Abstract
Phosphodiesterases (PDEs) are enzymes that degrade cellular cAMP and cGMP and are thus essential for regulating the cyclic nucleotides. At least 11 families of PDEs have been identified, each with a distinctive structure, activity, expression, and tissue distribution. The PDE type-3, -4, and -5 (PDE3, PDE4, PDE5) are localized to specific regions of the cardiomyocyte, such as the sarcoplasmic reticulum and Z-disc, where they are likely to influence cAMP/cGMP signaling to the end effectors of contractility. Several PDE inhibitors exhibit remarkable hemodynamic and inotropic properties that may be valuable to clinical practice. In particular, PDE3 inhibitors have potent cardiotonic effects that can be used for short-term inotropic support, especially in situations where adrenergic stimulation is insufficient. Most relevant to this review, PDE inhibitors have also been found to have cytoprotective effects in the heart. For example, PDE3 inhibitors have been shown to be cardioprotective when given before ischemic attack, whereas PDE5 inhibitors, which include three widely used erectile dysfunction drugs (sildenafil, vardenafil and tadalafil), can induce remarkable cardioprotection when administered either prior to ischemia or upon reperfusion. This article provides an overview of the current laboratory and clinical evidence, as well as the cellular mechanisms by which the inhibitors of PDE3, PDE4 and PDE5 exert their beneficial effects on normal and ischemic hearts. It seems that PDE inhibitors hold great promise as clinically applicable agents that can improve cardiac performance and cell survival under critical situations, such as ischemic heart attack, cardiopulmonary bypass surgery, and heart failure.
Similar content being viewed by others
Introduction
The 3′,5′-cyclic nucleotide phosphodiesterases (PDE) are a class of enzymes that are capable of cleaving the phosphodiester bond in either cAMP or cGMP to yield 5′-cyclic nucleotides. To date, a large number of gene products forming 11 families of PDE have been classified based upon common characteristics, such as protein sequence, structure, enzymatic properties, and sensitivity to inhibitors1. Although some redundancy in hydrolyzing cAMP and cGMP is apparent among these families, it has become increasingly clear that this complexity has functional relevance and that multiple PDE isoforms are involved in creating heterogeneous cyclic nucleotide signaling within the cell2, 3.
At least five PDE isoforms (PDE1, PDE2, PDE3, PDE4, and PDE5) have been observed in the heart2. Of the remaining types, PDE6 is solely associated with photoreceptors in the retina while the expression and function of PDE7-PDE11 in the heart have not been studied in depth due to a lack of specific inhibitors or antibodies. The relative expression levels and importance of each PDE within the heart varies by species, stage of development, and level of cardiac stimulation4. Recently, numerous studies have focused on PDE3, PDE4, and PDE5 and their inhibition for treating of a myriad of human diseases, such as acute heart failure syndromes5, intermittent claudication6, depression7, psychosis8, chronic obstructive pulmonary disease9, pulmonary hypertension10, and erectile dysfunction11. There is significant interest in the potential of PDE inhibitors in the treatment of cardiovascular disease. Several recent review articles have discussed the structure and biochemistry1, 3 of PDE, and the crosstalk12, compartmentalization13, inhibition14, and various physiological and pathological functions of individual PDEs2, 4, 15, 16, 17. A monograph dedicated to PDE has also recently been published18. The primary goal of this article is to provide an overview of the effects of PDE3, PDE4, and PDE5 inhibitors on myocyte contractility and survival in normal and ischemic hearts. Table 1 provides a brief summary of the key information on PDE3, PDE4, and PDE5, including the basic characteristics and tissue distribution of these PDE isozymes, as well as the names and relative selectivity of their inhibitors, which will be discussed in greater detail within their respective sections.
Phosphodiesterases and cyclic nucleotide signaling
The secondary messengers cAMP and cGMP regulate a wide variety of cellular functions and morphological processes in the heart, including inotropism, chronotropism, apoptosis, and hypertrophy19, 20, 21, 22. The dysfunction of cAMP or cGMP plays a role in heart failure19, 23 and cardiomyopathy24. The cytosolic levels of cAMP and cGMP are influenced by their production and degradation. The transmembrane protein adenylyl cyclase converts ATP to 3′,5′-cyclic AMP (cAMP) and pyrophosphate25. This conversion can be initiated by a Gs mechanism where agonal stimulation of localized receptors, such as the β1 adrenergic receptor, causes the α subunit of an associated G-protein to bind GTP, dissociate, and bind to adenylyl cyclase thereby activating cAMP production. Since adenylyl cyclase continues to produce cAMP until its stimulation by Gα-GTP is removed, there is an amplification of the original signal. Adenylyl cyclase can also be inhibited by stimulation of the α-adrenergic receptor through a Gi mechanism26. cAMP binds to the regulatory subunits of cAMP-dependent protein kinase (PKA) leading to activation of the catalytic subunits. Nitric oxide (NO) activates soluble guanylate cyclase (sGC) to produce cGMP and pyrophosphate from GTP27. cGMP may also be produced by membrane-bound particulate fraction guanylate cyclases (pGC), receptor-linked enzymes that are activated by ligands, such as atrial naturetic factor28. cGMP activates the cGMP-dependent protein kinase (PKG), which phosphorylates downstream targets.
A classic example of the contractile effects of modified cyclic nucleotide signaling is the inotropic and chronotropic response that is due to the elevation of cAMP during β-adrenergic stimulation19. On the other hand, an increase in levels of cGMP is generally thought to reduce contractility29, but dose-specific inotropic responses to cGMP activation by NO donors were also reported in isolated cardiomyocytes30, 31, 32. In addition, investigators also demonstrated that the NO donor-induced contractile enhancement in frog33 and rodent34 cardiomyocytes was mediated through a cGMP-independent mechanism. The exact nature of this response remains to be elucidated. The two main factors that contribute to cardiac contractility are calcium transient and myofilament calcium sensitivity. The calcium transient is regulated by many factors, including proteins involved in the excitation-contraction coupling and calcium-induced calcium release, such as L-type voltage gated calcium channel (VGCC)35, 36, 37 and ryanodine receptor (RyR)38, 39, as well as proteins involved in calcium reuptake, such as sarco/endoplasmic reticulum calcium ATPase (SERCA)40, the SERCA regulatory protein, phospholamban (PLB)41, Na+-Ca2+ exchanger (NCX)42, and the NCX regulatory protein, phospholemman (PLM)43, 44. The sensitivity of the myocardial contractile fibers to Ca2+ is regulated by troponin I (cTnI)45, 46, 47, 48, 49, 50, 51. The phosphorylation of specific residues on these proteins by PKA and PKG could affect their structure and activity, and thus influence cardiac contractility. In the coronary vasculature, cAMP and cGMP promote vasodilation that may indirectly affect cardiac performance52, 53. In addition, the above-mentioned cGMP-independent positive inotropic effects of NO in cardiomyocytes33, 34 might be achieved through direct S-nitrosylation of the excitation-contraction coupling elements, such as the L-type calcium channels34, 54 and RyR34, 55 (Figure 1).
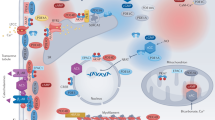
PDE inhibitors and cardiomyocyte contractile signaling crosstalk. PDEs modulate crosstalk and feedback signaling through cAMP, cGMP, and their associated kinases. PDE3, 4, and 5 are phosphorylated and activated by PKA while PDE5 is also phosphorylated by PKG. These PDEs in turn degrade cAMP or cGMP. Ultimately, this signaling cascade influences the activity of PKA and PKG which phosphorylate and modulate end effectors of myocyte contractility. Inhibition of PDE3, 4, and 5 isozymes in this system is thus expected to affect contractility. In particular, PDE3 or PDE4 inhibition may increase cAMP levels and result in enhanced contractility while contractile effects of PDE5 inhibitors may be dose dependent due to crosstalk effects mediated by PDE3. VGCC=Voltage-Gated Calcium Channel; PLB= Phospholamban; PLM=Phospholemman; RyR= Ryanodine Receptor; cTnI= Cardiac Troponin I; S-NO=S-Nitrosylation; L-Ca= L-type Calcium Channel.
Phosphodiesterase 3
Overview of PDE3
The PDE3 family, consisting of the subfamilies PDE3A and PDE3B, which are expressed in cardiac, vascular smooth muscle, and other tissues, has been well studied as a target of therapeutic drug development56. Members of the PDE3 family are capable of hydrolyzing cAMP and cGMP with high affinity14. However, PDE3 has a Vmax, the maximum enzyme velocity, for cAMP that is ten times greater than its Vmax for cGMP, which when taken with its greater affinity for cGMP, means that PDE3 is functionally a cAMP-hydrolyzing enzyme and cGMP is a competitive inhibitor (Ki≈0.6 μmol/L)3, 57. Thus, PDE3 is a crucial communicator between the cGMP and cAMP signaling pathways and is likely the point where low concentrations of NO donors are able to exert a positive inotropic effect32. Additionally, PDE3 contributes to the majority of the cAMP-hydrolyzing activity in subcellular fractions of human myocardium under basal conditions (not Ca2+ stimulated)58.
The PDE3A gene gives rise to three isoforms (PDE3A1/ 2/3) that differ only in the sequence near the N-terminus. PDE3A1 is a 136-kDa protein that is reported to be found only in particulate fractions of human myocardium and contains both NHR1 and NHR2, as well as PKB and PKA phosphorylation sites3, 12. PDE3A2 is a 118-kDa protein found in both the particulate and cytosolic fractions that is a truncated version of PDE3A1 lacking the NHR1 region and likely the PKB phosphorylation site3, 12. PDE3A3 is a 94-kDa protein found only in the cytosolic fraction that lacks both NHRs and all three phosphorylation sites3, 12. The PDE3A and PDE3B isoforms are largely homologous in their amino acid sequence and have similar enzyme kinetics. In the heart, PDE3A and PDE3B are activated by PKA resulting in a negative feedback on the levels of cAMP within the cell59. In fact, it has been suggested that PDE3 may mediate tachyphylaxis in response to long term β-agonist stimulation3. PDE3A1 is not well expressed in cardiomyocytes, but PDE3A2 and PDE3A3 are highly expressed in both cardiomyocytes and vascular smooth muscle56. Multiple PDE3B proteins of differing sizes have been found, but their origin is still unclear3. PDE3B1 has recently been found in the heart tissue of mice and is reported to account for about 30% of the PDE3 activity60. However, it is unknown whether PDE3B1 is found in cardiomyocytes rather than other cell types in the heart tissue.
Regulation of myocardial contractility by PDE3
PDE3 is a crucial modulator of contractility through its control of cAMP signaling. PDE3 inhibitors have long been studied for their positive inotropic effects. Subfamily specific regulation has recently been studied with PDE3A and PDE3B knockout mice61. PDE3A, but not PDE3B, knockout mice showed increased heart rate and contractility compared to wild type mice and these parameters were unresponsive to PDE3 inhibition with cilostamide61. This suggests that the PDE3A isoform is mainly responsible for control of contractility in the heart.
There is increasing evidence that suggests that regulation of many cellular processes results from local modulation and compartmentalization of cyclic nucleotide signaling, rather than global effects13. In neonatal rat cardiomyocytes, β-adrenergic stimulation selectively increases cAMP concentrations in the microdomain near the Z-disc and T-tubules62. Interestingly, studies of the isolated rabbit atria have shown that stimulation of sGC or pGC produced different results on cAMP levels depending on the source of cGMP, suggesting that PDE3 mediates this compartmentalization of signal63. PDE3 activity in the canine myocardium has been shown to be localized to the sarcoplasmic reticulum (SR), making it possible that PDE3 regulates phosphorylation of PLB64. If so, a decrease in PDE3 activity would increase PLB phosphorylation and result in positive inotropy.
In addition, PDE3 could control cardiac contractility through closely regulating cAMP-dependent phosphorylation of VGCC and thus Ca2+ entry into the cardiomyocyte65. cGMP inhibition of PDE3 increases cAMP-stimulated ICa in isolated human atrial myocytes66 and frog ventricular myocytes67. Indeed, it has been suggested that PDE3 plays a role in increasing ICa in nanomolar concentrations of NO donors while PDE2 decreases ICa in micromolar concentrations of NO donors68. This suggestion is consistent with a number of studies showing an increased cardiac contractility under conditions with moderate increases in cGMP levels30, 31, 32. Glucagon inhibition of PDE3 in frog cardiac myocytes also causes ICa stimulation69. Finally, it is conceivable that inhibition of PDE3 in noncardiac tissues may also affect cardiac contractility. Inhibition of PDE3 in rat juxtaglomerular cells contributes to the stimulation of renin secretion70. This in turn causes an increase in angiotensin II, which induces both a downregulation of PDE3A expression and an upregulation of the inducible cAMP early repressor (ICER), a pro-apoptotic protein, in cardiac myocytes71. ICER causes further repression of PDE3A expression and the resulting increase in cAMP/PKA signaling increases ICER expression (PDE3A-ICER feedback loop)72. This positive feedback cycle may increase contractility, but likely represents a pathological condition that leads to heart failure where PDE3 expression is also suppressed2.
Effects of PDE3 inhibitors on ventricular contractility
The PDE3 inhibitory activity of cilostamide was first identified in platelets73. Since then, PDE3 specific inhibitors have been studied for treating chronic heart disease due to the positive inotropic effects of these inhibitors. Clinically relevant PDE3 specific inhibitors include the bipyridine derivatives, milrinone (Primacor®) and amrinone (Inocor®), as well as the imidazole derivative enoximone (Perfan®), all of which are used to treat heart failure. Milrinone is the most well studied and extensively used and is also more potent than amrinone and enoximone. At high concentrations, all three drugs also inhibit PDE4 and PDE514. A clinically relevant PDE3 inhibitor, cilostazol (Pletal®), also inhibits adenosine uptake and is used for the treatment of intermittent claudication74. Another PDE3 inhibitor, pimobendan, which also increases myofilament sensitivity, has been tested to treat congestive heart failure (CHF) in dogs75. Cilostamide, cilostazol, and amrinone appear to have a greater effect on PDE3A than PDE3B14. A recent study comparing the dose dependent effects of milrinone, amrinone, and enoximone in isolated guinea pig hearts found that milrinone increased contractility at much lower concentrations compared to the other PDE3 inhibitors76. At high concentrations, milrinone also increased coronary flow and thus oxygen supply to a greater extent than the other drugs76.
