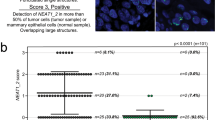
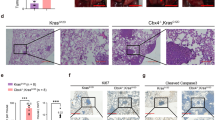
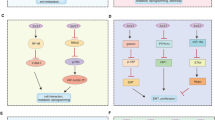
Abstract
Although Betaig-h3 gene has been suggested to modulate cell adhesion and tumor formation, its physiological functions are not well understood. Using human papillomavirus immortalized human bronchial epithelial (BEP2D) cells, we found that Betaig-h3 expression was markedly decreased in asbestos-induced tumorigenic cells. Fusion of tumorigenic and control BEP2D cells resulted in the recovery of Betaig-h3 gene expression to control level and the loss of tumorigenic phenotype. Furthermore, ectopic expression of Betaig-h3 gene in asbestos-induced tumorigenic cells inhibited cell growth in vitro, anchorage independent phenotype, as well as tumorigenicity in nude mice. Betaig-h3 gene is ubiquitously expressed in various normal human tissues, with the exception of the brain, where there is little or no expression. In contrast, there was a decrease or absence in expression of the Betaig-h3 gene in 14 human tumor cell lines of diverse histological types examined, when compared with normal human cells or tissues. The result strongly suggests that loss of Betaig-h3 expression is a frequent event in human cancer and causally related to acquisition of tumorigenic phenotype in asbestos-treated BEP2D cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Billings PC, Herrick DJ, Howard PS, Kucich U, Engelsberg BN, Rosenbloom J . 2000a Am. J. Respir. Cell Mol. Biol. 22: 352–359
Billings PC, Herrick DJ, Kucich U, Engelsberg BN, Abrams WR, Macarak EJ, Rosenbloom J, Howard PS . 2000b J. Cell Biochem. 79: 261–273
Brezinova J, Zemanova Z, Cermak J, Michalova K . 2000 Cancer Genet. Cytogenet. 117: 45–49
Brody AR, Liu JY, Brass D, Corti M . 1997 Environ. Health Perspect. 105: suppl 5 1165–1171
Bron AJ . 2000 Cornea 19: 699–711
Fearon ER, Pierceall WE . 1995 Cancer Surv. 24: 3–17
Fitzpatrick DR, Peroni DJ, Bielefeldt-Ohmann H . 1995 Am. J. Respir. Cell Mol. Biol. 12: 455–460
Genini M, Schwalbe P, Scholl FA, Schafer BW . 1996 Int. J. Cancer 66: 571–577
Gerwin BI . 1994 Am. J. Respir. Cell Mol. Biol. 11: 507–508
Hei TK, Piao CQ, He ZY, Vannais D, Waldren CA . 1992 Cancer Res. 52: 6305–6309
Hei TK, Wu LJ, Piao CQ . 1997 Environ. Health Perspect. 105: suppl 5 1085–1088
Hei TK, Xu A, Louie D, Zhao YL . 2000 Inhal. Toxicol. 12: suppl 3 141–147
Heintz NH, Janssen YM, Mossman BT . 1993 Proc. Natl. Acad. Sci. USA 90: 3299–3304
Jagirdar J, Lee TC, Reibman J, Gold LI, Aston C, Begin R, Rom WN . 1997 Environ. Health Perspect. 105: suppl 5 1197–1203
Jaurand MC . 1997 Environ. Health Perpect. 105: suppl 5 1073–1084
Kamp DW, Weitzman SA . 1999 Thorax 54: 638–652
Kane AB . 1996 IARC Sci. Publ. 140: 11–34
Kim JE, Kim EH, Han EH, Park RW, Park IH, Jun SH, Kim JC, Young MF, Kim IS . 2000 J. Cell Biochem. 77: 169–178
Mossman BT, Kamp DW, Weitzman SA . 1996 Cancer Invest. 14: 464–478
Mossman BT, Faux S, Janssen Y, Jimenez LA, Timblin C, Zanella C, Goldberg J, Walsh E, Barchowsky A, Driscoll K . 1997 Environ. Health Perspect. 105: suppl 5 1121–1125
Murthy SS, Testa JR . 1999 J. Cell. Physiol. 180: 150–157
Ni Z, Liu YQ, Keshava N, Zhou G, Whong W, Ong T . 2000 Mutation Res. 468: 87–92
Pass HI, Mew DJ . 1996 J. Cell Biochem. Suppl. 24: 142–151
Peralta RC, Casson AG, Wang RN, Keshavjee S, Redston M, Bapat B . 1998 Int. J. Cancer 78: 600–605
Perdue TD, Brody AR . 1994 J. Histochem. Cytochem. 42: 1061–1070
Piao CQ, Zhao YL, Hei TK . 2001 Oncogene 20: 7301–7306
Robledo R, Mossman B . 1999 J. Cell. Physiol. 180: 158–166
Salih HR, Nussler V . 2001 Eur. J. Med. Res. 6: 323–332
Schenker T, Trueb B . 1998 Exp. Cell Res. 239: 161–168
Skonier J, Neubauer M, Madisen L, Bennett K, Plowman GD, Purchio AF . 1992 DNA Cell Biol. 11: 511–522
Skonier J, Bennett K, Rothwell V, Kosowski S, Plowman G, Wallace P, Edelhoff S, Disteche C, Neubauer M, Marquardt H . 1994 DNA Cell Biol. 13: 571–584
Suzuki M, Piao CQ, Zhao YL, Hei TK . 2001 Int. J. Mol. Med. 8: 43–47
Willey JC, Broussoud A, Sleemi A, Bennett WP, Cerutti P, Curtis CC . 1991 Cancer Res. 51: 5370–5377
Wu X, Zhao Y, Kemp BL, Amos CI, Siciliano MJ, Spitz MR . 1998 Int. J. Cancer 79: 490–493
Xu A, Wu LJ, Santella RM, Hei TK . 1999 Cancer Res. 59: 5922–5926
Zanella CL, Posada J, Tritton TR, Mossman BT . 1996 Cancer Res. 56: 5334–5338
Zhao YL, Piao CQ, Wu LJ, Suzuki M, Hei TK . 2000 Carcinogenesis 21: 2005–2010
Acknowledgements
The author thanks Dr Paul C Billings for kindly providing anti-Betaigh3 antibody. Work supported in part by grants from the National Institute of Health CA49062, ES07890 and Environmental Health Center Grant P30 ES 09089.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Y., Piao, C. & Hei, T. Downregulation of Betaig-h3 gene is causally linked to tumorigenic phenotype in asbestos treated immortalized human bronchial epithelial cells. Oncogene 21, 7471–7477 (2002). https://doi.org/10.1038/sj.onc.1205891
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205891
Keywords
This article is cited by
-
Soluble TGFBI aggravates the malignancy of cholangiocarcinoma through activation of the ITGB1 dependent PPARγ signalling pathway
Cellular Oncology (2022)
-
The role of TGFBI (βig-H3) in gastrointestinal tract tumorigenesis
Molecular Cancer (2015)
-
TGFBI expression is an independent predictor of survival in adjuvant-treated lung squamous cell carcinoma patients
British Journal of Cancer (2014)
-
The role of TGFBI in mesothelioma and breast cancer: association with tumor suppression
BMC Cancer (2012)
-
TGFBI promoter hypermethylation correlating with paclitaxel chemoresistance in ovarian cancer
Journal of Experimental & Clinical Cancer Research (2012)