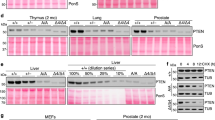
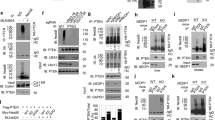
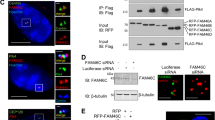
Abstract
Phosphatase and tensin homolog deleted on chromosome 10 (PTEN) is an important negative regulator of cell growth and a tumor suppressor. Its growth-attenuating activity is based on the dephosphorylation of phosphatidylinositol 3,4,5-trisphosphate (PIP3), an essential second messenger for the phosphoinositide 3-kinase/Akt signaling pathway. This activity may require localization of PTEN to cytoplasmic membranes. Yet PTEN can also localize to the cell nucleus where its functions remain unclear. Here we present data that define a short sequence in the N-terminal region of PTEN required for cytoplasmic localization. We will refer to this sequence as cytoplasmic localization signal (CLS). It could function as a non-canonical signal for nuclear export or as a cytoplasmic retention signal of PTEN. Mutations within the CLS induce nuclear localization and impair growth suppressive activities of PTEN while preserving lipid phosphatase activity. We propose that nuclear localization of PTEN is not compatible with plasma membrane-targeted growth suppressive functions of PTEN.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bonneau D, Longy M . (2000). Mutations of the human PTEN gene. Hum Mutat 16: 109–122.
Chung JH, Eng C . (2005). Nuclear-cytoplasmic partitioning of phosphatase and tensin homologue deleted on chromosome 10 (PTEN) differentially regulates the cell cycle and apoptosis. Cancer Res 65: 8096–8100.
Chung JH, Ginn-Pease ME, Eng C . (2005). Phosphatase and tensin homologue deleted on chromosome 10 (PTEN) has nuclear localization signal-like sequences for nuclear import mediated by major vault protein. Cancer Res 65: 4108–4116.
Das S, Dixon JE, Cho W . (2003). Membrane-binding and activation mechanism of PTEN. Proc Natl Acad Sci USA 100: 7491–7496.
Duerr EM, Rollbrocker B, Hayashi Y, Peters N, Meyer-Puttlitz B, Louis DN et al. (1998). PTEN mutations in gliomas and glioneuronal tumors. Oncogene 16: 2259–2264.
Fabbro M, Henderson BR . (2003). Regulation of tumor suppressors by nuclear-cytoplasmic shuttling. Exp Cell Res 282: 59–69.
Furnari FB, Lin H, Huang HS, Cavenee WK . (1997). Growth suppression of glioma cells by PTEN requires a functional phosphatase catalytic domain. Proc Natl Acad Sci USA 94: 12479–12484.
Gil A, Andres-Pons A, Fernandez E, Valiente M, Torres J, Cervera J et al. (2006). Nuclear localization of PTEN by Ran-dependent mechanism enhances apoptosis: involvement of an N-terminal nuclear localization domain and multiple nuclear exclusion motifs. Mol Biol Cell 17: 4002–4013.
Leslie NR, Downes CP . (2004). PTEN function: how normal cells control it and tumour cells lose it. Biochem J 382: 1–11.
Leslie NR, Yang X, Downes CP, Weijer CJ . (2005). The regulation of cell migration by PTEN. Biochem Soc Trans 33: 1507–1508.
Li DM, Sun H . (1997). TEP1, encoded by a candidate tumor suppressor locus, is a novel protein tyrosine phosphatase regulated by transforming growth factor beta. Cancer Res 57: 2124–2129.
Li DM, Sun H . (1998). PTEN/MMAC1/TEP1 suppresses the tumorigenicity and induces G1 cell cycle arrest in human glioblastoma cells. Proc Natl Acad Sci USA 95: 15406–15411.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Lian Z, Di Cristofano A . (2005). Class reunion: PTEN joins the nuclear crew. Oncogene 24: 7394–7400.
Liu JL, Sheng X, Hortobagyi ZK, Mao Z, Gallick GE, Yung WK . (2005). Nuclear PTEN-mediated growth suppression is independent of Akt down-regulation. Mol Cell Biol 25: 6211–6224.
Maehama T, Dixon JE . (1998). The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 273: 13375–13378.
Maehama T, Dixon JE . (1999). PTEN: a tumour suppressor that functions as a phospholipid phosphatase. Trends Cell Biol 9: 125–128.