PDE3 inhibitors almost certainly produce their inotropic effects in cardiomyocytes through stimulation of ICa by regulation of the phosphorylation of VGCC via the cAMP-PKA dependent mechanism described above. This effect is independent of β1 adrenergic receptor stimulation in isolated rat hearts because PDE3 inhibition by amrinone still produced a significant inotropic response despite β1 blockade with atenolol77. Milrinone inhibited 70% of the total phospohodiesterase activity in cultured embryonic chick ventricular cells and caused a 43% increase in sarcolemmal uptake of Ca2+ 78. In isolated human and rabbit atrial myocytes, pimobendan increased the basal ICa amplitude79. Milrinone and amrinone also increased calcium influx into calf cardiac Purkinje fiber80. In guinea pig ventricular myocytes, both milrinone and cilostazol increased ICa with milrinone having a more potent effect81. Evidence also exists that supports a mechanism involving PKA phosphorylation of VGCC. The PKA inhibitor H89 blocked the positive inotropy induced by milrinone81. The positive inotropic effects of milrinone in guinea pig82 and dog83 can be attenuated by a calcium channel blocker. Interestingly, the effect of PDE3 inhibition may differ from species to species as amrinone reduced ICa in normal hamster ventricular myocytes and produced no inotropic effects in cardiomyopathic hamsters84.
There is also evidence that PDE3 inhibitors cause positive inotropy independently of increases in ICa and are likely due to increases in cAMP and PKA function in other localized areas resulting in altered calcium handling by SR. For instance, milrinone but not enoximone stimulates Ca2+ release from SR by RyR2 in sheep85. This effect may be due to cAMP-PKA mediated phosphorylation of RyR2. Milrinone increases SR Ca2+ uptake by Ca2+-ATPase in a cAMP-PKA dependent mechanism86, which is also involved in the stimulation of NCX that could conceivably contribute to the inotropic effects of PDE3 inhibitors42. However, high concentrations of milrinone cause a small inhibitory effect on Ca2+ uptake due to the NCX in bovine cardiac sarcolemmal membranes, an effect that may limit excessive inotropic responses80. Cilostazol may have effects secondary to its inhibition of adenosine uptake since ryanodine receptor Ca2+ release is also reduced by adenosine receptor stimulation87. Indeed, increases in both the interstitial and circulatory adenosine concentrations by cilostazol seems to antagonize its inotropic effects by PDE3 stimulation in isolated rabbit hearts6. Conversely, milrinone analogues may also be involved in adenosine receptor blockade, which would increase contractility by disinhibition of adenylyl cyclase near A1 receptors88. Milrinone does not seem to have a calcium sensitizing effect on myofilaments81.
Effects of PDE3 inhibitors on myocardial ischemia-reperfusion injury
A large number of endogenous and exogenous agents are known to cause preconditioning-like effects and there have been significant interests in determining their mechanism of action and application to clinical cardiology. PDE inhibitors are among these agents and the vasodilatory and contractile effects of some PDE inhibitors, in addition to possible cardioprotection, makes research on these agents an especially promising area of investigation. It is notable that cyclic nucleotide levels and PDE activity have been shown to be altered by ischemic preconditioning in isolated perfused rat hearts89.
Early evidence showed that milrinone, when added to the perfusion solution, improved contractile recovery in the isolated perfused rabbit heart following hypoxia/reperfusion injury, suggesting a cardioprotective effect90. Pimobendan, when administered after reperfusion of stunned myocardium in dogs, was able to decrease the contractile dysfunction due to ischemia91. However, pimobendan increased the mortality rate and length of ventricular fibrillation following coronary occlusion in rats92. Amrinone also improved contractile recovery after ischemia-reperfusion (IR) in the isolated rat heart in an extracellular calcium dependent manner93.
Furthermore, injection of milrinone or olprinone before IR reduced infarct size in dogs94. Further dissection of the signaling pathways involved in olprinone-mediated protection revealed that it induced cardioprotection through cAMP/PKA and p38-MAPK dependent, but not PKC dependent mechanisms94. The authors proposed that the end-effectors of p38-MAPK may include opening of the mitochondrial ATP-sensitive potassium channel (mitoKATP) channel or translocation of Hsp27 to the Z-disc to stabilize the myofibril and cytoskeleton. Milrinone, administered before ischemia or at the onset of reperfusion, has recently been shown to attenuate myocardial stunning in pigs95. However, milrinone did not reduce the incidence of arrhythmias due to IR injury in dogs96. The post-myocardial infarction (MI) treatment with milrinone had no significant effect on survival rate in rats97. On the other hand, amrinone preconditioning in isolated perfused rabbit hearts reduced both the infarct size and incidences of ventricular fibrillation as compared to both control and ischemically preconditioned hearts98. Amrinone also decreased IR injury in isolated rat hearts as shown by decreased levels of creatine kinase and lactate dehydrogenase in the coronary effluent99. The authors attributed the protective effects to the possibility of altered Ca2+ handling by amrinone in the myocardium. Pharmacologic inhibition of PDE3 in the post-MI failing heart also provides cardioprotection during subsequent incidences of ischemia, even when ischemic preconditioning was ineffective100. In addition, PDE3 inhibitors are capable of altering protection by other preconditioning modalities. Cilostazol, likely due to inhibition of adenosine uptake, is capable of reducing the transient ischemia duration required for ischemic preconditioning101. However, this study found that cilostazol by itself offered no protection nor was milrinone capable of reducing the time required for ischemic preconditioning101. Cilostazol has recently been shown to potentiate the cardioprotective effects of low-dose atorvastatin in rats by increasing phosphorylation of eNOS102. Indeed, eNOS phosphorylation was about 258% of control values when cilostazol and atorvastatin were given in conjunction. Additionally, cilostazol, but not atorvastatin alone, decreased infarct size while the combination of both drugs provided even greater infarct size reduction102.
The differential cardioprotective effects of PDE3 inhibitors may be due to their differences in modulating inflammatory signaling. In rat cardiomyocytes, all tested concentrations (50, 100, 250 μmol/L) of amrinone inhibit NFκB activation by LPS, but it has a biphasic modulatory effect on TNF driven NFκB activation103. In contrast, milrinone does not affect LPS induced NFκB activation and potentiates TNF driven NFκB activation103. Additionally, amrinone reduces LPS and TNF induction of iNOS while milrinone has no effect on LPS induction of iNOS, but has a biphasic response to TNF induction of iNOS103. Amirone also does not affect production of COX-2 while milrinone increases COX-2 production103. This is notable because NFκB, iNOS, and COX-2 are all known mediators of delayed preconditioning. These findings suggest that many PDE3 inhibitors are capable of causing preconditioning-like effects either by themselves or in conjunction with other approaches. Although the mechanisms still remain to be elucidated through additional studies, the cardioprotective effects that include improvements in both myocyte contractility and viability are indeed very attractive.
Finally, the cardioprotective effects of PDE3 inhibitors may be utilized in cardioplegic preparations for the preservation of myocardium. Amrinone along with St. Thomas Hospital's solution preserved isolated rat hearts after six hours of global ischemia caused by cardioplegic arrest better than combinations of St Thomas Hospital's solution with milrinone104. This was shown by a decrease in lactate dehydrogenase leakage, representing lower ischemic damage that could result from the cardioprotective effects of amrinone. Enoximone, when added to a cardioplegic solution, has also been shown to improve myocardial performance after reperfusion105.
Clinical applications of PDE3 inhibitors
The clinical use of PDE3 inhibitors has focused on treating heart failure patients through positive inotropy. In theory, the hemodynamic and vasodilatory effects of PDE3 inhibitors are also attractive. However, clinical trials of PDE3 inhibitors for these purposes have been somewhat disappointing. An early study published in 1989 using milrinone and/or digoxin in patients with heart failure found increased incidence of ventricular arrhythmias106. The Prospective Randomized Milrinone Survival Evaluation (PROMISE) was a double-blind study involving 1,088 patients with New York Heart Association (NYHA) class III or IV advanced heart failure107. Milrinone therapy (40 mg oral daily) was associated with a 28% increase in mortality in the total population and a 53% increase in mortality in NYHA class IV patients compared to placebo. There was no survival benefit for any subgroup and the milrinone treated patients had a higher incidence of hypotension and syncope. More recently, Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF) looked at milrinone use in 951 patients who were hospitalized with an exacerbation of chronic heart failure (NYHA class III or IV), but did not require inotropic support108. Milrinone (0.5 μg·kg−1·min−1 infusion for 48 h) treated patients had a higher incidences of hypotension and new atrial arrhythmias, but this study did not find a significant increase in mortality. Post-hoc analysis of the findings revealed that patients with heart failure resulting from ischemia fared worse with milrinone compared to placebo while patients with heart failure unrelated to ischemia did better with milrinone when compared to placebo109.
Likewise, despite the promising initial results suggesting that amrinone may improve hemodynamics110, ejection fraction111, and myocardial energy metabolism112, the subsequent larger and better controlled studies have found little or no benefit. For instance, a multicenter, double-blinded study published in 1985 had investigated the effects of long-term oral amrinone therapy (started with 1.5 mg/kg t.i.d and increased to a maximum dosage of 200 mg t.i.d.) in 99 patients with NYHA class III or IV heart failure113. The investigators found no symptomatic improvement or enhanced exercise tolerance after the amrinone therapy. In particular, there were no significant differences in the ejection fraction, cardiothoracic ratio, or incidence of ventricular premature depolarization. Importantly, the adverse side-effects, which are most commonly gastrointestinal complaints, abnormal liver function, thrombocytopenia, and fatigue, occurred in 83% of patients and necessitated withdrawal of amrinone in 34% of the patients. Furthermore, other large studies showed that long-term amrinone treatment did not give sustained benefits after controlled withdrawal from the drug114, 115. However, in a study involving 46 patients with refractory heart failure, intravenous treatment with amrinone (10 μg·kg−1·min−1) was comparable to dobutamine in increasing the cardiac index116. Amrinone also had a higher individualized response rate of pulmonary arterial wedge pressure, more consistent diuresis, and more predictable effective dose as compared to dobutamine. Additionally, in elderly patients (>75 years old) with severe heart failure (NYHA class IV), amrinone (0.75 mg/kg loading followed by 5 or 10 μg·kg−1·min−1 infusion) was found to improve hemodynamics similarly to dobutamine, but resulted in fewer arrhythmic side-effects117.
Enoximone (50 or 75, t.i.d, and titrated up to 100 or 150 mg/day as needed), in a double-blinded trial in 1990 involving 102 patients, was initially found to have no benefit in patients with heart failure (NYHA class II or III) and increased mortality rate118. However, a more recent study involving enoximone used at a lower dosage (25 or 50 mg t.i.d.) in 105 patients with NYHA class II or III heart failure found an increase in exercise capacity with no increase in mortality or incidence of arrhythmias119. The Pimobendan in Congestive Heart Failure (PICO) was a randomized, double-blind trial involving 317 patients with NYHA class II or III moderate heart failure, which compared 2.5 mg and 5 mg daily doses of pimobendan on exercise capacity120. This study found that there was indeed enhanced exercise capacity, but the pimobendan treated patients also had a 1.8 times increase in hazard of death compared to patients on placebo. However in 2002, the Effects of Pimobendan on Chronic Heart Failure (EPOCH) study of 306 patients with NYHA class II or III heart failure found no difference between mortality rates at a dosage of 1.25 or 2.5 mg twice daily121. In fact, there was a decrease in adverse cardiac events in the pimobendan treated groups. Although PDE3 inhibitors are commonly used in the treatment of heart failure, in light of these seemingly contradictory findings, a determination of whether the risks of treatment outweigh the benefits remains to be established.
Nevertheless, the strong inotropic effects of PDE3 inhibitors may prove useful in some situations requiring acute support, such as in cardiac surgery. In a study of 99 patients after cardiac surgery who had low cardiac output, milrinone treatment (50 μg/kg loading dose followed by continuous infusion of 0.375, 0.5, or 0.75 μg·kg−1·min−1 for at least 12 hours) significantly increased the cardiac index and decreased pulmonary capillary wedge pressure122. Milrinone (50 μg/kg loading followed by 0.5 μg·kg−1·min−1 infusion) was also effective in weaning surgical patients off bypass in a randomized, double-blind clinical study involving 32 high risk patients with LV dysfunction or pulmonary hypertension123. More recently, a smaller dose of milrinone (25 μg/kg loading followed by 0.25 μg·kg−1·min−1) has also been shown to be effective in weaning patients with mitral valve stenosis or pulmonary hypertension off bypass124. Milrinone (0.5 μg·kg−1·min−1 infusion, no loading) may also be effective in restoring peripheral circulation after cardiac surgery125. A single bolus (1 or 1.5 mg/kg) of amrinone was sufficient to improve ventricular function126 and maintain circulating blood volume127 following cardiopulmonary bypass. More recently, a prospective double-blind study involving 234 patients found that amrinone (1.5 mg/kg loading followed by 10 μg·kg−1·min−1 infusion) given upon release of aortic cross-clamp was successful in weaning more patients off of the cardiopulmonary bypass as compared with controls (21% vs 7%)128.
PDE3 inhibition is also useful in conditions where standard treatment with β-agonists may not be effective. Whereas downregulation of β-adrenergic receptors suppressed the inotropic reaction to isoproterenol, milrinone and amrinone were still able to exert their contractile effects in a rat model of cardiac hypertrophy129. Because of this feature, PDE3 inhibition may be useful as a pharmacologic bridge to cardiac transplantation in patients with inadequate response to β-agonists. A report of the use of milrinone in ten patients with severe decompensated CHF found that milrinone was an effective treatment, allowing seven patients to survive to transplant130. One patient who was resistant to vasodilators, β-agonists, and balloon counterpulsation was stabilized with milrinone for 21 days and survived to transplant three months later. The other nine patients were stabilized on milrinone between 11 and 51 days. Milrinone therapy was also effective as a bridge to cardiac transplantation in the case of a patient who underwent the removal of a left ventricle (LV) assist device due to sepsis131. Interestingly, the inotropic effects of milrinone may have some applicability in cardiopulmonary resuscitation (CPR). In a pig model of CPR following ligation of the circumflex artery and induction of ventricular fibrillation, the combination of milrinone and vasopressin increased the cardiac index more than other combinations of vasopressin, milrinone, or epinephrine132. Enoximone was reported to be successful in the CPR protocol of two patients who suffered cardiac arrest from overdose of the β-blocker propranolol where conventional advanced life support was ineffective133.
