Abstract
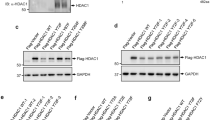
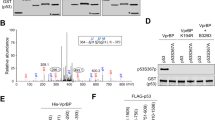
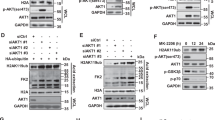
p14ARF is a tumour suppressor which plays a critical role in p53-dependent or -independent cell growth control. Several studies have recently provided evidence that p14ARF can also interfere either directly or indirectly with some components of the RB signalling pathway to mediate its antiproliferative activity. The aim of this study was to explore the existence of direct relationships between p14ARF and RB proteins. We show that p14ARF promotes the accumulation of a hypoacetylated RB protein, when it is upregulated in a model of stable-inducible clones or physiologically induced following cell exposure to cytotoxic agents. Looking for the mechanisms involved in this process, we demonstrate that the histone acetyl transferase Tip60 directly interacts with RB and stimulates its degradation by the proteasome through acetylation of its C-terminus. Furthermore, and consistent with p14ARF-induced RB accumulation, we provide evidence that p14ARF prevents Tip60-mediated RB acetylation, therefore precluding its proteasomal degradation. Overall, our results identify a novel mechanism by which p14ARF controls the RB pathway to trigger its antiproliferative function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
31 July 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/s41388-023-02789-1
References
Bates S, Phillips AC, Clark PA, Stott F, Peters G, Ludwig RL et al. (1998). Nature 395: 124–125.
Berns K, Hijmans EM, Mullenders J, Brummelkamp TR, Velds A, Heimerikx M et al. (2004). Nature 428: 431–437.
Carnero A, Hudson JD, Price CM, Beach DH . (2000). Nat Cell Biol 2: 148–155.
Caron C, Boyault C, Khochbin S . (2005). BioEssays 27: 408–415.
Carrozza MJ, Utley RT, Workman JL, Cote J . (2003). Trends Genet 19: 321–329.
Chan HM, Krstic-Demonacos M, Smith L, Demonacos C, La Thangue NB . (2001). Nat Cell Biol 3: 667–674.
Chau BN, Wang JY . (2003). Nat Rev Cancer 3: 130–138.
Datta A, Nag A, Raychaudhuri P . (2002). Mol Cell Biol 22: 8398–8408.
Eymin B, Gazzeri S, Brambilla C, Brambilla E . (2002). Oncogene 21: 2750–2761.
Eymin B, Karayan L, Seite P, Brambilla C, Brambilla E, Larsen CJ et al. (2001). Oncogene 20: 1033–1041.
Eymin B, Leduc C, Coll JL, Brambilla E, Gazzeri S . (2003). Oncogene 22: 1822–1835.
Ito A, Kawaguchi Y, Lai CH, Kovacs JJ, Higashimoto Y, Appella E et al. (2002). EMBO J 21: 6236–6245.
Legube G, Linares LK, Lemercier C, Scheffner M, Khochbin S, Trouche D . (2002). EMBO J 21: 1704–1712.
Lemercier C, Legube G, Caron C, Louwagie M, Garin J, Trouche D et al. (2003). J Biol Chem 278: 4713–4718.
Lipinski MM, Jacks T . (1999). Oncogene 18: 7873–7882.
Martelli F, Hamilton T, Silver DP, Sharpless NE, Bardeesy N, Rokas M et al. (2001). Proc Natl Acad Sci USA 98: 4455–4460.
Mason SL, Loughran O, La Thangue NB . (2002). Oncogene 21: 4220–4230.
Nguyen DX, Baglia LA, Huang SM, Baker CM, McCance DJ . (2004). EMBO J 23: 1609–1618.
Sherr CJ . (1998). Genes Dev 12: 2984–2991.
Sherr CJ . (2001). Nat Rev Mol Cell Biol 2: 731–737.
Sherr CJ, Roberts JM . (1999). Genes Dev 13: 1501–1512.
Tan X, Wang JY . (1998). Trends Cell Biol 8: 116–120.
Uchida C, Miwa S, Kitagawa K, Hattori T, Isobe T, Otani S et al. (2005). EMBO J 24: 160–169.
Acknowledgements
We thank Dr Bruno Amati (Milan, Italy) for the generous gift of anti-Tip60 antibody and Dr Didier trouche (Toulouse, France), Pr Lars-Gunnar Larsson (Upsala, Sweden) and Dr McCance for the gift of various plasmids used in this work. We are also grateful to Celine Lampreia and Pascal Perron for technical assistance. This work was supported by a grant to EB from La Ligue Nationale Contre le Cancer as an ‘équipe labellisée’ and by the Region Rhône Alpes (Thématique prioritaire cancer and Canceropole 2003, 2005: Oncocell, Epimed, INACancer) (EPIPRO). SK laboratory is supported by a sidaction grant and EC is supported by sidaction from 2004 as well as by ANRS (2002-2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc)
Supplementary information
About this article
Cite this article
Leduc, C., Claverie, P., Eymin, B. et al. RETRACTED ARTICLE: p14ARF promotes RB accumulation through inhibition of its Tip60-dependent acetylation. Oncogene 25, 4147–4154 (2006). https://doi.org/10.1038/sj.onc.1209446
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209446
Keywords
This article is cited by
-
Protein post-translational modifications in the regulation of cancer hallmarks
Cancer Gene Therapy (2023)
-
Post-translational modifications on the retinoblastoma protein
Journal of Biomedical Science (2022)
-
Mutually exclusive acetylation and ubiquitylation of the splicing factor SRSF5 control tumor growth
Nature Communications (2018)
-
p14ARF inhibits the growth of lung adenocarcinoma cells harbouring an EGFR L858R mutation by activating a STAT3-dependent pro-apoptotic signalling pathway
Oncogene (2013)
-
Acetylation controls Notch3 stability and function in T-cell leukemia
Oncogene (2012)