Abstract
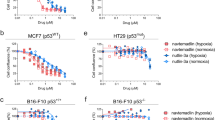
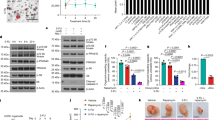
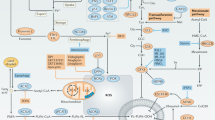
We have previously reported tumour-selective killing by the sigma (σ) receptor ligand rimcazole. We now report that rimcazole elevates hypoxia inducible factor-1α (HIF-1α) protein levels under normoxic conditions in colorectal (HCT-116) and mammary carcinoma (MDA MB 231) cells but fails to induce HIF-1α in normal fibroblasts or mammary epithelial cells. Combining the σ-1 agonist (+)-pentazocine with rimcazole substantially reduces the accumulation of HIF-1α, confirming that the effect is mediated at least partly by antagonism of σ-1 sites. HIF-1α knockdown by RNA interference attenuates rimcazole-induced cell death in both cell types. Thus, the induction of HIF-1α by rimcazole contributes to tumour cell killing. In a comparison of HCT-116p53+/+ and HCT-116p53−/− cells, HIF-1α levels are consistently higher after rimcazole treatment in HCT-116p53+/+ cells. Furthermore, although rimcazole kills HCT-116p53−/− cells, it has a more potent apoptosis-inducing effect in HCT-116p53+/+ cells. This suggests that the presence of functional p53 protein may enhance death induction by rimcazole in part through greater induction of HIF-1α. p53 is not required, however, for the rimcazole-induced engagement of HIF-1α in proapoptotic mode as HIF-1α knockdown attenuates rimcazole-induced death to comparable extents in p53 mutant and wild-type cell systems. Knowledge of HIF-1α involvement may assist the re-profiling of rimcazole and other σ ligands as cancer therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Achison M, Hupp TR . (2003). Hypoxia attenuates the p53 response to cellular damage. Oncogene 22: 3431–3440.
An WG, Kanekal M, Simon MC, Maltepe E, Blagosklonny MV, Neckers LM . (1998). Stabilization of wild-type p53 by hypoxia-inducible factor 1alpha. Nature 392: 405–408.
Arends JW . (2000). Molecular interactions in the Vogelstein model of colorectal carcinoma. J Pathol 190: 412–416.
Bacon AL, Harris AL . (2004). Hypoxia-inducible factors and hypoxic cell death in tumour physiology. Ann Med 36: 530–539.
Bunz F, Hwang PM, Torrance C, Waldman T, Zhang Y, Dillehay L et al. (1999). Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J Clin Invest 104: 263–269.
Chen D, Li M, Luo J, Gu W . (2003). Direct interactions between HIF-1 alpha and Mdm2 modulate p53 function. J Biol Chem 278: 13595–13598.
Chun AC, Jin DY . (2003). Transcriptional regulation of mitotic checkpoint gene MAD1 by p53. J Biol Chem 278: 37439–37450.
Fels DR, Koumenis C . (2005). HIF-1alpha and p53: the ODD couple? Trends Biochem Sci 30: 426–429.
Fukuda R, Hirota K, Fan F, Jung YD, Ellis LM, Semenza GL . (2002). Insulin-like growth factor 1 induces hypoxia-inducible factor 1-mediated vascular endothelial growth factor expression, which is dependent on MAP kinase and phosphatidylinositol 3-kinase signaling in colon cancer cells. J Biol Chem 277: 38205–38211.
Giaccia AJ, Brown JM, Wouters B, Denko N, Koumenis C . (1998). Cancer therapy and tumor physiology. Science 279: 12–13.
Giaccia AJ, Kastan MB . (1998). The complexity of p53 modulation: emerging patterns from divergent signals. Genes Dev 12: 2973–2983.
Gilmore DL, Liu Y, Matsumoto RR . (2004). Review of the pharmacological and clinical profile of rimcazole. CNS Drug Rev 10: 1–22.
Graeber TG, Osmanian C, Jacks T, Housman DE, Koch CJ, Lowe SW et al. (1996). Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours. Nature 379: 88–91.
Greijer AE, van der Wall E . (2004). The role of hypoxia inducible factor 1 (HIF-1) in hypoxia induced apoptosis. J Clin Pathol 57: 1009–1014.
Hammond EM, Dorie MJ, Giaccia AJ . (2003). ATR/ATM targets are phosphorylated by ATR in response to hypoxia and ATM in response to reoxygenation. J Biol Chem 278: 12207–12213.
Hansson LO, Friedler A, Freund S, Rudiger S, Fersht AR . (2002). Two sequence motifs from HIF-1alpha bind to the DNA-binding site of p53. Proc Natl Acad Sci USA 99: 10305–10309.