Phosphodiesterase 4
Overview of PDE4
The PDE4 family comprises four genes PDE4A, PDE4B, PDE4C, and PDE4D, which represent over 20 isoforms. In humans, PDE4 expression is ubiquitous, but is highly expressed in brain and heart1. PDE4 activity was discovered early on to be inhibited by rolipram. PDE4 enzymes are cAMP specific with a Vmax lower than those for PDE1 and PDE2, but similar to those of other PDEs3. The PDE4 family is composed of a very large number of splice variants that are organized based upon variations of the region near the N-terminus. PDE4 isoforms are categorized into long forms, short forms, and super-short forms. Members of the PDE4 family have unique and highly conserved upstream conserved regions (UCRs) near the N-terminus. UCR1, formed by 55 amino acids, is proximal to the N-terminus while UCR2, formed by 76 amino acids, is distal15. UCR1 contains a phosphorylation site that results in about a 60%–250% increase in activity of PDE4 when targeted by PKA134, 135. There is an additional phosphorylation site for ERK near the C-terminus that reduces PDE4 activity136. Thus, it is likely that the UCRs help determine the effect of phosphorylation on overall activity of PDE4. PDE4 is involved in numerous cellular processes and is particularly noted for its involvement in cAMP signal compartmentalization and regulation of β-adrenergic signaling. PDE4D3 is localized to the sarcomeric Z-disc137.
Regulation of myocyte contractility by PDE4
PDE4D3 has been determined to be a crucial part of the RyR2 complex on the SR membrane138. The RyR2 is the major Ca2+ release channel on the SR in cardiomyocytes whose open state probability is affected by a number of factors, including cytosolic [Ca2+] and phosphorylation. PDE4D3, along with protein phosphatases, have the purpose of preventing hyperphosphorylation of RyR2 by PKA at Ser2809139. Hyperphosphorylation increases the likelihood of Ca2+ release by the channel in response to stimulation and may cause depletion of the SR resulting in arrhythmia and decreased contractility. These “leaky” RyR2 channels have been observed in human and animal-model failing hearts140. Additionally, PDE4D3 deficient mice demonstrate arrhythmia and cardiomyopathy as well as accelerated heart failure after myocardial infarction138. These effects were ameliorated in mice with RyR2 that could not be PKA phosphorylated138. This provides strong evidence that hyperphospohrylation of RyR2 by PKA is due to the altered regulation by PDE4 and results in cardiac dysfunction.
All PDE4 isoforms can also be closely associated with β-arrestins, which are scaffold proteins that downregulate β2-adrenergic signaling. β-arrestins preferentially bring PDE4D5 to cellular regions that are important for cAMP production under agonal stimulation141. Further evidence demonstrated that PDE4D regulated the β2-adrenergic receptor (β2AR), but not the β1-adrenergic receptor, in neonatal rat cardiomyocytes142. This is important because in cardiomyocytes, PKA is localized to β2AR and PKA phosphorylation of β2AR decreases coupling to Gs and switches coupling to Gi143. The recruitment of PDE4D5 to this region creates a localized sink that regulates cAMP production and PKA activity near β2AR. Indeed, siRNA knockdown of PDE4D5 in human embryonic kidney cells (HEK293) increased phosphorylation of β2AR144. Additionally, catalytically inactive PDE4D5 (Asp556Ala) overexpression in HEK293 cells by adenoviral gene delivery amplified PKA activity at the plasma membrane, but not in the cytoplasm145. Catalytically inactive PDE4D5 expression in neonatal rat cardiomyocytes also caused increased Gs to Gi switching of the β2AR under isoproterenol stimulation, as measured by ERK1/2 activation, an observation that may be the result of increased phosphorylation by PKA145. These findings all suggest that PDE4 regulates PKA phosphorylation of β2AR, which modifies cAMP production with subsequent downstream effects within the cell, and PDE4 is responsible for modulating the amplitude and duration of cAMP response to β-adrenergic stimulation146.
Effects of PDE4 inhibitors on ventricular contractility
The PDE4 inhibitor rolipram was first developed in the 1970s as an effective antidepressant. Rolipram analogues also quickly became the focus of development of drugs for treating asthma and chronic obstructive pulmonary disease (COPD). However, clinical development was halted due to significant side effects, including strong stimulation of emesis and nausea147. Recently, new PDE4 inhibitors with fewer side effects have been developed. Clinically relevant examples include cilomilast (Ariflo®) and roflumilast (Daxas®), which are both under investigation for the treatment of inflammatory airway disease148.
In the heart, PDE4 inhibitors seem to affect contractility only when the myocardium is already stimulated. This is consistent with the finding that β-adrenergic stimulation creates microdomains of increased cAMP concentration close to Z-disc and T tubules in neonatal rat cardiac myocytes62 and the observation that PDE4 regulates β2AR and is localized to the Z-disc. In isolated human and rabbit atrial myocytes, rolipram was unable to increase basal ICa79. Additionally, in isolated frog cardiomyocytes, rolipram was unable to elevate ICa in the absence of elevated cAMP, but increased ICa in submaximal levels of elevated cAMP65. In isolated rat ventricular myocytes, Ro 20-1724, a rolipram analogue and PDE4 inhibitor, by itself could not increase ICa, but was able to produce a significant increase with the PDE3 inhibitor cilostamide149. In rat ventricular myocardium, rolipram by itself did not affect either cAMP levels or basal contractility, but did enhance the contractile responses to glucagon, decreased fade due to tachyphylaxis, and also enhanced the effects of dobutamine150. This was also seen in guinea pig left atria where rolipram and Ro 20-1724 produced negligible or slight increases in contractility at basal conditions, but exerted positive inotropic effects in the presence of forskolin, an adenylyl cyclase activator, and SK&F 94120, a PDE3 inhibitor151. PDE4 inhibition by Ro 20-1724 has been observed to enhance contractility and cAMP accumulation under stimulation by isoproterenol152. In another study, rolipram by itself did not cause increases in contractility in rat papillary muscle, but it markedly potentiated the positive inotropy of milrinone153. Interestingly, the contractile effects of rolipram are conserved in a rat model of hypertrophy where there is downregulation of the β-adrenergic receptor129. However, even under stimulated conditions there is some evidence that PDE4 inhibition may not increase contractility. In rats, Ro 20-1724 did not significantly alter norepinephrine, epinephrine, or dobutamine induced changes but actually significantly attenuated isoproterenol induced increases in the maximum dp/dt, a measure of cardiac contractility154. Additionally, in a case where positive inotropic effects of Ro 20-1724 and rolipram had been found in the dog, these effects were abolished by the β-blockers, propranolol and nadolol, suggesting that at least some level of stimulation is necessary for positive inotropy155.
Effects of PDE4 inhibitors on myocyte viability in ischemic myocardium
The selective PDE4 inhibitor, rolipram (1 mg·kg−1 bolus before ischemia and 1 mg·kg−1·h−1 during reperfusion), alone was unable to protect the canine myocardium in one study of IR injury in vivo156. Rolipram, administered at the time of reperfusion, reduced inflammation, but did not reduce infarct size in a rat model of myocardial infarction157. However, a much lower nonvasodilatory dose of rolipram (1 ng·kg−1·min−1) was able to significantly reduce inflammation and infarct size in another study of IR injury in canine myocardium158. In this study, rolipram also potentiated the cardioprotective effects of the A2A receptor agonist, ATL-146e158. The authors attributed their results to rolipram-induced reduction of inflammation as well as a possible increased level of endogenous adenosine available due to their preparation and occlusion technique158. Importantly, rolipram has been shown to increase the incidence of arrythmias following IR injury in rabbits159. It is still unknown whether other PDE4 inhibitors have cardioprotective effects.
Clinical applications of PDE4 inhibitors
The two selective PDE4 inhibitors, cilomilast and roflimilast, have been shown to be effective in the treatment of asthma or COPD without the emetic side effect of rolipram148. There is limited information available regarding the cardiovascular effects of PDE4 inhibitors in the clinical setting. In five independent studies investigating the pharmokinetics of cilmoliast in healthy volunteers, the drug appeared to be well tolerated at dosages of ≤15 mg/d160. Cilomilast had no significant effects on cardiac function or ECG. In another study in healthy young adults, cilomilast had no significant pharmokinetic interaction with digoxin161. A study investigating the pharmokinetics of roflumilast in 19 healthy volunteers found dose dependent adverse effects, such as headache, asthenia, and abdominal discomfort, that were moderate in severity162. It did not find any clinically relevant changes in blood pressure, heart rate, or ECG.
However, in a study of distal human pulmonary artery smooth muscle cells (PASMC), intracellular cAMP was increased by rolipram, cilomilast, and roflumilast163. All three PDE4 inhibitors significantly attenuated DNA production. Additionally, roflumilast attenuated cell proliferation and MMP-2 and MMP-9 production. The authors suggested that PDE4 inhibitors may prove useful in treating pulmonary artery hypertension by reducing proliferation of PASMCs.
Pretreatment of rats with rolipram before administration of LPS prevented the decline in cardiac index and rise in plasma levels of TNF-α164. Additionally, Ro 20-1724 increased contractility in endotoxemic rats, indicating the potential use of PDE4 inhibitors in the treatment of sepsis165. In addition, rolipram has been investigated in protecting the rat heart against doxorubicin (DOX) induced cardiomyopathy166. In this study, rolipram, given before DOX administration and every two weeks during the experiment, was able to blunt the cardiotoxic effects of both DOX and LPS. Rolipram also reduced apoptosis under these conditions. The authors attributed the cardioprotection to the TNF-α inhibiting effect of rolipram.
These findings indicate that PDE4 inhibitors may have some potential use in cardiovascular protection and contractility. However, the overall picture of these drugs remains vague and additional research is necessary to determine the mechanisms of these actions and clinical applicability.
Phosphodiesterase 5
Overview of PDE5
PDE5 activity was first found in platelets, but it is now known to also be expressed in lung, vascular smooth muscle, heart, and many other tissues. It was originally believed to be similar to another cGMP-specific PDE found in rod and cone photoreceptors (PDE6) but was differentiated by sequence comparison167. The high expression of PDE5 in the corpus cavernosum has been of particular interest due to the ability of PDE5 inhibitors to treat male erectile dysfunction. Recently, inhibition of PDE5 in the lung has also been pursued in the treatment of pulmonary hypertension. There has been controversy over whether PDE5 exists in the cardiomyocyte168. A recent study indicated that PDE5 was not present in normal human left and right ventricular myocardium169. However, there has been strong evidence showing PDE5 expression in the left ventricle and isolated cardiomyocytes from dog170 and mouse171, 172 as well as hypertrophied human left and right ventricles169. PDE5 enzymes are specific in the hydrolysis of cGMP and also contain high affinity binding sites for cGMP173. Interestingly, PDE5 appears to regulate a distinct pool of cGMP that is separate from that produced by atrial natriuretic peptide stimulation of pGC suggesting that it plays a role in compartmentalization of cGMP signaling172. To date, only one family of PDE5, PDE5A, has been described. Three splice variants of PDE5A, PDE5A1/2/3, have been found in humans and differ in their N-terminal region but have similar Km values174. All variants of PDE5A contain two approximately 120 amino acid GAF domains (GAF-A and GAF-B) near the N-terminus, which bind cGMP. Association of cGMP with the main allosteric binding site, GAF-A, stimulates PDE5 activity 9 to 11 fold175, 176, 177. All variants of PDE5 also have a single serine PKA/PKG phosphorylation site, located at Ser102 of human PDE5A1, proximal to the N-terminus from the GAF domains that increases enzyme activity in vitro178, 179. Binding of cGMP to GAF-A appears to regulate phosphorylation178. Indeed, while PKG is the major enzyme responsible for phosphorylation, PKA can also phosphorylate this site when GAF-A is occupied by cGMP179. Conversely, phosphorylation also stabilizes the binding of cGMP to GAF-A179. Interestingly, only PKG activates PDE5 in smooth muscle cells180. PDE5 is localized to the cytosolic fraction and PDE5A is found near the Z-disc in cardiac myocytes170. NOS activity also appears to be necessary for this localization since cardiomyocytes from eNOS knockout mice and wild-type cardiomyocytes chronically treated with L-NAME had diffuse PDE5A distribution181. The diffuse PDE5A distribution has also been seen in isolated cardiomyocytes from a canine model of heart failure170. Furthermore, PDE5 expression is increased in human right ventricle hypertrophy169.
Regulation of myocardial contractility by PDE5
PDE5 is not known to be specifically complexed or associated directly with an end-effector of contractility. Indeed, there is still little knowledge concerning the mechanism of PDE5A localization to the Z-disc. However, this localization may be important since PDE5 activity may influence other structures located near the Z-disc, such as β2AR or RyR2. Such an effect would likely be mediated by cGMP, which dampens many stress responses. In isolated guinea pig right atriums, 8-Br-cGMP exerts a small negative inotropic effect, increases the EC50 of isoproterenol, and decreases the cardiotonic effects of PDE3 inhibition182. Activation of PKG and phosphorylation of troponin I, which results in decreased Ca2+ sensitivity, is a possible mechanism for this effect. cGMP may also decrease Ca2+ flux and Ca2+-dependent action potentials as had been shown in isolated frog ventricles183. A cGMP driven crosstalk effect between PDE5 and PDE3 likely contributes to this regulation as well. In human atrial myocytes, cGMP has been shown to influence Ca2+ level in a concentration dependent manner with PDE3 inhibition and Ca2+ increase at low concentrations and PDE2 stimulation and Ca2+ decrease at high concentrations184. Further evidence for the role PDE3 plays is provided by the observation that in guinea pigs, stimulation of ICa by 8-Br-cAMP, which is resistant to PDE hydrolysis, was not affected by cGMP, but stimulation by isoproterenol or forskolin was potentiated or inhibited by cGMP in a biphasic, concentration-dependent manner185.
Effects of PDE5 inhibitors on ventricular contractility
Specific inhibitors of PDE5 have been the most clinically successful of the class of drugs that inhibit PDEs. Originally conceived as a treatment for angina pectoris, sildenafil has been widely used for the treatment of erectile dysfunction (Viagra®) and pulmonary hypertension (Revatio®). However, limitations such as inhibition of PDE6, resulting in visual effects, and a short half life have led to the development of other specific PDE5 inhibitors such as vardenafil (Levitra®) and tadalafil (Cialis®), which either have more potent action, longer half life, and/or lower PDE6 inhibitory activity.
