Abstract
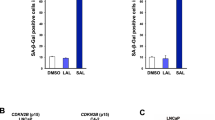
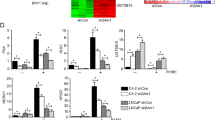
It has been suggested in many studies that combined treatment with chemotherapeutic agents and apoptosis-inducing ligands belonging to TNFR family is a more effective strategy for cancer treatment. However, the role of androgen regulation of TNFR family-induced apoptosis in prostate cancer is poorly understood. In this study, we investigated the dose-dependent effects of androgen on TNF-α and TRAIL-mediated apoptosis in LNCaP. To investigate the interaction between the androgen receptor (AR) and the caspase-2 gene, chromatin immunoprecipitation analysis was used, and we are the first to identify that AR interacts in vivo with an androgen-responsive elements in intron 8 of caspase-2 gene. We have found that DHT inhibited apoptosis in dose-dependent manner. There is a direct, androgen-dependent correlation between the levels of activated Akt and caspase activation after treatment with TNF-α and TRAIL. We have also found that there are at least two different regulatory mechanisms of p53 expression by androgen: at the gene and protein levels. At the same time, the level of AR was found to be higher in LNCaP-si-p53 compared to LNCaP-mock cells. These data indicate that there is a mutual regulation of expression between p53 and AR. Our study suggests that androgen-dependent outcome of apoptotic treatment can occur, at least in part, via the caspase-2, Akt and p53-mediated pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bennet M, Macdonald K, Chan SW, Luzio JP, Simari R and Weissberg P . (1998). Science, 282, 290–293.
Brazil DP, Park J and Hemmings BA . (2002). Cell, 111, 293–303.
Budanov AV, Sablina AA, Feinstein E, Koonin EV and Chumakov PM . (2004). Science, 304, 596–600.
Chan TO, Rittenhouse SE and Tsichlis PN . (1999). Annu. Rev. Biochem., 68, 965–1014.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vessella R, Rosenfeld MG and Sawyers CL . (2004). Nat. Med., 10, 33–39.
Coffey RNT, Watson RWG, O'Neill AJ, Mc Eleny K and Fitzpatrick JM . (2002). Prostate, 53, 300–309.
Degterev A, Boyce M and Yuan J . (2003). Oncogene, 22, 8543–8567.
Gnanapragasam VJ, Robinson MC, Marsh C, Robson CN, Hamdy FC and Leung HY . (2003). Br. J. Cancer, 88, 1432–1438.
Guo Y, Srinivasula SM, Druilhe A, Fernandes-Alnemri T and Alnemri ES . (2002). J. Biol. Chem., 277, 13430–13437.
Guseva NV, Taghiyev AF, Rokhlin OW and Cohen MB . (2004). J. Cell. Biochem., 91, 70–99.
Heisler LE, Evangelou A, Lew AM, Trachtenberg J, Elsholtz HP and Brown TJ . (1997). Mol. Cell. Endocrinol., 126, 59–73.
Hill MM and Hemmings BA . (2002). Pharmacol. Ther., 93, 243–251.
Huang H, Muddiman DC and Tindall DJ . (2004a). J. Biol. Chem., 279, 13866–13877.
Huang H, Zegarra-Moro OL, Benson D and Tindall DJ . (2004b). Oncogene, 23, 2161–2176.
Isaacs JT and Isaacs WB . (2004). Nat. Med., 10, 26–27.
Johnstone RW, Ruefli AA and Lowe SW . (2002). Cell, 108, 153–164.
Lassus P, Opitz-Araya X and Lazebnik Y . (2002). Science, 297, 1352–1354.
Linja MJ, Savinainen KJ, Saramaki OR, Tamella TLJ, Vessella RL and Visakorpi T . (2001). Cancer Res., 61, 3550–3555.
Marchenko ND, Zaika A and Moll UM . (2000). J. Biol. Chem., 275, 16202–16212.
Mayo LD and Donner DB . (2002). Trends Biochem. Sci., 27, 462–467.
Murillo H, Huang H, Schmidt LJ, Smith DI and Tindall DJ . (2001). Endocrinology, 142, 4795–4805.