Kim JY, Ahn HJ, Ryu JH, Suk K, Park JH . (2004). BH3-only protein Noxa is a mediator of hypoxic cell death induced by hypoxia-inducible factor 1alpha. J Exp Med 199: 113–124.
Koumenis C, Alarcon R, Hammond E, Sutphin P, Hoffman W, Murphy M et al. (2001). Regulation of p53 by hypoxia: dissociation of transcriptional repression and apoptosis from p53-dependent transactivation. Mol Cell Biol 21: 1297–1310.
Krishnamachary B, Berg-Dixon S, Kelly B, Agani F, Feldser D, Ferreira G et al. (2003). Regulation of colon carcinoma cell invasion by hypoxia-inducible factor 1. Cancer Res 63: 1138–1143.
Langa F, Codony X, Tovar V, Lavado A, Gimenez E, Cozar P et al. (2003). Generation and phenotypic analysis of sigma receptor type I (sigma 1) knockout mice. Eur J Neurosci 18: 2188–2196.
Laughner E, Taghavi P, Chiles K, Mahon PC, Semenza GL . (2001). HER2 (neu) signaling increases the rate of hypoxia-inducible factor 1alpha (HIF-1alpha) synthesis: novel mechanism for HIF-1-mediated vascular endothelial growth factor expression. Mol Cell Biol 21: 3995–4004.
Magnusson KP, Satalino R, Qian W, Klein G, Wiman KG . (1998). Is conversion of solid into more anoxic ascites tumors associated with p53 inactivation? Oncogene 17: 2333–2337.
Nitta M, Okamura H, Aizawa S, Yamaizumi M . (1997). Heat shock induces transient p53-dependent cell cycle arrest at G1/S. Oncogene 15: 561–568.
Raval RR, Lau KW, Tran MG, Sowter HM, Mandriota SJ, Li JL . (2005). Contrasting properties of hypoxia-inducible factor 1 (HIF-1) and HIF-2 in von Hippel–Lindau-associated renal cell carcinoma. Mol Cell Biol 25: 5675–5686.
Sanchez-Puig N, Veprintsev DB, Fersht AR . (2005). Binding of natively unfolded HIF-1alpha ODD domain to p53. Mol Cell 17: 11–21.
Schmid T, Zhou J, Kohl R, Brune B . (2004). p300 relieves p53-evoked transcriptional repression of hypoxia-inducible factor-1 (HIF-1). Biochem J 380: 289–295.
Semenza GL . (1999). Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol 15: 551–578.
Semenza GL . (2001). HIF-1 and mechanisms of hypoxia sensing. Curr Opin Cell Biol 13: 167–171.
Shoshani T, Faerman A, Mett I, Zelin E, Tenne T, Gorodin S et al. (2002). Identification of a novel hypoxia-inducible factor 1-responsive gene, RTP801, involved in apoptosis. Mol Cell Biol 22: 2283–2293.
Spruce BA, Campbell LA, McTavish N, Cooper MA, Appleyard MV, O'Neill M et al. (2004). Small molecule antagonists of the sigma-1 receptor cause selective release of the death program in tumor and self-reliant cells and inhibit tumor growth in vitro and in vivo. Cancer Res 64: 4875–4886.
Waldman T, Kinzler KW, Vogelstein B . (1995). p21 is necessary for the p53-mediated G1 arrest in human cancer cells. Cancer Res 55: 5187–5190.
Williams AC, Collard TJ, Paraskeva C . (1999). An acidic environment leads to p53 dependent induction of apoptosis in human adenoma and carcinoma cell lines: implications for clonal selection during colorectal carcinogenesis. Oncogene 18: 3199–3204.
Yim S, Choi SM, Choi Y, Lee N, Chung J, Park H . (2003). Insulin and hypoxia share common target genes but not the hypoxia-inducible factor-1alpha. J Biol Chem 278: 38260–38268.
Zhou J, Schmid T, Brune B . (2003). Tumor necrosis factor-alpha causes accumulation of a ubiquitinated form of hypoxia inducible factor-1alpha through a nuclear factor-kappaB-dependent pathway. Mol Biol Cell 14: 2216–2225.
Acknowledgements
This research was funded in part by a Programme Grant from Cancer Research UK (TRH) and an award from NESTech University Challenge Seed Fund (BAS, MA). We thank Dr Anna Woodward for critical reading of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Achison, M., Boylan, M., Hupp, T. et al. HIF-1α contributes to tumour-selective killing by the sigma receptor antagonist rimcazole. Oncogene 26, 1137–1146 (2007). https://doi.org/10.1038/sj.onc.1209890
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209890
Keywords
This article is cited by
-
Roles of sigma-1 receptors on mitochondrial functions relevant to neurodegenerative diseases
Journal of Biomedical Science (2017)