In anesthetized dogs, high doses of sildenafil were capable of producing positive chronotropic and inotropic effects186. However, this is unlikely to be caused by the direct effect of sildenafil on the myocardium. Instead, it can be explained by reflex sympathetic stimulation caused by vasodilation. Indeed, in humans, sildenafil reduces vagal modulation and increases sympathetic stimulation of the heart187 and also slightly increases contractility in healthy human volunteers188 although the effect in this study is suspect since the measurements were taken 75 min following dobutamine infusion. Sildenafil-induced sympathetic activation seems to be general, as shown by increases in plasma norepinephrine levels and muscle sympathetic nerve activity189.
A number of studies have demonstrated that sildenafil does not seem to have a direct effect on myocardial contractility. Sildenafil (up to 10 μmol/L) did not produce a positive inotropic effect in human atrium, ventricle, or failing ventricle190. Even at high concentrations (10 μmol/L), sildenafil had no effect on the contractility of dog trabeculae carneae191 and this drug did not cause a positive contractile response in human or dog atrial tissue192. In isolated mouse ventricular myocytes, sildenafil slightly enhanced basal shortening, but did not alter calcium transients181. Sildenafil only caused small transient increases in the cardiac index in healthy volunteers193 and caused no changes in patients with coronary artery disease194.
On the other hand, some in vitro or ex vivo studies suggest that sildenafil could increase myocardial contractility. For instance, 10 μmol/L sildenafil treatment causes a net increase in cAMP levels in canine ventricular membrane preparations and bovine heart, an effect that would be expected to increase contractility195. The authors attributed the effect to a possible nonselective inhibition of PDE3 by sildenafil. In fact, in human cardiac tissue, sildenafil (0.1–1.0 μmol/L) increased cAMP levels almost as much as milrinone196.
One possible explanation for the seemingly contradictory observations is that sildenafil may also have negative contractile effects that counteract with any positive inotropic effects. PDE5 inhibition may lead to increased phosphorylation of cTnI by PKG resulting in decreased Ca2+ sensitivity. Further evidence for a PKG related mechanism blunting positive inotropy is that in a rat model of right ventricular hypertrophy (RVH), PDE5 inhibition by sildenafil or MY-5445 significantly increases contractility169. In the same study, there were also decreases in contractility of hearts with RVH following treatment with both PDE5 inhibitors and H89, a PKA inhibitor, suggesting that the positive inotropic effects are mediated through cAMP-PKA, and that there is a net negative inotropic effect from the elevation of cGMP-PKG signaling under PKA inhibition169.
It is generally accepted that PDE5 inhibitors have their greatest effects when related cellular signaling mechanisms are concurrently stimulated. The prime example of this is the observation that the therapeutic effects of PDE5 inhibitors in the corpus cavernosum only occurs secondary to the release of NO. Interestingly, PDE5 inhibitors seem to be capable of negatively modulating β-adrenergic stimulation. Dobutamine-mediated enhancement of systolic and diastolic function in dog was blunted by PDE5 inhibition170. This effect was not seen in dogs with pacing-induced heart failure170. In isoproterenol-stimulated mouse hearts and isolated cardiomyocytes, sildenafil suppressed contractility coincident with cGMP elevation and an increase in PKG activity181. This effect was not observed in eNOS knockout mice but was restored with adenoviral transfer of active eNOS181. Sildenafil also slightly attenuated the isoproterenol-induced increase in force of contraction in human myocardial tissue190, and contractile response to dobutamine was blunted by sildenafil in healthy human volunteers188.
Effects of PDE5 inhibitors on myocardial ischemia-reperfusion injury
The PDE5 inhibitors, sildenafil197, vardenafil197, and tadalafil198, have all been shown to have cardioprotective effects against myocardial IR injury. This phenomenon has been discussed in detail in several recent review articles199, 200, 201. In summary, sildenafil induces myocardial preconditioning in adult rabbits202, infant rabbits203, mice204, dogs205, and rats206 by a mechanism that involves NO signaling and the opening of mitoKATP. A recent study of sildenafil delayed preconditioning in isolated mouse heart has also revealed a role for mitochondrial Ca2+-activated K+ channels in cardioprotection207. Vardenafil and tadalafil also reduce myocardial infarct size in rats198 and rabbits208.
The acute cardioprotective effects of PDE5 inhibitors appear to be dose dependent. In the isolated rat heart, low concentrations of sildenafil (20-50 nmol/L) has been shown to reduce infarct size while higher concentrations had no effect209. This study also showed that sildenafil elevated cGMP and suppressed cAMP levels in the ischemic heart. Sildenafil induces delayed preconditioning 24 h after treatment through a NOS dependent mechanism in the mouse heart204. Sildenafil and vardenafil also have cardioprotective effects when administered at the onset of reperfusion, which depends on the opening of mitoKATP197. Sildenafil protects cardiac myocytes against necrosis and apoptosis through NO signaling and upregulation of Bcl-2171. Sildenafil has additive cardioprotective effects with low-dose atorvastatin by enhancing expression and phosphorylation of eNOS and iNOS210. This PDE5 inhibitor also reduces the incidence of severe arrhythmias during ischemia in dogs211.
Based on numerous studies dissecting the cardioprotective mechanism of action of PDE5 inhibitors, the signaling cascade likely involves activation of endogenous triggers and mediators of cytoprotection. In particular, phosphorylation or induction of NOS results in an elevation of NO and cGMP production from sGC. cGMP then activates PKG, which opens the mitoKATP that prevents the loss of ionic gradients and allows for continued ATP production and calcium transport199, 200. The protective effects of sildenafil are also associated with PKG-dependent phosphorylation of ERK and GSK3β212. The role of mitoKATP is essential since the cardioprotective effects of PDE5 inhibitors can be abrogated by the putative blocker of mitoKATP, 5-HD197, 202. PKC signaling also appears to be involved since the PKC inhibitor, chelerythrine, abolishes sildenafil-induced cardioprotection against IR injury in rabbits213. Interestingly, a low dose of sildenafil (0.06 mg/kg) given at reperfusion did not alter cGMP levels, but was able to reduce infarct size in the wild-type, eNOS null, and iNOS null mice after IR214. Sildenafil's stimulation of the sympathetic nervous system in vivo may also play a role since the autonomic nervous system is involved in remote ischemic preconditioning215. Certain disease states may affect the cardioprotective effect of PDE5 inhibitors. In particular, the infarct size reducing effect of sildenafil in the heart is abolished in diabetic dogs216 and mice214. This blunting effect may be a result of abolishment of sildenafil-induced enhancement of PKG expression in the diseased hearts216.
Clinical applications of PDE5 inhibitors
The hemodynamic profile of PDE5 inhibitors is predominantly characterized by a weak systemic vasodilation, which causes a slight, often transient, drop in systolic and diastolic blood pressure and has been observed in healthy human volunteers treated with sildenafil193, vardenafil217, and tadalafil218. These vasodilatory effects are potentiated by nitrates, which are contraindicated with PDE5 inhibitors193, 218. A hypothetical steal effect of PDE5 inhibitors that may cause detrimental reductions in coronary blood flow has not been observed in studies done on dogs219, 220.
To the contrary, PDE5 inhibition for treatment of pulmonary artery hypertension (PAH) has been well explored. Sildenafil attenuated hypoxia-induced PAH in rats when given before and during the chronic hypoxic treatment (10% O2 for up to 42 days)221. In the clinical setting, a study of 13 patients with PAH found that sildenafil significantly increased cardiac output, decreased pulmonary artery pressure, and decreased pulmonary resistance222. Sildenafil was as effective as inhaled NO for pulmonary vasodilation in children with congenital heart disease223 and was superior to inhaled NO in 13 adult patients with PAH because it increased cardiac output without increasing wedge pressure224. In light of these promising preliminary findings, the Sildenafil Use in Pulmonary HypERtension (SUPER) study included 278 patients with PAH who were randomly assigned to receive placebo, or sildenafil (20, 40, or 80 mg po daily) for 12 weeks225. All groups of patients treated with sildenafil experienced increased exercise capacity as measured by 6-minute walking distance as well as decreased mean pulmonary arterial pressure, and increased cardiac index. Patients who completed the 12 week study could elect to enter a 2-year long term extension trial. Those patients who had previously received placebo, but then were titrated to 80 mg of sildenafil, had a significant increase in exercise capacity at week 12 of the extension period. Recently, the United States Food and Drug Administration approved the use of 20 mg dose of sildenafil for the treatment of PAH. Interestingly, sildenafil, vardenafil, and tadalafil have differential effects in a study of 60 patients with PAH226. The authors found that vardenafil had the most rapid effect on pulmonary arterial pressure, sildenafil and tadalafil, but not vardenafil, caused a reduction in the pulmonary to systemic vascular resistance ratio, and only sildenafil significantly improved arterial oxygenation. A new specific PDE5 inhibitor, udenafil (Zydena®), has also been shown to attenuate PAH227 and the development of compensatory right ventricular hypertrophy due to PAH228 in rats. The hemodynamic effects of sildenafil may also be beneficial in cases of acute pulmonary embolism (APE). In a canine study of APE, sildenafil infusion decreased pulmonary arterial pressure as well as pulmonary vascular resistance229.
Furthermore, initial clinical data showed that sildenafil improved vasodilation in patients with chronic heart failure as analyzed by ultrasound imaging of the brachial artery230 . Sildenafil also dialated epicardial coronary arteries, improved endothelial dysfunction and inhibited platelet activation in a clinical study of 12 patients with coronary artery disease231. Flow-mediated dilation of the brachial artery was also improved by acute and long term treatment with sildenafil in patients with type 2 diabetes232. This is especially interesting because myocardial protection is lost in animal models of diabetes205, 214. Very importantly, the sildenafil induced protection of the endothelium from IR injury was demonstrated in a study involving 10 healthy male volunteers233. These subjects were randomly selected to receive placebo or 50 mg oral sildenafil. Two hours later, endothelium-dependent, flow-mediated dilatation of the radial artery was measured before and after IR induced by 15 min of ischemia at the level of the brachial artery followed by 15 min of reperfusion. Seven days later, all subjects were given the other treatment and underwent the same protocol. Sildenafil blunted the impairment of endothelium-dependent vasodilation. This same study also showed that the protective effect was abolished by glibenclamide, a blocker of KATP channels. Thus, these studies demonstrate that in humans, PDE5 inhibition by sildenafil improves endothelial function during disease states and after IR, possibly mediated by KATP channel.
In a dog model of heart failure, researchers found that sildenafil did not affect coronary flow or myocardial oxygen consumption at rest or during exercise234. In fact, PDE5 expression appears to decrease in failing canine hearts170. However, more recent studies showed an augmented PDE5 activity and/or expression in the systemic and pulmonary vasculature and in the kidney in dogs suffering heart failure235 and in the hypertrophied human right ventricle169. There are a number of clinical studies showing the beneficial effects of sildenafil in heart failure patients. For example, in a study of 23 patients with chronic heart failure, sildenafil significantly increased exercise capacity236. Another study of 20 patients with chronic heart failure and ejection fractions <35% found that the increased cardiac output and exercise capacity following acute treatment with sildenafil can be largely explained by a decrease in LV load, which is caused by the reduced peripheral resistance, aortic and large artery stiffness, as well as wave reflection from peripheral sites237. Thus, sildenafil is a promising therapy in the treatment of heart failure, especially in the presence of endothelial dysfunction. A recent dog study suggested that oral administration of sildenafil may be an efficient, more cost-effective, and better tolerated alternative to intravenous B-type natriuretic peptide infusion for treating the decompensated heart failure235.
Sildenafil may interact with various types of cardiomyopathies. In hypertensive cardiomyopathy induced by L-NAME (20 mg/d, po) in rats, chronic sildenafil treatment (45 mg·kg−1·d−1 po) over eight weeks significantly reduced arterial blood pressure and peripheral vascular resistance238. Importantly, sildenafil also attenuated the decrease in cardiac output and cardiac remodeling. In a mouse model of doxorubicin (5 mg/kg, ip) induced cardiomyopathy, prophylactic treatment with sildenafil (0.7 mg/kg, ip) prevented apoptosis and left ventricular dysfunction239. Sildenafil also prevented the ST interval elongation caused by doxorubicin cardiotoxicity. Additionally, sildenafil also prevented cardiomyopathic changes in the hearts of dystrophin knockout mice240. Sildenafil treatment reduced mRNA levels of atrial natriuretic factor, an indicator of cardiac remodeling, and cardiomyoctes from sildenafil-treated mice were protected against isoproterenol-induced injuries240. However, in a report of a hypertrophic cardiomyopathy patient, Holter monitoring for 24 h before treatment with sildenafil revealed 255 ventricular premature beats and no episode of ventricular tachycardia. After treatment with sildenafil, Holter monitoring revealed 1210 ventricular premature beats and six episodes of unsustained ventricular tachycardia241. This pro-arrhythmic effect of sildenafil needs to be further characterized.
Finally, the cardioprotective and hemodynamic profile of PDE5 inhibitors may have applicability in cardiac surgery. It has been suggested that PDE5 inhibitors could be used: 1) to pharmacologically precondition the heart against ischemia/hypoxia insults caused by aortic cross-clamping and cardiopulmonary bypass; 2) to postcondition the heart with the drug added into the perfusate for reperfusing the ischemic myocardium; and 3) to be used as maintenance therapy to improve coronary flow and perfusion of the myocardium242. However, the true fraction of patients who experience postoperative complications from reperfusion injury appears relatively small and adequately powered trials are necessary to determine effective dosages and protocols for using PDE5 inhibitors as cardioprotectants in the clinical settings201.
Regulatory role of other phosphodiesterases in the heart
Although our review article predominantly focuses on the inhibitors of PDE3, PDE4 and PDE5, we should not underestimate the potentially important role played by PDE1 and PDE2 in regulating cyclic nucleotides and cardiac contractility. In brief, PDE1 is a Ca2+/calmodulin stimulated cytosolic phosphodiesterase found in human heart and is one of the main contributors to cAMP hydrolyzing activity in the presence of Ca2+ in subcellular fractions of human left ventricular myocardium58. It also has been found to be responsible for the majority of cGMP hydrolyzing activity in human ventricular tissue191. However, relevance of PDE1 in human cardiomyocytes is still unclear since PDE1 activity was not identified in isolated rat cardiomyocytes, but instead it was detected in whole myocardium12, 243. There are several nonspecific inhibitors of PDE1 that include nimodipine, which also inhibits the L-type Ca2+ channel244, vinpocetine, which activates the Ca2+-activated K+ channels245, and zaprinast, which also inhibits PDE5246. Many inhibitors that were initially reported to be specific for PDE1 (such as zaprinast, SCH 51866, and IBMX) were later found to inhibit PDE5 as well14. To date, there are no effective and selective PDE1 inhibitors available14. However, the new PDE inhibitor, IC224, holds great promise (selectivity ratio of 127 for PDE1), but it is still unknown whether it is equally selective for the PDE1 subtypes247.