Mayo LD, Dixon JE, Durden DL, Tonks NK, Donner DB . (2002). PTEN protects p53 from Mdm2 and sensitizes cancer cells to chemotherapy. J Biol Chem 277: 5484–5489.
Myers MP, Pass I, Batty IH, Van der Kaay J, Stolarov JP, Hemmings BA et al. (1998). The lipid phosphatase activity of PTEN is critical for its tumor supressor function. Proc Natl Acad Sci USA 95: 13513–13518.
Myers MP, Stolarov JP, Eng C, Li J, Wang SI, Wigler MH et al. (1997). P-TEN, the tumor suppressor from human chromosome 10q23, is a dual-specificity phosphatase. Proc Natl Acad Sci USA 94: 9052–9057.
Ramaswamy S, Nakamura N, Vazquez F, Batt DB, Perera S, Roberts TM et al. (1999). Regulation of G1 progression by the PTEN tumor suppressor protein is linked to inhibition of the phosphatidylinositol 3-kinase/Akt pathway. Proc Natl Acad Sci USA 96: 2110–2115.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH et al. (1997). Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 15: 356–362.
Su JD, Mayo LD, Donner DB, Durden DL . (2003). PTEN and phosphatidylinositol 3′-kinase inhibitors up-regulate p53 and block tumor-induced angiogenesis: evidence for an effect on the tumor and endothelial compartment. Cancer Res 63: 3585–3592.
Sun H, Lesche R, Li DM, Liliental J, Zhang H, Gao J et al. (1999). PTEN modulates cell cycle progression and cell survival by regulating phosphatidylinositol 3,4,5,-trisphosphate and Akt/protein kinase B signaling pathway. Proc Natl Acad Sci USA 96: 6199–6204.
Trotman LC, Pandolfi PP . (2003). PTEN and p53: who will get the upper hand? Cancer Cell 3: 97–99.
Van Meir EG, Kikuchi T, Tada M, Li H, Diserens AC, Wojcik BE et al. (1994). Analysis of the p53 gene and its expression in human glioblastoma cells. Cancer Res 54: 649–652.
Vazquez F, Devreotes P . (2006). Regulation of PTEN function as a PIP3 gatekeeper through membrane interaction. Cell Cycle 5: 1523–1527.
Vazquez F, Matsuoka S, Sellers WR, Yanagida T, Ueda M, Devreotes PN . (2006). Tumor suppressor PTEN acts through dynamic interaction with the plasma membrane. Proc Natl Acad Sci USA 103: 3633–3638.
Vogt PK . (2001). PI 3-kinase, mTOR, protein synthesis and cancer. Trends Mol Med 7: 482–484.
Walker SM, Leslie NR, Perera NM, Batty IH, Downes CP . (2004). The tumour-suppressor function of PTEN requires an N-terminal lipid-binding motif. Biochem J 379: 301–307.
Wen S, Stolarov J, Myers MP, Su JD, Wigler MH, Tonks NK et al. (2001). PTEN controls tumor-induced angiogenesis. Proc Natl Acad Sci USA 98: 4622–4627.
Acknowledgements
The work of the authors is supported by grants from the National Cancer Institute (CA078230, CA078045 and CA94233) and a Fellowship of the American Cancer Society (PF-04-092-01-MGO). This is manuscript number 17452-MEM of The Scripps Research Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Denning, G., Jean-Joseph, B., Prince, C. et al. A short N-terminal sequence of PTEN controls cytoplasmic localization and is required for suppression of cell growth. Oncogene 26, 3930–3940 (2007). https://doi.org/10.1038/sj.onc.1210175
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210175
Keywords
This article is cited by
-
Molecular Characteristics, Phylogeny and Expression Profile of the PTEN Gene in Goats
Biochemical Genetics (2020)
-
Identification of diverse activating mutations of the RAS-MAPK pathway in histiocytic sarcoma
Modern Pathology (2019)
-
Precise definition of PTEN C-terminal epitopes and its implications in clinical oncology
npj Precision Oncology (2019)
-
PTEN/PTENP1: ‘Regulating the regulator of RTK-dependent PI3K/Akt signalling’, new targets for cancer therapy
Molecular Cancer (2018)
-
Notch-1-PTEN-ERK1/2 signaling axis promotes HER2+ breast cancer cell proliferation and stem cell survival
Oncogene (2018)