Nantermet PV, Xu J, Yu Y, Hodor P, Holder D, Adamski S, Gentile MA, Kimmel DB, Harada S-I, Tinel A and Tschopp J . (2004). Science, 304, 843–846.
Nelson WG, De Marzo AM and Isaacs WB . (2003). N. Engl. J. Med., 349, 366–381.
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T, Tokino T, Tanigushi T and Tanaka N . (2000). Science, 288, 1053–1058.
Paroni G, Henderson C, Schneider C and Brancolini C . (2002). J. Biol. Chem., 277, 15147–15161.
Pinski J, Parikh A, Bova S and Isaacs JT . (2001). Cancer Res., 61, 6372–6376.
Reed JC . (2003). Cancer Cell, 3, 17–22.
Robertson JD, Gogvadze V, Kropotov A, Vakifahmetoglu H, Zhivotovsky B and Orrenius S . (2004). EMBO Rep., 5, 643–648.
Rokhlin OW, Bishop GA, Hostager BS, Waldschmidt TJ, Sidorenko SP, Pavloff N, Kiefer MC, Umansky SR, Glover RA and Cohen MB . (1997). Cancer Res., 57, 1758–1768.
Rokhlin OW, Glover RA and Cohen MB . (1998). Cancer Res., 58, 5870–5875.
Rokhlin OW, Glover RA, Taghiyev AF, Guseva NV, Seftor REB, Shyshynova I, Gudkov AV and Cohen MB . (2002a). J. Biol. Chem., 277, 33213–33219.
Rokhlin OW, Gudkov AV, Kwek S, Glover RA, Gewies AS and Cohen MB . (2000). Oncogene, 19, 1959–1968.
Rokhlin OW, Guseva N, Taghiyev A, Knudson CM and Cohen MB . (2001). Oncogene, 20, 2836–2843.
Rokhlin OW, Guseva NV, Taghiyev AF, Glover RA and Cohen MB . (2004). Cancer Biol. Ther., 3, 761–768.
Rokhlin OW, Taghiyev AF, Guseva NV, Glover RA, Syrbu SI and Cohen MB . (2002b). Cancer Biol. Ther., 1, 631–637.
Sonnenschein C, Olea N, Pasanen ME and Soto AM . (1989). Cancer Res., 49, 3474–3481.
Sun M, Yang L, Feldman RI, Sun X-M, Bhalla KN, Jove R, Nicosia SV and Cheng JQ . (2003). J. Biol. Chem., 278, 42992–43000.
Thalman GN, Anezinis PA, Chang SM, Zhau HE, Kim EE, Hopwood VL, Pathak S, von Eschenbach AC and Chung LWK . (1994). Cancer Res., 54, 2577–2781.
Tinel A and Tschopp . (2004). Science, 304, 843–846.
Zornig M, Hueber A, Baum W and Evan G . (2001). Biochem. Biopys. Acta, 1551, F1–F37.
Acknowledgements
We thank Y Lazebnik (Cold Spring Harbor Lab) for monoclonal and polyclonal antibodies to caspase-2 and the expression vector with mutated caspase-2. We thank TJ Brown (The University of Toronto, Canada) for the PC-3(AR)2 cell line and expression vector pCEP4/hAR. This work is supported by NIH Grant CA 87717.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rokhlin, O., Taghiyev, A., Guseva, N. et al. Androgen regulates apoptosis induced by TNFR family ligands via multiple signaling pathways in LNCaP. Oncogene 24, 6773–6784 (2005). https://doi.org/10.1038/sj.onc.1208833
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208833
Keywords
This article is cited by
-
Metformin regulates multiple signaling pathways within castration-resistant human prostate cancer cells
BMC Cancer (2022)
-
Single-cell analysis reveals androgen receptor regulates the ER-to-Golgi trafficking pathway with CREB3L2 to drive prostate cancer progression
Oncogene (2021)
-
Molecular evidence of IGFBP-3 dependent and independent VD3 action and its nonlinear response on IGFBP-3 induction in prostate cancer cells
BMC Cancer (2020)
-
Der Einfluss westlicher Ernährung
Der Deutsche Dermatologe (2019)
-
Genetic and cellular studies highlight that A Disintegrin and Metalloproteinase 19 is a protective biomarker in human prostate cancer
BMC Cancer (2016)