PDE2 also hydrolyzes cAMP and cGMP with high affinities and is stimulated by cGMP. Unlike other phosphodiesterases, the regulation of PDE2 by phosphorylation has yet to be described in detail3. PDE2 may serve as a mediator of cyclic nucleotide crosstalk12. PDE2 attenuates the rise in cAMP resulting from β-adrenergic stimulation in rat ventricular myocytes248. cGMP stimulation of PDE2 also inhibits the cAMP-mediated increase of ICa in amphibian ventricular myocytes68 as well as human atrial myocytes249. The first specific inhibitor discovered for PDE2 is erythro-9-(2-hydroxy-3-nonyl)adenine (EHNA), but its use has been limited due to its inhibitory effects on adenosine deaminase even at low concentrations250. Four more PDE2 specific inhibitors, IC933247, Bay 60-7550251, (9-(6-phenyl-2-oxohex-3-yl)-2-(3,4-dimethoxybenzyl)-purin-6-one (PDP)252, and oxindole253, have recently been described. Nevertheless, since the highly specific inhibitors of PDE1 and PDE2 are still relatively new in experimental studies and far from any clinical use targeting the heart, their potential impact on cardiac contractility cannot be adequately described at the present time.
Summary
This article has extensively reviewed the most updated clinical and laboratory evidence demonstrating the remarkable cardiotonic and/or cytoprotective effects afforded by PDE3 and PDE5 inhibitors, and to the lesser extent, by PDE4 inhibitors (summarized in Table 2). We have further discussed the putative molecular and cellular mechanisms underlying the modulatory effects of PDE3, PDE4, and PDE5 inhibitors on ventricular contractile function and myocyte viability in the normal and ischemic hearts. The cAMP, cGMP, NOS, mitoKATP, and numerous protein kinases are apparently among the principal players forming the signal transduction cascades that result in the positive inotropic (Figure 1) and/or pro-survival (Figure 2) phenotype counteracting myocardial ischemia-reperfusion injury.
Proposed mechanisms for PDE inhibitor-induced cytoprotection against myocardial ischemia-reperfusion injury. PDE5 inhibitors increase cGMP content, which activates PKG. PKG can subsequently activate mitoKATP channel and prevent the loss of ionic gradients in the necrotic pathway of ischemia/reperfusion injury. PDE5 inhibitors simultaneously initiate a signaling cascade involving PKC and ERK, leading to phosphorylation/induction of nitric oxide synthases (NOS) and release of nitric oxide (NO). This pathway also leads to opening of mitoKATP channel. Alternatively, PDE3, and PDE4 inhibitors may increase the activity of PKA leading to phosphorylation of eNOS and p38 MAPK. p38 MAPK activation may potentially result in translocation of 27 kDa heat shock protein (Hsp27) to the Z-disc, an event that may to stabilize the cytoskeleton and contractile fibers.
Ischemic heart diseases remain the number one cause of morbidity and mortality in Western countries and its prevalence has rapidly increased in many developing countries, such as China, due to the profound economical, nutritional, social, and environmental changes. There has been a great deal of demand for advanced surgical or interventional procedures as well as new drug therapies to ameliorate or prevent the devastating functional outcomes of ischemic heart diseases, such as congestive heart failure. Therefore, further vigorous investigations in this dynamic area will undoubtedly pave the way for a series of well designed and controlled clinical trials, which may eventually lead to the use of PDE3 and/or PDE5 inhibitors as the primary line of drug therapy in promoting cardiac performance and cell survival under critical situations, such as ischemic heart attack, cardiopulmonary bypass surgery, and heart failure.
References
Omori K, Kotera J . Overview of PDEs and their regulation. Circ Res 2007; 100: 309–27.
Yan C, Miller CL, Abe J . Regulation of phosphodiesterase 3 and inducible cAMP early repressor in the heart. Circ Res 2007; 100: 489–501.
Bender AT, Beavo JA . Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol Rev 2006; 58: 488–520.
Osadchii OE . Myocardial phosphodiesterases and regulation of cardiac contractility in health and cardiac disease. Cardiovasc Drugs Ther 2007; 21: 171–94.
Shin DD, Brandimarte F, De Luca L, Sabbah HN, Fonarow GC, Filippatos G, et al. Review of current and investigational pharmacologic agents for acute heart failure syndromes. Am J Cardiol 2007; 99: 4A–23A.
Wang S, Cone J, Fong M, Yoshitake M, Kambayashi J, Liu Y . Interplay between inhibition of adenosine uptake and phosphodiesterase type 3 on cardiac function by cilostazol, an agent to treat intermittent claudication. J Cardiovasc Pharmacol 2001; 38: 775–83.
Bobon D, Breulet M, Gerard-Vandenhove MA, Guiot-Goffioul F, Plomteux G, Hernandez M, et al. Is phosphodiesterase inhibition a new mechanism of antidepressant action? A double blind double-dummy study between rolipram and desipramine in hospitalized major and/or endogenous depressives. Eur Arch Psychiatry Neurol Sci 1988; 238: 2–6.
Kanes SJ, Tokarczyk J, Siegel SJ, Bilker W, Abel T, Kelly MP . Rolipram: a specific phosphodiesterase 4 inhibitor with potential antipsychotic activity. Neuroscience 2007; 144: 239–46.
Calverley PM, Sanchez-Toril F, McIvor A, Teichmann P, Bredenbroeker D, Fabbri LM . Effect of 1-year treatment with roflumilast in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2007; 176: 154–61.
Ghofrani HA, Osterloh IH, Grimminger F . Sildenafil: from angina to erectile dysfunction to pulmonary hypertension and beyond. Nat Rev Drug Discov 2006; 5: 689–702.
Corbin JD . Mechanisms of action of PDE5 inhibition in erectile dysfunction. Int J Impot Res 2004; 16 Suppl 1: S4–S7.
Zaccolo M, Movsesian MA . cAMP and cGMP signaling cross-talk: role of phosphodiesterases and implications for cardiac pathophysiology. Circ Res 2007; 100: 1569–78.
Fischmeister R, Castro LR, Abi-Gerges A, Rochais F, Jurevicius J, Leroy J, et al. Compartmentation of cyclic nucleotide signaling in the heart: the role of cyclic nucleotide phosphodiesterases. Circ Res 2006; 99: 816–28.
Lugnier C . Cyclic nucleotide phosphodiesterase (PDE) superfamily: a new target for the development of specific therapeutic agents. Pharmacol Ther 2006; 109: 366–98.
Houslay MD, Baillie GS, Maurice DH . cAMP-Specific phosphodiesterase-4 enzymes in the cardiovascular system: a molecular toolbox for generating compartmentalized cAMP signaling. Circ Res 2007; 100: 950–66.
Kass DA, Takimoto E, Nagayama T, Champion HC . Phosphodiesterase regulation of nitric oxide signaling. Cardiovasc Res 2007; 75: 303–14.
Kass DA, Champion HC, Beavo JA . Phosphodiesterase type 5: expanding roles in cardiovascular regulation. Circ Res 2007; 101: 1084–95.
Beavo JA, Houslay MD, Francis S (editors). Cyclic Nucleotide Phosphodiesterases in Health and Disease. Boca Raton, FL: CRC Press; 2007.
Lohse MJ, Engelhardt S, Eschenhagen T . What is the role of beta-adrenergic signaling in heart failure? Circ Res 2003; 93: 896–906.
Lohmann SM, Fischmeister R, Walter U . Signal transduction by cGMP in heart. Basic Res Cardiol 1991; 86: 503–14.
Tomita H, Nazmy M, Kajimoto K, Yehia G, Molina CA, Sadoshima J . Inducible cAMP early repressor (ICER) is a negative-feedback regulator of cardiac hypertrophy and an important mediator of cardiac myocyte apoptosis in response to beta-adrenergic receptor stimulation. Circ Res 2003; 93: 12–22.
Booz GW . Putting the brakes on cardiac hypertrophy: exploiting the NO-cGMP counter-regulatory system. Hypertension 2005; 45: 341–6.
Sucharov CC . Beta-adrenergic pathways in human heart failure. Expert Rev Cardiovasc Ther 2007; 5: 119–24.
Movsesian MA . Altered cAMP-mediated signalling and its role in the pathogenesis of dilated cardiomyopathy. Cardiovasc Res 2004; 62: 450–9.
Cooper DM . Regulation and organization of adenylyl cyclases and cAMP. Biochem J 2003; 375: 517–29.
Shannon R, Chaudhry M . Effect of alpha1-adrenergic receptors in cardiac pathophysiology. Am Heart J 2006; 152: 842–50.
Denninger JW, Marletta MA . Guanylate cyclase and the NO/cGMP signaling pathway. Biochim Biophys Acta 1999; 1411: 334–50.
Shah AM, MacCarthy PA . Paracrine and autocrine effects of nitric oxide on myocardial function. Pharmacol Ther 2000; 86: 49–86.
Shah AM, Spurgeon HA, Sollott SJ, Talo A, Lakatta EG . 8-bromo-cGMP reduces the myofilament response to Ca2+ in intact cardiac myocytes. Circ Res 1994; 74: 970–8.
Vila-Petroff MG, Younes A, Egan J, Lakatta EG, Sollott SJ . Activation of distinct cAMP-dependent and cGMP-dependent pathways by nitric oxide in cardiac myocytes. Circ Res 1999; 84: 1020–31.
Mohan P, Brutsaert DL, Paulus WJ, Sys SU . Myocardial contractile response to nitric oxide and cGMP. Circulation 1996; 93: 1223–9.
Kojda G, Kottenberg K, Nix P, Schluter KD, Piper HM, Noack E . Low increase in cGMP induced by organic nitrates and nitrovasodilators improves contractile response of rat ventricular myocytes. Circ Res 1996; 78: 91–101.
Chesnais JM, Fischmeister R, Mery PF . Positive and negative inotropic effects of NO donors in atrial and ventricular fibres of the frog heart. J Physiol 1999; 518: 449–61.
Paolocci N, Ekelund UE, Isoda T, Ozaki M, Vandegaer K, Georgakopoulos D, et al. cGMP-independent inotropic effects of nitric oxide and peroxynitrite donors: potential role for nitrosylation. Am J Physiol Heart Circ Physiol 2000; 279: H1982–H1988.
Yamaoka K, Kameyama M . Regulation of L-type Ca2+ channels in the heart: overview of recent advances. Mol Cell Biochem 2003; 253: 3–13.
Kamp TJ, Hell JW . Regulation of cardiac L-type calcium channels by protein kinase A and protein kinase C. Circ Res 2000; 87: 1095–102.
Wang Y, Wagner MB, Joyner RW, Kumar R . cGMP-dependent protein kinase mediates stimulation of L-type calcium current by cGMP in rabbit atrial cells. Cardiovasc Res 2000; 48: 310–22.
Xiao B, Zhong G, Obayashi M, Yang D, Chen K, Walsh MP, et al. Ser-2030, but not Ser-2808, is the major phosphorylation site in cardiac ryanodine receptors responding to protein kinase A activation upon beta-adrenergic stimulation in normal and failing hearts. Biochem J 2006; 396: 7–16.
Zalk R, Lehnart SE, Marks AR . Modulation of the ryanodine receptor and intracellular calcium. Annu Rev Biochem 2007; 76: 367–85.
Vangheluwe P, Raeymaekers L, Dode L, Wuytack F . Modulating sarco(endo)plasmic reticulum Ca2+ ATPase 2 (SERCA2) activity: cell biological implications. Cell Calcium 2005; 38: 291–302.
MacLennan DH, Kranias EG . Phospholamban: a crucial regulator of cardiac contractility. Nat Rev Mol Cell Biol 2003; 4: 566–77.
Wei SK, Ruknudin A, Hanlon SU, McCurley JM, Schulze DH, Haigney MC . Protein kinase A hyperphosphorylation increases basal current but decreases beta-adrenergic responsiveness of the sarcolemmal Na+-Ca2+ exchanger in failing pig myocytes. Circ Res 2003; 92: 897–903.
Song J, Zhang XQ, Ahlers BA, Carl LL, Wang J, Rothblum LI, et al. Serine 68 of phospholemman is critical in modulation of contractility, [Ca2+]i transients, and Na+/Ca2+ exchange in adult rat cardiac myocytes. Am J Physiol Heart Circ Physiol 2005; 288: H2342–H2354.
Zhang XQ, Ahlers BA, Tucker AL, Song J, Wang J, Moorman JR, et al. Phospholemman inhibition of the cardiac Na+/Ca2+ exchanger. Role of phosphorylation. J Biol Chem 2006; 281: 7784–92.
al Hillawi E, Bhandari DG, Trayer HR, Trayer IP . The effects of phosphorylation of cardiac troponin-I on its interactions with actin and cardiac troponin-C. Eur J Biochem 1995; 228: 962–70.
Bilchick KC, Duncan JG, Ravi R, Takimoto E, Champion HC, Gao WD, et al. Heart failure-associated alterations in troponin I phosphorylation impair ventricular relaxation-afterload and force-frequency responses and systolic function. Am J Physiol Heart Circ Physiol 2007; 292: H318–25.
Hunlich M, Begin KJ, Gorga JA, Fishbaugher DE, LeWinter MM, VanBuren P . Protein kinase A mediated modulation of acto-myosin kinetics. J Mol Cell Cardiol 2005; 38: 119–25.
Layland J, Solaro RJ, Shah AM . Regulation of cardiac contractile function by troponin I phosphorylation. Cardiovasc Res 2005; 66: 12–21.
Li MX, Wang X, Sykes BD . Structural based insights into the role of troponin in cardiac muscle pathophysiology. J Muscle Res Cell Motil 2004; 25: 559–79.
