Abstract
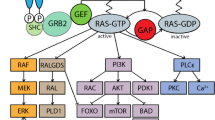
While both nitric oxide synthase-2 (NOS-2) and low molecular weight GTPases, such as Ras and Rho, have been implicated in malignant transformation, the cross talk between these important proteins is ill understood. In this study we examined the ability of H-Ras, RhoA, RhoB and Rac1 to modulate cytokine-induced NOS2. In the normal human liver AKN-1 cell line and in the human non-small cell lung carcinoma cell line, A-549, the ability of the cytokines (INF-γ, IL-1β and TNF-α) to activate NOS-2 was blocked by activated L61-H-Ras whereas dominant negative N17-H-Ras enhanced NOS-2 activation. Consistent with this dominant negative Erk2 as well as a MEK inhibitor also enhanced cytokine activation of NOS-2. Furthermore, activated L63-RhoA blocked whereas activated V14-RhoB enhanced cytokine NOS-2 activation. Activated I115-Racl did not affect NOS-2 activation. These results demonstrate that the Ras/Erk and the Ras/RhoA pathways negatively regulate whereas RhoB enhances cytokine-induced NOS-2. This is the first demonstration that genes that promote malignant transformation such as Ras and RhoA inhibit, whereas genes with tumor suppresser activity such as RhoB enhance NOS2 induction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Abbreviations
- NO:
-
nitric oxide
- NOS:
-
nitric oxide synthase
- INF:
-
interferon
- IL:
-
interleukin
- TNF:
-
tumor necrosis factor
- LPS:
-
lypopolysaccharides
- MAPK:
-
mitogen-activated protein kinase.
References
Barbacid M . 1987 Annu. Rev. Biochem. 56: 779–827
Baron R, Fourcade E, Lajoie-Mazenc I, Allal C, Couderc B, Barbaras R, Favre G, Faye J-C, Pradines A . 2000 Proc. Natl. Acad. Sci. USA 97: 11626–11631
Campbell SL, Khosravi-Far R, Rossman KL, Clark GJ, Der CJ . 1998 Oncogene 17: 1395–1413
Chen Z, Sun J, Pradines A, Favre G, Adnane J, Sebti SM . 2000 J. Biol. Chem. 275: 17974–17978
Cunha FQ, Moncada S, Liew FY . 1992 Biochem Biophys Res. Commun. 182: 1155–1159
Curley SA, Roh MS, Feig B, Oyedeji C, Kleinerman ES, Klostergaard J . 1993 J. Leukoc Biol. 53: 715–721
de Vera ME, Shapiro RA, Nussler AK, Mudgett JS, Simmons RL, Morris Jr SM, Billiar TR, Geller DA . 1996 Proc. Natl. Acad. Sci. USA 93: 1054–1059
Dong Z, Staroselsky AH, Qi X, Xie K, Fidler IJ . 1994 Cancer Res. 54: 789–793
Finder JD, Litz JL, Blaskovich MA, McGuire TF, Qian Y, Hamilton AD, Davies P, Sebti SM . 1997 J. Biol. Chem. 272: 13484–13488
Her J–H, Lakhani S, Zu K, Vila J, Dent P, Sturgill TW, Weber MJ . 1993 Mol. Cell. Biol. 19: 7519–7528
Juang SH, Xie K, Xu L, Wang Y, Yoneda J, Fidler IJ . 1997 Cancer Biother. Radiopharm. 12: 167–175
Khosravi-Far R, Solski PA, Clark GJ, Kinch MS, Der CJ . 1995 Mol. Cell Biol. 15: 6443–6453
Lepoivre M, Boudbid H, Petit JF . 1989 Cancer Res. 49: 1970–1976
Mordan LJ, Burnett TS, Zhang LX, Tom J, Cooney RV . 1993 Carcinogenesis 14: 1555–1559
Muniyappa R, Xu R, Ram JL, Sowers JR . 2000 Am. J. Physiol. Heart Circ. Physiol. 278: H1762–H1768
Nathan CF, Hibbs Jr JB . 1991 Curr. Opin. Immunol. 3: 65–70
Nussler AK, Vergani G, Gollin SM, Dorko K, Morris Jr SM, Demetris AJ, Nomoto M, Beger HG, Strom SC . 1999 In Vitro Cell Dev. Biol. Anim. 35: 190–197
Pahan K, Liu X, McKinney MJ, Wood C, Sheikh FG, Raymond JR . 2000 J. Neurochem 74: 2288–2295
Pahan K, Sheikh FG, Khan M, Namboodiri AM, Singh I . 1998 J. Biol. Chem. 273: 2591–2600
Palmer RM, Ferrige AG, Moncada S . 1987 Nature 327: 524–526
Sambrook J, Fritsch EF, Miniatis T . 1989 Molecular Cloning. A Laboratory Manual 2nd edn Cold Spring Harbor Laboratory Press
Schini VB, Durante W, Elizondo E, Scott-Burden T, Junquero DC, Schafer AI, Vanhoutte PM . 1992 Eur. J. Pharmacol 216: 379–383
Schmidt HH, Walter U . 1994 Cell 78: 919–925
Shi Q, Xiong Q, Wang B, Le X, Khan NA, Xie K . 2000 Cancer Res. 60: 2579–2583
Singh K, Balligand JL, Fischer TA, Smith TW, Kelly RA . 1996 J. Biol. Chem. 271: 1111–1117
Stuehr DJ . 1997 Annu. Rev. Pharmacol Toxicol 37: 339–359
Wang B, Xiong Q, Shi Q, Le X, Abbruzzese JL, Xie K . 2001 Cancer Res. 61: 71–75
Zohn IM, Campbell SL, Khosravi-Far R, Rossman KL, Der CJ . 1998 Oncogene 17: 1415–1438
Acknowledgements
We would like to thank Dr Channing Der for the wild-type and mutants of Ras, RhoA and Rac1, Dr Richard Jove for dominant negative Erk (Her et al., 1993) and Dr Allan Hall for the RhoB mutants. This work was supported by NCI grant CA67771 (SM Sebti).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Delarue, F., Taylor, B. & Sebti, S. Ras and RhoA suppress whereas RhoB enhances cytokine-induced transcription of nitric oxide synthase-2 in human normal liver AKN-1 cells and lung cancer A-549 cells. Oncogene 20, 6531–6537 (2001). https://doi.org/10.1038/sj.onc.1204801
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204801
Keywords
This article is cited by
-
Dynamic transcriptomic analysis of Ischemic Injury in a Porcine Pre-Clinical Model mimicking Donors Deceased after Circulatory Death
Scientific Reports (2018)
-
RhoB is involved in lipopolysaccharide-induced inflammation in mouse in vivo and in vitro
Journal of Physiology and Biochemistry (2013)
-
RhoB mRNA is stabilized by HuR after UV light
Oncogene (2005)
-
Rho GTPases in human carcinogenesis: a tale of excess
Revista de Oncología (2003)