Abstract
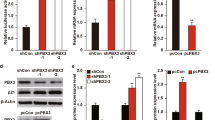
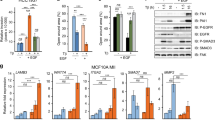
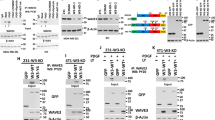
Transforming growth factor-β type 1 (TGF-β) has been implicated as both a tumor suppressor and a tumor promoter in many solid epithelial cancers. We have previously demonstrated that the cyclin dependent kinase (CDK) inhibitor p21 acts as a molecular switch in determining a growth inhibitory versus growth proliferative response to TGF-β in the spontaneously immortalized human mammary epithelial cell line MCF-10A. We now demonstrate that this proliferative effect of TGF-β is mediated through the proinflammatory cytokine, interleukin-1α (IL-1α). Using gene expression array analysis, we identified IL-1α as a cytokine specifically upregulated only in cells lacking p21 and only upon TGF-β stimulation. Cell proliferation assays verified that recombinant IL-1α was capable of inducing a growth proliferative response in p21 null MCF-10A cells, while neutralizing antibodies against IL-1α prevented the growth proliferative effects of TGF-β. Mechanistically, both the CDK and proliferating cell nuclear antigen (PCNA) inhibitory functions of p21 were responsible for preventing TGF-β induced cell proliferation, but only PCNA inhibition by p21 regulated IL-1α gene expression. These studies demonstrate a novel role for IL-1α in mediating a proliferative response to TGF-β signaling, and suggest that therapies directed against IL-1α could abate the growth proliferative effects of TGF-β without compromising its tumor suppressive function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- IL-1α:
-
interleukin-1 alpha
- TGF-β:
-
transforming growth factor-beta
- CDK:
-
cyclin dependent kinase
- PCNA:
-
proliferating cell nuclear antigen
References
Bachman KE, Park BH . (2005). Curr Opin Oncol 17: 49–54.
Bachman KE, Blair BG, Brenner K, Bardelli A, Arena S, Zhou S et al. (2004). Cancer Biol Ther 3: 221–225.
Bittner M, Meltzer P, Chen Y, Jiang Y, Seftor E, Hendrix M et al. (2000). Nature 406: 536–540.
Cayrol C, Knibiehler M, Ducommun B . (1998). Oncogene 16: 311–320.
Chen CR, Kang Y, Massague J . (2001). Proc Natl Acad Sci USA 98: 992–999.
Chou JL, Fan Z, DeBlasio T, Koff A, Rosen N, Mendelsohn J . (1999). Breast Cancer Res Treat 55: 267–283.
Cordenonsi M, Dupont S, Maretto S, Insinga A, Imbriano C, Piccolo S . (2003). Cell 113: 301–314.
Derynck R, Akhurst RJ, Balmain A . (2001). Nat Genet 29: 117–129.
Grille SJ, Bellacosa A, Upson J, Klein-Szanto AJ, van Roy F, Lee-Kwon W et al. (2003). Cancer Res 63: 2172–2178.
Hahn SA, Schutte M, Hoque AT, Moskaluk CA, da Costa LT, Rozenblum E et al. (1996). Science 271: 350–353.
Iavarone A, Massague J . (1997). Nature 387: 417–422.
Islam S, Kim JB, Trendel J, Wheelock MJ, Johnson KR . (2000). J Cell Biochem 78: 141–150.
Kloen P, Jennings CL, Gebhardt MC, Springfield DS, Mankin HJ . (1994a). Int J Cancer 58: 440–445.
Kloen P, Jennings CL, Gebhardt MC, Springfield DS, Mankin HJ . (1994b). Eur J Cancer 30A: 678–682.
Lei X, Bandyopadhyay A, Le T, Sun L . (2002). Oncogene 21: 7514–7523.
Luo Y, Hurwitz J, Massague J . (1995). Nature 375: 159–161.
Markowitz S, Wang J, Myeroff L, Parsons R, Sun L, Lutterbaugh J et al. (1995). Science 268: 1336–1338.
Seoane J, Le HV, Shen L, Anderson SA, Massague J . (2004). Cell 117: 211–223.
Siegel PM, Massague J . (2003). Nat Rev Cancer 3: 807–821.
Theocharis SE, Margeli AP, Klijanienko JT, Kouraklis GP . (2004). Histopathology 45: 103–118.
Turner M, Chantry D, Buchan G, Barrett K, Feldmann M . (1989). J Immunol 143: 3556–3561.
Wakefield LM, Winokur TS, Hollands RS, Christopherson K, Levinson AD, Sporn MB . (1990). J Clin Invest 86: 1976–1984.
Yin JJ, Selander K, Chirgwin JM, Dallas M, Grubbs BG, Wieser R et al. (1999). J Clin Invest 103: 197–206.
Acknowledgements
We thank Dr Bernard Ducommun for providing the wild type and mutant p21 plasmids. This work was supported by an NIH Breast SPORE Grant P50 CA88843, the Maryland Cigarette Restitution Fund, The American Cancer Society (#IRG-58-005-41), The Entertainment Industry Foundation, The Department of Defense Breast Cancer Research Program (DAMD17-03-1-0241), NIH/NCI R01CA109274-01A2 and the Avon Foundation. This work was supported in part by funds from the intramural research program of the National Institute on Aging. BHP is an Avon Scholar for Breast Cancer Research and also receives generous support from The V Foundation for Cancer Research and The Flight Attendant's Medical Research Institute (FAMRI).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Karakas, B., Weeraratna, A., Abukhdeir, A. et al. Interleukin-1 alpha mediates the growth proliferative effects of transforming growth factor-beta in p21 null MCF-10A human mammary epithelial cells. Oncogene 25, 5561–5569 (2006). https://doi.org/10.1038/sj.onc.1209540
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209540
Keywords
This article is cited by
-
Selective therapeutic strategy for p53-deficient cancer by targeting dysregulation in DNA repair
Communications Biology (2021)
-
Robust gene expression programs underlie recurrent cell states and phenotype switching in melanoma
Nature Cell Biology (2020)
-
Integrative analysis of survival-associated gene sets in breast cancer
BMC Medical Genomics (2015)
-
Genetic knockouts and knockins in human somatic cells
Nature Protocols (2007)