Messer AE, Jacques AM, Marston SB . Troponin phosphorylation and regulatory function in human heart muscle: Dephosphorylation of Ser23/24 on troponin I could account for the contractile defect in end-stage heart failure. J Mol Cell Cardiol 2007; 42: 247–59.
Sakthivel S, Finley NL, Rosevear PR, Lorenz JN, Gulick J, Kim S, et al. In vivo and in vitro analysis of cardiac troponin I phosphorylation. J Biol Chem 2005; 280: 703–14.
Qin X, Zheng X, Qi H, Dou D, Raj JU, Gao Y . cGMP-dependent protein kinase in regulation of basal tone and in nitroglycerin- and nitric-oxide-induced relaxation in porcine coronary artery. Pflugers Arch 2007; 454: 913–23.
Kudej RK, Zhang XP, Ghaleh B, Huang CH, Jackson JB, Kudej AB, et al. Enhanced cAMP-induced nitric oxide-dependent coronary dilation during myocardial stunning in conscious pigs. Am J Physiol Heart Circ Physiol 2000; 279: H2967–H2974.
Sun J, Picht E, Ginsburg KS, Bers DM, Steenbergen C, Murphy E . Hypercontractile female hearts exhibit increased S-nitrosylation of the L-type Ca2+ channel alpha1 subunit and reduced ischemia/reperfusion injury. Circ Res 2006; 98: 403–11.
Gonzalez DR, Fernandez IC, Ordenes PP, Treuer AV, Eller G, Boric MP . Differential role of S-nitrosylation and the NO-cGMP-PKG pathway in cardiac contractility. Nitric Oxide 2008; 18: 157–67.
Wechsler J, Choi YH, Krall J, Ahmad F, Manganiello VC, Movsesian MA . Isoforms of cyclic nucleotide phosphodiesterase PDE3A in cardiac myocytes. J Biol Chem 2002; 277: 38072–8.
Degerman E, Belfrage P, Manganiello VC . Structure, localization, and regulation of cGMP-inhibited phosphodiesterase (PDE3). J Biol Chem 1997; 272: 6823–6.
Hambleton R, Krall J, Tikishvili E, Honeggar M, Ahmad F, Manganiello VC, et al. Isoforms of cyclic nucleotide phosphodiesterase PDE3 and their contribution to cAMP hydrolytic activity in subcellular fractions of human myocardium. J Biol Chem 2005; 280: 39168–74.
Shakur Y, Holst LS, Landstrom TR, Movsesian M, Degerman E, Manganiello V . Regulation and function of the cyclic nucleotide phosphodiesterase (PDE3) gene family. Prog Nucleic Acid Res Mol Biol 2001; 66: 241–77.
Patrucco E, Notte A, Barberis L, Selvetella G, Maffei A, Brancaccio M, et al. PI3Kgamma modulates the cardiac response to chronic pressure overload by distinct kinase-dependent and -independent effects. Cell 2004; 118: 375–87.
Sun B, Li H, Shakur Y, Hensley J, Hockman S, Kambayashi J, et al. Role of phosphodiesterase type 3A and 3B in regulating platelet and cardiac function using subtype-selective knockout mice. Cell Signal 2007; 19: 1765–71.
Zaccolo M, Pozzan T . Discrete microdomains with high concentration of cAMP in stimulated rat neonatal cardiac myocytes. Science 2002; 295: 1711–5.
Wen JF, Cui X, Jin JY, Kim SM, Kim SZ, Kim SH, et al. High and low gain switches for regulation of cAMP efflux concentration: distinct roles for particulate GC- and soluble GC-cGMP-PDE3 signaling in rabbit atria. Circ Res 2004; 94: 936–43.
Kauffman RF, Crowe VG, Utterback BG, Robertson DW . LY195115: a potent, selective inhibitor of cyclic nucleotide phosphodiesterase located in the sarcoplasmic reticulum. Mol Pharmacol 1986; 30: 609–16.
Fischmeister R, Hartzell HC . Regulation of calcium current by low-Km cyclic AMP phosphodiesterases in cardiac cells. Mol Pharmacol 1990; 38: 426–33.
Kirstein M, Rivet-Bastide M, Hatem S, Benardeau A, Mercadier JJ, Fischmeister R . Nitric oxide regulates the calcium current in isolated human atrial myocytes. J Clin Invest 1995; 95: 794–802.
Frace AM, Mery PF, Fischmeister R, Hartzell HC . Rate-limiting steps in the beta-adrenergic stimulation of cardiac calcium current. J Gen Physiol 1993; 101: 337–53.
Mery PF, Pavoine C, Belhassen L, Pecker F, Fischmeister R . Nitric oxide regulates cardiac Ca2+ current. Involvement of cGMP-inhibited and cGMP-stimulated phosphodiesterases through guanylyl cyclase activation. J Biol Chem 1993; 268: 26286–95.
Mery PF, Brechler V, Pavoine C, Pecker F, Fischmeister R . Glucagon stimulates the cardiac Ca2+ current by activation of adenylyl cyclase and inhibition of phosphodiesterase. Nature 1990; 345: 158–61.
Friis UG, Jensen BL, Sethi S, Andreasen D, Hansen PB, Skott O . Control of renin secretion from rat juxtaglomerular cells by cAMP-specific phosphodiesterases. Circ Res 2002; 90: 996–1003.
Ding B, Abe J, Wei H, Huang Q, Walsh RA, Molina CA, et al. Functional role of phosphodiesterase 3 in cardiomyocyte apoptosis: implication in heart failure. Circulation 2005; 111: 2469–76.
Ding B, Abe J, Wei H, Xu H, Che W, Aizawa T, et al. A positive feedback loop of phosphodiesterase 3 (PDE3) and inducible cAMP early repressor (ICER) leads to cardiomyocyte apoptosis. Proc Natl Acad Sci USA 2005; 102: 14771–6.
Hidaka H, Hayashi H, Kohri H, Kimura Y, Hosokawa T, Igawa T, et al. Selective inhibitor of platelet cyclic adenosine monophosphate phosphodiesterase, cilostamide, inhibits platelet aggregation. J Pharmacol Exp Ther 1979; 211: 26–30.
Liu Y, Shakur Y, Yoshitake M, Kambayashi Ji J . Cilostazol (pletal): a dual inhibitor of cyclic nucleotide phosphodiesterase type 3 and adenosine uptake. Cardiovasc Drug Rev 2001; 19: 369–86.
Gordon SG, Miller MW, Saunders AB . Pimobendan in heart failure therapy — a silver bullet? J Am Anim Hosp Assoc 2006; 42: 90–3.
Zausig YA, Stowe DF, Zink W, Grube C, Martin E, Graf BM . A comparison of three phosphodiesterase type III inhibitors on mechanical and metabolic function in guinea pig isolated hearts. Anesth Analg 2006; 102: 1646–52.
Osadchii OE, Woodiwiss AJ, Norton GR . Contractile responses to selective phosphodiesterase inhibitors following chronic beta-adrenoreceptor activation. Pflugers Arch 2006; 452: 155–63.
Olson EM, Kim D, Smith TW, Marsh JD . Mechanism of the positive inotropic effect of milrinone in cultured embryonic chick ventricular cells. J Mol Cell Cardiol 1987; 19: 95–104.
Kajimoto K, Hagiwara N, Kasanuki H, Hosoda S . Contribution of phosphodiesterase isozymes to the regulation of the L-type calcium current in human cardiac myocytes. Br J Pharmacol 1997; 121: 1549–56.
Sutko JL, Kenyon JL, Reeves JP . Effects of amrinone and milrinone on calcium influx into the myocardium. Circulation 1986; 73: III52–III58.
Matsui K, Kiyosue T, Wang JC, Dohi K, Arita M . Effects of pimobendan on the L-type Ca2+ current and developed tension in guinea-pig ventricular myocytes and papillary muscle: comparison with IBMX, milrinone, and cilostazol. Cardiovasc Drugs Ther 1999; 13: 105–13.
Alousi AA, Johnson DC . Pharmacology of the bipyridines: amrinone and milrinone. Circulation 1986; 73: III10–III24.
Farah AE, Canniff PC, Bentley RG, Frangakis CJ . The effect of extracellular Ca2+ and related ions on the cardiac action of milrinone. J Cardiovasc Pharmacol 1988; 11: 591–600.
Xiong W, Ferrier GR, Howlett SE . Diminished inotropic response to amrinone in ventricular myocytes from myopathic hamsters is linked to depression of high-gain Ca2+-induced Ca2+ release. J Pharmacol Exp Ther 2004; 310: 761–73.
Holmberg SR, Williams AJ . Phosphodiesterase inhibitors and the cardiac sarcoplasmic reticulum calcium release channel: differential effects of milrinone and enoximone. Cardiovasc Res 1991; 25: 537–45.
Yano M, Kohno M, Ohkusa T, Mochizuki M, Yamada J, Kohno M, et al. Effect of milrinone on left ventricular relaxation and Ca2+ uptake function of cardiac sarcoplasmic reticulum. Am J Physiol Heart Circ Physiol 2000; 279: H1898–H1905.
Ghelardoni S, Frascarelli S, Carnicelli V, Ronca-Testoni S, Zucchi R . Modulation of cardiac sarcoplasmic reticulum calcium release by aenosine: a protein kinase C-dependent pathway. Mol Cell Biochem 2006; 288: 59–64.
Floreani M, Fossa P, Gessi S, Mosti L, Borea PA, Dorigo P . New milrinone analogues: in vitro study of structure-activity relationships for positive inotropic effect, antagonism towards endogenous adenosine, and inhibition of cardiac type III phosphodiesterase. Naunyn Schmiedebergs Arch Pharmacol 2003; 367: 109–18.
Lochner A, Genade S, Tromp E, Opie L, Moolman J, Thomas S, et al. Role of cyclic nucleotide phosphodiesterases in ischemic preconditioning. Mol Cell Biochem 1998; 186: 169–75.
Lowe H, Jacobsohn E . Protection of the hypoxic myocardium by the bispyridine derivatives AWD 122–14 and milrinone: studies on isolated, working right rabbit hearts. Pharmazie 1992; 47: 452–5.
Fukutomi T, Satoh K, Ogoshi S, Ichihara K . Effects of pimobendan and EGIS 9377, cardiotonic agents, and OG-VI, a nucleoside-nucleotide mixture, administered during reperfusion after ischemia on stunned myocardium in dogs. Coron Artery Dis 2000; 11: 83–90.
Carceles MD, Aleixandre F, Fuente T, Lopez-Vidal J, Laorden ML . Effects of rolipram, pimobendan and zaprinast on ischaemia-induced dysrhythmias and on ventricular cyclic nucleotide content in the anaesthetized rat. Eur J Anaesthesiol 2003; 20: 205–11.
Zucchi R, Poddighe R, Mariani M, Ronca G . Effect of amrinone in the working rat heart: influence of ischaemic damage, adenosine and calcium. Drugs Exp Clin Res 1990; 16: 187–95.
Sanada S, Kitakaze M, Papst PJ, Asanuma H, Node K, Takashima S, et al. Cardioprotective effect afforded by transient exposure to phosphodiesterase III inhibitors: the role of protein kinase A and p38 mitogen-activated protein kinase. Circulation 2001; 104: 705–10.
Use T, Makita T, Ureshino H, Cho S, Yoshitomi O, Akiyama D, et al. Milrinone administered before ischemia or just after reperfusion, attenuates myocardial stunning in anesthetized swine. Cardiovasc Drugs Ther 2006; 20: 327–34.
Papp JG, Pollesello P, Varro AF, Vegh AS . Effect of levosimendan and milrinone on regional myocardial ischemia/reperfusion-induced arrhythmias in dogs. J Cardiovasc Pharmacol Ther 2006; 11: 129–35.
Levijoki J, Pollesello P, Kaheinen P, Haikala H . Improved survival with simendan after experimental myocardial infarction in rats. Eur J Pharmacol 2001; 419: 243–8.
Saltman AE, Gaudette GR, Levitsky S, Krukenkamp IB . Amrinone preconditioning in the isolated perfused rabbit heart. Ann Thorac Surg 2000; 70: 609–13.
Rechtman MP, Van der Zypp A, Majewski H . Amrinone reduces ischaemia-reperfusion injury in rat heart. Eur J Pharmacol 2000; 402: 255–62.
Nomura Y, Horimoto H, Mieno S, Nakahara K, Okawa H, Yoshida M, et al. Repetitive preischemic infusion of phosphodiesterase III inhibitor olprinone elicits cardioprotective effects in the failing heart after myocardial infarction. Mol Cell Biochem 2003; 248: 179–84.
Liu Y, Fong M, Cone J, Wang S, Yoshitake M, Kambayashi J . Inhibition of adenosine uptake and augmentation of ischemia-induced increase of interstitial adenosine by cilostazol, an agent to treat intermittent claudication. J Cardiovasc Pharmacol 2000; 36: 351–60.
Manickavasagam S, Ye Y, Lin Y, Perez-Polo RJ, Huang MH, Lui CY, et al. The cardioprotective effect of a statin and cilostazol combination: relationship to Akt and endothelial nitric oxide synthase activation. Cardiovasc Drugs Ther 2007; 21: 321–30.
Chanani NK, Cowan DB, Takeuchi K, Poutias DN, Garcia LM, del Nido PJ, et al. Differential effects of amrinone and milrinone upon myocardial inflammatory signaling. Circulation 2002; 106: I284–9.
Besirli K, Burhani SM, Arslan C, Suzer O, Sayin AG . Effect of combining phosphodiesterase III inhibitors with St Thomas Hospital's solution used as transplantation preservative solution in isolated rat hearts. Transplant Proc 2006; 38: 1253–8.
Koksal C, Suzer O, Bozkurt AK, Koseoglu S . Comparison of enoximone, amrinone, or levosimendan enriched St. Thomas' hospital cardioplegic solutions used for myocardial preservation in isolated guinea pig hearts. Acta Medica (Hradec Kralove) 2002; 45: 93–7.
DiBianco R, Shabetai R, Kostuk W, Moran J, Schlant RC, Wright R . A comparison of oral milrinone, digoxin, and their combination in the treatment of patients with chronic heart failure. N Engl J Med 1989; 320: 677–83.
Packer M, Carver JR, Rodeheffer RJ, Ivanhoe RJ, DiBianco R, Zeldis SM, et al. Effect of oral milrinone on mortality in severe chronic heart failure. The PROMISE Study Research Group. N Engl J Med 1991; 325: 1468–75.
