005). Inhibition of insulin-like growth factor-1 receptor signaling enhances growth-inhibitory and proapoptotic effects of gefitinib (Iressa) in human breast cancer cells. Breast Cancer Res 7: R570–R579.
Chernicky CL, Tan H, Yi L, Loret de Mola JR, Ilan J . (2002). Treatment of murine breast cancer cells with antisense RNA to the type I insulin-like growth factor receptor decreases the level of plasminogen activator transcripts, inhibits cell growth in vitro, and reduces tumorigenesis in vivo. Mol Pathol 55: 102–109.
Chernicky CL, Yi L, Tan H, Gan SU, Ilan J . (2000). Treatment of human breast cancer cells with antisense RNA to the type I insulin-like growth factor receptor inhibits cell growth, suppresses tumorigenesis, alters the metastatic potential, and prolongs survival in vivo. Cancer Gene Ther 7: 384–395.
Danial NN, Losman JA, Lu T, Yip N, Krishnan K, Krolewski J et al. (1998). Direct interaction of Jak1 and v-Abl is required for v-Abl-induced activation of STATs and proliferation. Mol Cell Biol 18: 6795–6804.
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM et al. (2001). Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344: 1031–1037.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S et al. (1996). Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 2: 561–566.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hines SJ, Organ C, Kornstein MJ, Krystal GW . (1995). Coexpression of the c-kit and stem cell factor genes in breast carcinomas. Cell Growth Differ 6: 769–779.
Holbro T, Hynes NE . (2004). ErbB receptors: directing key signaling networks throughout life. Annu Rev Pharmacol Toxicol 44: 195–217.
Hynes NE . (2000). Tyrosine kinase signalling in breast cancer. Breast Cancer Res 2: 154–157.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ . (2007). Cancer statistics, 2007. CA Cancer J Clin 57: 43–66.
Kauffman EC, Robinson VL, Stadler WM, Sokoloff MH, Rinker-Schaeffer CW . (2003). Metastasis suppression: the evolving role of metastasis suppressor genes for regulating cancer cell growth at the secondary site. J Urol 169: 1122–1133.
Kharas MG, Fruman DA . (2005). ABL oncogenes and phosphoinositide 3-kinase: mechanism of activation and downstream effectors. Cancer Res 65: 2047–2053.
Levy DE, Gilliland DG . (2000). Divergent roles of STAT1 and STAT5 in malignancy as revealed by gene disruptions in mice. Oncogene 19: 2505–2510.
Liliensiek SJ, Schell K, Howard E, Nealey P, Murphy CJ . (2006). Cell sorting but not serum starvation is effective for SV40 human corneal epithelial cell cycle synchronization. Exp Eye Res 83: 61–68.
Marra F, Choudhury GG, Abboud HE . (1996). Interferon-gamma-mediated activation of STAT1alpha regulates growth factor-induced mitogenesis. J Clin Invest 98: 1218–1230.
Morgensztern D, McLeod HL . (2005). PI3K/Akt/mTOR pathway as a target for cancer therapy. Anticancer Drugs 16: 797–803.
Nagano K, Itagaki C, Izumi T, Nunomura K, Soda Y, Tani K et al. (2006). Rb plays a role in survival of Abl-dependent human tumor cells as a downstream effector of Abl tyrosine kinase. Oncogene 25: 493–502.
Nahta R, Yuan LX, Zhang B, Kobayashi R, Esteva FJ . (2005). Insulin-like growth factor-I receptor/human epidermal growth factor receptor 2 heterodimerization contributes to trastuzumab resistance of breast cancer cells. Cancer Res 65: 11118–11128.
Noren NK, Foos G, Hauser CA, Pasquale EB . (2006). The EphB4 receptor suppresses breast cancer cell tumorigenicity through an Abl-Crk pathway. Nat Cell Biol 8: 815–825.
Pendergast AM, Carella AM, Daley GQ, Eaves CJ, Goldman JM, Helmann R . (2001) (eds). Chronic Myeloid Leukaemia: Biology and Treatment. Martin Dunitz, Lt: London, pp 19–39.
Pendergast AM . (2002). The Abl family kinases: mechanisms of regulation and signaling. Adv Cancer Res 85: 51–100.
Plattner R, Pendergast AM . (2003). Activation and signaling of the Abl tyrosine kinase: bidirectional link with phosphoinositide signaling. Cell Cycle 2: 273–274.
Santen RJ, Song RX, McPherson R, Kumar R, Adam L, Jeng MH et al. (2002). The role of mitogen-activated protein (MAP) kinase in breast cancer. J Steroid Biochem Mol Biol 80: 239–256.
Singer CF, Hudelist G, Lamm W, Mueller R, Czerwenka K, Kubista E . (2004). Expression of tyrosine kinases in human malignancies as potential targets for kinase-specific inhibitors. Endocr Relat Cancer 11: 861–869.
Srinivasan D, Plattner R . (2006). Activation of Abl tyrosine kinases promotes invasion of aggressive breast cancer cells. Cancer Res 66: 5648–5655.
van Diest PJ, van der Wall E, Baak JP . (2004). Prognostic value of proliferation in invasive breast cancer: a review. J Clin Pathol 57: 675–681.
Vantaggiato C, Formentini I, Bondanza A, Bonini C, Naldini L, Brambilla R . (2006). ERK1 and ERK2 mitogen-activated protein kinases affect Ras-dependent cell signaling differentially. J Biol 5: 14.
Vigneri P, Wang JY . (2001). Induction of apoptosis in chronic myelogenous leukemia cells through nuclear entrapment of BCR-ABL tyrosine kinase. Nat Med 7: 228–234.
Widschwendter A, Tonko-Geymayer S, Welte T, Daxenbichler G, Marth C, Doppler W . (2002). Prognostic significance of signal transducer and activator of transcription 1 activation in breast cancer. Clin Cancer Res 8: 3065–3074.
Wu J, Shen ZZ, Lu JS, Jiang M, Han QX, Fontana JA et al. (1999). Prognostic role of p27Kip1 and apoptosis in human breast cancer. Br J Cancer 79: 1572–1578.
Acknowledgements
We thank Jennifer Strange and Greg Bauman for assistance with FACS, Dr Lawrence Brewer for assistance with statistics and Drs Natasha Kyprianou, Vivek Rangnekar and Sayan Mitra for critically reading the manuscript. This work was supported by a Concern Foundation Young Investigator Award, American Cancer Society Institutional Pilot Grant 85-001-16 IRG, and NIH P20 RR20171 from the National Center for Research Resources to RP. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Srinivasan, D., Sims, J. & Plattner, R. Aggressive breast cancer cells are dependent on activated Abl kinases for proliferation, anchorage-independent growth and survival. Oncogene 27, 1095–1105 (2008). https://doi.org/10.1038/sj.onc.1210714
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210714
Keywords
This article is cited by
-
VNTR polymorphism in the breakpoint region of ABL1 and susceptibility to bladder cancer
BMC Medical Genomics (2021)
-
Enabling Tumor Growth and Progression: Recent Progress in Unraveling the Functions of ABL Kinases in Solid Tumor Cells
Current Pharmacology Reports (2018)
-
Gene expression profiles and signaling mechanisms in α2B-adrenoceptor-evoked proliferation of vascular smooth muscle cells
BMC Systems Biology (2017)
-
Mutant p53 dictates the oncogenic activity of c-Abl in triple-negative breast cancers
Cell Death & Disease (2017)
-
c-Abl and Arg induce cathepsin-mediated lysosomal degradation of the NM23-H1 metastasis suppressor in invasive cancer
Oncogene (2014)