Cuffe MS, Califf RM, Adams KF Jr.,, Benza R, Bourge R, Colucci WS, et al. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial. JAMA 2002; 287: 1541–7.
Felker GM, Benza RL, Chandler AB, Leimberger JD, Cuffe MS, Califf RM, et al. Heart failure etiology and response to milrinone in decompensated heart failure: results from the OPTIME-CHF study. J Am Coll Cardiol 2003; 41: 997–1003.
Maskin CS, Forman R, Klein NA, Sonnenblick EH, LeJemtel TH . Long-term amrinone therapy in patients with severe heart failure: drug-dependent hemodynamic benefits despite progression of disease. Am J Med 1982; 72: 113–8.
Wynne J, Malacoff RF, Benotti JR, Curfman GD, Grossman W, Holman BL, et al. Oral amrinone in refractory congestive heart failure. Am J Cardiol 1980; 45: 1245–9.
Benotti JR, Grossman W, Braunwald E, Carabello BA . Effects of amrinone on myocardial energy metabolism and hemodynamics in patients with severe congestive heart failure due to coronary artery disease. Circulation 1980; 62: 28–34.
Massie B, Bourassa M, DiBianco R, Hess M, Konstam M, Likoff M, et al. Long-term oral administration of amrinone for congestive heart failure: lack of efficacy in a multicenter controlled trial. Circulation 1985; 71: 963–71.
DiBianco R, Shabetai R, Silverman BD, Leier CV, Benotti JR . Oral amrinone for the treatment of chronic congestive heart failure: results of a multicenter randomized double-blind and placebo-controlled withdrawal study. J Am Coll Cardiol 1984; 4: 855–66.
Rettig G, Sen S, Frohlig G, Schieffer H, Bette L . Withdrawal of long-term amrinone therapy in patients with congestive heart failure: a placebo controlled trial. Eur Heart J 1986; 7: 628–31.
Marcus RH, Raw K, Patel J, Mitha A, Sareli P . Comparison of intravenous amrinone and dobutamine in congestive heart failure due to idiopathic dilated cardiomyopathy. Am J Cardiol 1990; 66: 1107–12.
Rich MW, Woods WL, Davila-Roman VG, Morello PJ, Kurz H, Barbarash R, et al. A randomized comparison of intravenous amrinone versus dobutamine in older patients with decompensated congestive heart failure. J Am Geriatr Soc 1995; 43: 271–4.
Uretsky BF, Jessup M, Konstam MA, Dec GW, Leier CV, Benotti J, et al. Multicenter trial of oral enoximone in patients with moderate to moderately severe congestive heart failure. Lack of benefit compared with placebo. Enoximone Multicenter Trial Group. Circulation 1990; 82: 774–80.
Lowes BD, Higginbotham M, Petrovich L, DeWood MA, Greenberg MA, Rahko PS, et al. Low-dose enoximone improves exercise capacity in chronic heart failure. Enoximone Study Group. J Am Coll Cardiol 2000; 36: 501–8.
Lubsen J, Just H, Hjalmarsson AC, La Framboise D, Remme WJ, Heinrich-Nols J, et al. Effect of pimobendan on exercise capacity in patients with heart failure: main results from the Pimobendan in Congestive Heart Failure (PICO) trial. Heart 1996; 76: 223–31.
Effects of Pimobendan on Chronic Heart Failure Study (EPOCH Study). Effects of pimobendan on adverse cardiac events and physical activities in patients with mild to moderate chronic heart failure: the effects of pimobendan on chronic heart failure study (EPOCH study). Circ J 2002; 66: 149–57.
Feneck RO . Effects of variable dose milrinone in patients with low cardiac output after cardiac surgery. European Multicenter Trial Group. Am Heart J 1991; 121: 1995–9.
Doolan LA, Jones EF, Kalman J, Buxton BF, Tonkin AM . A placebo-controlled trial verifying the efficacy of milrinone in weaning high-risk patients from cardiopulmonary bypass. J Cardiothorac Vasc Anesth 1997; 11: 37–41.
Oztekin I, Yazici S, Oztekin DS, Goksel O, Issever H, Canik S . Effects of low-dose milrinone on weaning from cardiopulmonary bypass and after in patients with mitral stenosis and pulmonary hypertension. Yakugaku Zasshi 2007; 127: 375–83.
Kochi K, Kyo S, Yokote Y, Ueda K, Asano H, Koyanagi T, et al. The effect of milrinone for the shock patients after cardiac surgery. Kyobu Geka 1999; 52: 239–42.
Kikura M, Levy JH, Bailey JM, Shanewise JS, Michelsen LG, Sadel SM . A bolus dose of 1.5 mg/kg amrinone effectively improves low cardiac output state following separation from cardiopulmonary bypass in cardiac surgical patients. Acta Anaesthesiol Scand 1998; 42: 825–33.
Hamada Y, Kawachi K, Nakata T, Tsunooka N, Takano S, Kohtani T . Effects of a phosphodiesterase III inhibitor on circulating blood volume after cardiopulmonary bypass. Heart Vessels 2000; 15: 70–3.
Lewis KP, Appadurai IR, Pierce ET, Halpern EF, Bode RH Jr., Prophylactic amrinone for weaning from cardiopulmonary bypass. Anaesthesia 2000; 55: 627–33.
Osadchii O, Norton G, Woodiwiss A . Inotropic responses to phosphodiesterase inhibitors in cardiac hypertrophy in rats. Eur J Pharmacol 2005; 514: 201–8.
Sherry KM, Locke TJ . Use of milrinone in cardiac surgical patients. Cardiovasc Drugs Ther 1993; 7: 671–5.
Koyanagi T, Kyo S, Ouchi H, Asano H, Yokote Y, Omoto R . Clinical experience of milrinone as a pharmacological bridge to heart transplantation. Jpn J Thorac Cardiovasc Surg 2000; 48: 460–3.
Palmaers T, Albrecht S, Heuser F, Leuthold C, Schuettler J, Schmitz B . Milrinone combined with vasopressin improves cardiac index after cardiopulmonary resuscitation in a pig model of myocardial infarction. Anesthesiology 2007; 106: 100–6.
Sandroni C, Cavallaro F, Caricato A, Scapigliati A, Fenici P, Antonelli M . Enoximone in cardiac arrest caused by propranolol: two case reports. Acta Anaesthesiol Scand 2006; 50: 759–61.
Richter W, Jin SL, Conti M . Splice variants of the cyclic nucleotide phosphodiesterase PDE4D are differentially expressed and regulated in rat tissue. Biochem J 2005; 388: 803–11.
Sette C, Conti M . Phosphorylation and activation of a cAMP-specific phosphodiesterase by the cAMP-dependent protein kinase. Involvement of serine 54 in the enzyme activation. J Biol Chem 1996; 271: 16526–34.
Dodge-Kafka KL, Soughayer J, Pare GC, Carlisle Michel JJ, Langeberg LK, Kapiloff MS, et al. The protein kinase A anchoring protein mAKAP coordinates two integrated cAMP effector pathways. Nature 2005; 437: 574–8.
Verde I, Pahlke G, Salanova M, Zhang G, Wang S, Coletti D, et al. Myomegalin is a novel protein of the golgi/centrosome that interacts with a cyclic nucleotidephosphodiesterase. J Biol Chem 2001; 276: 11189–98.
Lehnart SE, Wehrens XH, Reiken S, Warrier S, Belevych AE, Harvey RD, et al. Phosphodiesterase 4D deficiency in the ryanodine-receptor complex promotes heart failure and arrhythmias. Cell 2005; 123: 25–35.
Lehnart SE . Novel targets for treating heart and muscle disease: stabilizing ryanodine receptors and preventing intracellular calcium leak. Curr Opin Pharmacol 2007; 7: 225–32.
Wehrens XH, Lehnart SE, Reiken S, Vest JA, Wronska A, Marks AR . Ryanodine receptor/calcium release channel PKA phosphorylation: a critical mediator of heart failure progression. Proc Natl Acad Sci USA 2006; 103: 511–8.
Bolger GB, McCahill A, Huston E, Cheung YF, McSorley T, Baillie GS, et al. The unique amino-terminal region of the PDE4D5 cAMP phosphodiesterase isoform confers preferential interaction with beta-arrestins. J Biol Chem 2003; 278: 49230–8.
Xiang Y, Naro F, Zoudilova M, Jin SL, Conti M, Kobilka B . Phosphodiesterase 4D is required for beta2 adrenoceptor subtype-specific signaling in cardiac myocytes. Proc Natl Acad Sci USA 2005; 102: 909–14.
Lefkowitz RJ, Pierce KL, Luttrell LM . Dancing with different partners: protein kinase a phosphorylation of seven membrane-spanning receptors regulates their G protein-coupling specificity. Mol Pharmacol 2002; 62: 971–4.
Lynch MJ, Baillie GS, Mohamed A, Li X, Maisonneuve C, Klussmann E, et al. RNA silencing identifies PDE4D5 as the functionally relevant cAMP phosphodiesterase interacting with beta arrestin to control the protein kinase A/AKAP79-mediated switching of the beta2-adrenergic receptor to activation of ERK in HEK293B2 cells. J Biol Chem 2005; 280: 33178–89.
Baillie GS, Sood A, McPhee I, Gall I, Perry SJ, Lefkowitz RJ, et al. beta-Arrestin-mediated PDE4 cAMP phosphodiesterase recruitment regulates beta-adrenoceptor switching from Gs to Gi. Proc Natl Acad Sci USA 2003; 100: 940–5.
Mongillo M, McSorley T, Evellin S, Sood A, Lissandron V, Terrin A, et al. Fluorescence resonance energy transfer-based analysis of cAMP dynamics in live neonatal rat cardiac myocytes reveals distinct functions of compartmentalized phosphodiesterases. Circ Res 2004; 95: 67–75.
Scott AI, Perini AF, Shering PA, Whalley LJ . In-patient major depression: is rolipram as effective as amitriptyline? Eur J Clin Pharmacol 1991; 40: 127–9.
Huang Z, Mancini JA . Phosphodiesterase 4 inhibitors for the treatment of asthma and COPD. Curr Med Chem 2006; 13: 3253–62.
Verde I, Vandecasteele G, Lezoualc'h F, Fischmeister R . Characterization of the cyclic nucleotide phosphodiesterase subtypes involved in the regulation of the L-type Ca2+ current in rat ventricular myocytes. Br J Pharmacol 1999; 127: 65–74.
Juan-Fita MJ, Vargas ML, Kaumann AJ, Hernandez Cascales J . Rolipram reduces the inotropic tachyphylaxis of glucagon in rat ventricular myocardium. Naunyn Schmiedebergs Arch Pharmacol 2004; 370: 324–9.
Muller B, Lugnier C, Stoclet JC . Involvement of rolipram-sensitive cyclic AMP phosphodiesterase in the regulation of cardiac contraction. J Cardiovasc Pharmacol 1990; 16: 796–803.
Katano Y, Endoh M . Differential effects of Ro 20–1724 and isobutylmethylxanthine on the basal force of contraction and beta-adrenoceptor-mediated response in the rat ventricular myocardium. Biochem Biophys Res Commun 1990; 167: 123–9.
Shahid M, Nicholson CD . Comparison of cyclic nucleotide phosphodiesterase isoenzymes in rat and rabbit ventricular myocardium: positive inotropic and phosphodiesterase inhibitory effects of Org 30029, milrinone and rolipram. Naunyn Schmiedebergs Arch Pharmacol 1990; 342: 698–705.
Herzer WA, Thomas NJ, Carcillo JA, Tofovic SP, Jackson EK . Effects of type IV phosphodiesterase inhibition on cardiac function in the presence and absence of catecholamines. J Cardiovasc Pharmacol 1998; 32: 769–76.
Weishaar RE, Kobylarz-Singer DC, Steffen RP, Kaplan HR . Subclasses of cyclic AMP-specific phosphodiesterase in left ventricular muscle and their involvement in regulating myocardial contractility. Circ Res 1987; 61: 539–47.
Simpson PJ, Schelm JA, Smallwood JK, Clay MP, Lindstrom TD . Inhibition of granulocyte cAMP-phosphodiesterase by rolipram in vivo is not sufficient to protect the canine myocardium from reperfusion injury. J Cardiovasc Pharmacol 1992; 19: 987–95.
McVey M, Perrone MH, Clark KL . Does tumor necrosis factor-alpha (TNF-alpha) contribute to myocardial reperfusion injury in anesthetized rats? Gen Pharmacol 1999; 32: 41–5.
Glover DK, Riou LM, Ruiz M, Sullivan GW, Linden J, Rieger JM, et al. Reduction of infarct size and postischemic inflammation from ATL-146e, a highly selective adenosine A2A receptor agonist, in reperfused canine myocardium. Am J Physiol Heart Circ Physiol 2005; 288: H1851–H1858.
Holbrook M, Coker SJ . Effects of zaprinast and rolipram on platelet aggregation and arrhythmias following myocardial ischaemia and reperfusion in anaesthetized rabbits. Br J Pharmacol 1991; 103: 1973–9.
Zussman BD, Benincosa LJ, Webber DM, Clark DJ, Cowley H, Kelly J, et al. An overview of the pharmacokinetics of cilomilast (Ariflo), a new, orally active phosphodiesterase 4 inhibitor, in healthy young and elderly volunteers. J Clin Pharmacol 2001; 41: 950–8.
Zussman BD, Kelly J, Murdoch RD, Clark DJ Mbchb, Schubert C, et al. Cilomilast: pharmacokinetic and pharmacodynamic interactions with digoxin. Clin Ther 2001; 23: 921–31.
Bethke TD, Bohmer GM, Hermann R, Hauns B, Fux R, Morike K, et al. Dose-proportional intraindividual single- and repeated-dose pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor. J Clin Pharmacol 2007; 47: 26–36.
Growcott EJ, Spink KG, Ren X, Afzal S, Banner KH, Wharton J . Phosphodiesterase type 4 expression and anti-proliferative effects in human pulmonary artery smooth muscle cells. Respir Res 2006; 7: 9.
Dutta P, Ryan DE, Tabrizchi R . The influence of phosphodiesterase inhibitor, rolipram, on hemodynamics in lipopolysaccharide-treated rats. Jpn J Pharmacol 2001; 85: 241–9.
Thomas NJ, Carcillo JA, Herzer WA, Mi Z, Tofovic SP, Jackson EK . Type IV phosphodiesterase inhibition improves cardiac contractility in endotoxemic rats. Eur J Pharmacol 2003; 465: 133–9.
Mohamed HE, Asker ME, Ali SI, el Fattah TM . Protection against doxorubicin cardiomyopathy in rats: role of phosphodiesterase inhibitors type 4. J Pharm Pharmacol 2004; 56: 757–68.
McAllister-Lucas LM, Sonnenburg WK, Kadlecek A, Seger D, Trong HL, Colbran JL, et al. The structure of a bovine lung cGMP-binding, cGMP-specific phosphodiesterase deduced from a cDNA clone. J Biol Chem 1993; 268: 22863–73.
Reffelmann T, Kloner RA . Therapeutic potential of phosphodiesterase 5 inhibition for cardiovascular disease. Circulation 2003; 108: 239–44.
Nagendran J, Archer SL, Soliman D, Gurtu V, Moudgil R, Haromy A, et al. Phosphodiesterase type 5 is highly expressed in the hypertrophied human right ventricle, and acute inhibition of phosphodiesterase type 5 improves contractility. Circulation 2007; 116: 238–48.
Senzaki H, Smith CJ, Juang GJ, Isoda T, Mayer SP, Ohler A, et al. Cardiac phosphodiesterase 5 (cGMP-specific) modulates beta-adrenergic signaling in vivo and is down-regulated in heart failure. FASEB J 2001; 15: 1718–26.
Das A, Xi L, Kukreja RC . Phosphodiesterase-5 inhibitor sildenafil preconditions adult cardiac myocytes against necrosis and apoptosis. Essential role of nitric oxide signaling. J Biol Chem 2005; 280: 12944–55.
Takimoto E, Belardi D, Tocchetti CG, Vahebi S, Cormaci G, Ketner EA, et al. Compartmentalization of cardiac beta-adrenergic inotropy modulation by phosphodiesterase type 5. Circulation 2007; 115: 2159–67.
Francis SH, Lincoln TM, Corbin JD . Characterization of a novel cGMP binding protein from rat lung. J Biol Chem 1980; 255: 620–6.
Lin CS, Lau A, Tu R, Lue TF . Expression of three isoforms of cGMP-binding cGMP-specific phosphodiesterase (PDE5) in human penile cavernosum. Biochem Biophys Res Commun 2000; 268: 628–35.
Liu L, Underwood T, Li H, Pamukcu R, Thompson WJ . Specific cGMP binding by the cGMP binding domains of cGMP-binding cGMP specific phosphodiesterase. Cell Signal 2002; 14: 45–51.
Zoraghi R, Bessay EP, Corbin JD, Francis SH . Structural and functional features in human PDE5A1 regulatory domain that provide for allosteric cGMP binding, dimerization, and regulation. J Biol Chem 2005; 280: 12051–63.
Rybalkin SD, Rybalkina IG, Shimizu-Albergine M, Tang XB, Beavo JA . PDE5 is converted to an activated state upon cGMP binding to the GAF A domain. EMBO J 2003; 22: 469–78.
Turko IV, Francis SH, Corbin JD . Binding of cGMP to both allosteric sites of cGMP-binding cGMP-specific phosphodiesterase (PDE5) is required for its phosphorylation. Biochem J 1998; 329: 505–10.
Corbin JD, Turko IV, Beasley A, Francis SH . Phosphorylation of phosphodiesterase-5 by cyclic nucleotide-dependent protein kinase alters its catalytic and allosteric cGMP-binding activities. Eur J Biochem 2000; 267: 2760–7.
Rybalkin SD, Rybalkina IG, Feil R, Hofmann F, Beavo JA . Regulation of cGMP-specific phosphodiesterase (PDE5) phosphorylation in smooth muscle cells. J Biol Chem 2002; 277: 3310–7.
Takimoto E, Champion HC, Belardi D, Moslehi J, Mongillo M, Mergia E, et al. cGMP catabolism by phosphodiesterase 5A regulates cardiac adrenergic stimulation by NOS3-dependent mechanism. Circ Res 2005; 96: 100–9.
Muller B, Lugnier C, Stoclet JC . Implication of cyclic AMP in the positive inotropic effects of cyclic GMP-inhibited cyclic AMP phosphodiesterase inhibitors on guinea pig isolated left atria. J Cardiovasc Pharmacol 1990; 15: 444–51.
Hartzell HC, Fischmeister R . Opposite effects of cyclic GMP and cyclic AMP on Ca2+ current in single heart cells. Nature 1986; 323: 273–5.
Vandecasteele G, Verde I, Rucker-Martin C, Donzeau-Gouge P, Fischmeister R . Cyclic GMP regulation of the L-type Ca2+ channel current in human atrial myocytes. J Physiol 2001; 533: 329–40.
Ono K, Trautwein W . Potentiation by cyclic GMP of beta-adrenergic effect on Ca2+ current in guinea-pig ventricular cells. J Physiol 1991; 443: 387–404.
Sugiyama A, Satoh Y, Shiina H, Takahara A, Yoneyama M, Hashimoto K . Cardiac electrophysiologic and hemodynamic effects of sildenafil, a PDE5 inhibitor, in anesthetized dogs. J Cardiovasc Pharmacol 2001; 38: 940–6.
Piccirillo G, Nocco M, Lionetti M, Moise A, Naso C, Marigliano V, et al. Effects of sildenafil citrate (viagra) on cardiac repolarization and on autonomic control in subjects with chronic heart failure. Am Heart J 2002; 143: 703–10.
Borlaug BA, Melenovsky V, Marhin T, Fitzgerald P, Kass DA . Sildenafil inhibits beta-adrenergic-stimulated cardiac contractility in humans. Circulation 2005; 112: 2642–9.
Phillips BG, Kato M, Pesek CA, Winnicki M, Narkiewicz K, Davison D, et al. Sympathetic activation by sildenafil. Circulation 2000; 102: 3068–73.
Cremers B, Scheler M, Maack C, Wendler O, Schafers HJ, Sudkamp M, et al. Effects of sildenafil (viagra) on human myocardial contractility, in vitro arrhythmias, and tension of internal mammaria arteries and saphenous veins. J Cardiovasc Pharmacol 2003; 41: 734–43.
Wallis RM, Corbin JD, Francis SH, Ellis P . Tissue distribution of phosphodiesterase families and the effects of sildenafil on tissue cyclic nucleotides, platelet function, and the contractile responses of trabeculae carneae and aortic rings in vitro. Am J Cardiol 1999; 83: 3C–12C.
Corbin J, Rannels S, Neal D, Chang P, Grimes K, Beasley A, et al. Sildenafil citrate does not affect cardiac contractility in human or dog heart. Curr Med Res Opin 2003; 19: 747–52.
Jackson G, Benjamin N, Jackson N, Allen MJ . Effects of sildenafil citrate on human hemodynamics. Am J Cardiol 1999; 83: 13C–20C.
Herrmann HC, Chang G, Klugherz BD, Mahoney PD . Hemodynamic effects of sildenafil in men with severe coronary artery disease. N Engl J Med 2000; 342: 1622–6.
Sugiyama A, Takeuchi N, Saegusa Y, Sugita M, Hashimoto K . Molecular mechanisms of cardiostimulatory effects of sildenafil. Jpn J Pharmacol 2002; 88: 362–4.
Stief CG, Uckert S, Becker AJ, Harringer W, Truss MC, Forssmann WG, et al. Effects of sildenafil on cAMP and cGMP levels in isolated human cavernous and cardiac tissue. Urology 2000; 55: 146–50.
Salloum FN, Takenoshita Y, Ockaili RA, Daoud VP, Chou E, Yoshida K, et al. Sildenafil and vardenafil but not nitroglycerin limit myocardial infarction through opening of mitochondrial KATP channels when administered at reperfusion following ischemia in rabbits. J Mol Cell Cardiol 2007; 42: 453–8.
Sesti C, Florio V, Johnson EG, Kloner RA . The phosphodiesterase-5 inhibitor tadalafil reduces myocardial infarct size. Int J Impot Res 2007; 19: 55–61.
Kukreja RC, Ockaili R, Salloum F, Yin C, Hawkins J, Das A, et al. Cardioprotection with phosphodiesterase-5 inhibition — a novel preconditioning strategy. J Mol Cell Cardiol 2004; 36: 165–73.
Kukreja RC, Salloum F, Das A, Ockaili R, Yin C, Bremer YA, et al. Pharmacological preconditioning with sildenafil: Basic mechanisms and clinical implications. Vascul Pharmacol 2005; 42: 219–32.
Raja SG . Cardioprotection with sildenafil: implications for clinical practice. Curr Med Chem 2006; 13: 3155–64.
Ockaili R, Salloum F, Hawkins J, Kukreja RC . Sildenafil (Viagra) induces powerful cardioprotective effect via opening of mitochondrial KATP channels in rabbits. Am J Physiol Heart Circ Physiol 2002; 283: H1263–H1269.
Bremer YA, Salloum F, Ockaili R, Chou E, Moskowitz WB, Kukreja RC . Sildenafil citrate (viagra) induces cardioprotective effects after ischemia/reperfusion injury in infant rabbits. Pediatr Res 2005; 57: 22–7.
Salloum F, Yin C, Xi L, Kukreja RC . Sildenafil induces delayed preconditioning through inducible nitric oxide synthase-dependent pathway in mouse heart. Circ Res 2003; 92: 595–7.
Krolikowski JG, Pagel PS, Warltier DC, Kersten JR . Diabetes abolishes sildenafil-induced preconditioning in dogs. FASEB J 2004; 18: A262.
Das S, Maulik N, Das DK, Kadowitz PJ, Bivalacqua TJ . Cardioprotection with sildenafil, a selective inhibitor of cyclic 3′,5′-monophosphate-specific phosphodiesterase 5. Drugs Exp Clin Res 2002; 28: 213–9.
Wang X, Fisher PW, Xi L, Kukreja RC . Essential role of mitochondrial Ca2+-activated and ATP-sensitive K+ channels in sildenafil-induced late cardioprotection. J Mol Cell Cardiol 2008; 44: 105–13.
Salloum FN, Ockaili RA, Wittkamp M, Marwaha VR, Kukreja RC . Vardenafil: a novel type 5 phosphodiesterase inhibitor reduces myocardial infarct size following ischemia/reperfusion injury via opening of mitochondrial KATP channels in rabbits. J Mol Cell Cardiol 2006; 40: 405–11.
du Toit EF, Rossouw E, Salie R, Opie LH, Lochner A . Effect of sildenafil on reperfusion function, infarct size, and cyclic nucleotide levels in the isolated rat heart model. Cardiovasc Drugs Ther 2005; 19: 23–31.
Rosanio S, Ye Y, Atar S, Rahman AM, Freeberg SY, Huang MH, et al. Enhanced cardioprotection against ischemia-reperfusion injury with combining sildenafil with low-dose atorvastatin. Cardiovasc Drugs Ther 2006; 20: 27–36.
Nagy O, Hajnal A, Parratt JR, Vegh A . Sildenafil (Viagra) reduces arrhythmia severity during ischaemia 24 h after oral administration in dogs. Br J Pharmacol 2004; 141: 549–51.
Das A, Xi L, Kukreja RC . Protein kinase G dependent cardioprotective mechanism of phosphodiesterase-5 inhibition involves phosphorylation of ERK and GSK3beta. J Biol Chem 2008; 283: 29572–85.
Das A, Ockaili R, Salloum F, Kukreja RC . Protein kinase C plays an essential role in sildenafil-induced cardioprotection in rabbits. Am J Physiol Heart Circ Physiol 2004; 286: H1455–H1460.
Elrod JW, Greer JJ, Lefer DJ . Sildenafil-mediated acute cardioprotection is independent of the NO/cGMP pathway. Am J Physiol Heart Circ Physiol 2007; 292: H342–H347.
Loukogeorgakis SP, Panagiotidou AT, Broadhead MW, Donald A, Deanfield JE, MacAllister RJ . Remote ischemic preconditioning provides early and late protection against endothelial ischemia-reperfusion injury in humans: role of the autonomic nervous system. J Am Coll Cardiol 2005; 46: 450–6.
Jamnicki-Abegg M, Weihrauch D, Chiari PC, Krolikowski JG, Pagel PS, Warltier DC, et al. Diabetes abolishes sildenafil-induced cGMP-dependent protein kinase-I expression and cardioprotection. J Cardiovasc Pharmacol 2007; 50: 670–6.
Bischoff E . Vardenafil preclinical trial data: potency, pharmacodynamics, pharmacokinetics, and adverse events. Int J Impot Res 2004; 16 Suppl 1: S34–S37.
Kloner RA, Mitchell M, Emmick JT . Cardiovascular effects of tadalafil. Am J Cardiol 2003; 92: 37M–46M.
Traverse JH, Chen YJ, Du R, Bache RJ . Cyclic nucleotide phosphodiesterase type 5 activity limits blood flow to hypoperfused myocardium during exercise. Circulation 2000; 102: 2997–3002.
Przyklenk K, Kloner RA . Sildenafil citrate (Viagra) does not exacerbate myocardial ischemia in canine models of coronary artery stenosis. J Am Coll Cardiol 2001; 37: 286–92.
Sebkhi A, Strange JW, Phillips SC, Wharton J, Wilkins MR . Phosphodiesterase type 5 as a target for the treatment of hypoxia-induced pulmonary hypertension. Circulation 2003; 107: 3230–5.
Bhatia S, Frantz RP, Severson CJ, Durst LA, McGoon MD . Immediate and long-term hemodynamic and clinical effects of sildenafil in patients with pulmonary arterial hypertension receiving vasodilator therapy. Mayo Clin Proc 2003; 78: 1207–13.
Schulze-Neick I, Hartenstein P, Li J, Stiller B, Nagdyman N, Hubler M, et al. Intravenous sildenafil is a potent pulmonary vasodilator in children with congenital heart disease. Circulation 2003; 108 Suppl 1: II167–II173.
Michelakis E, Tymchak W, Lien D, Webster L, Hashimoto K, Archer S . Oral sildenafil is an effective and specific pulmonary vasodilator in patients with pulmonary arterial hypertension: comparison with inhaled nitric oxide. Circulation 2002; 105: 2398–403.
Galie N, Ghofrani HA, Torbicki A, Barst RJ, Rubin LJ, Badesch D, et al. Sildenafil citrate therapy for pulmonary arterial hypertension. N Engl J Med 2005; 353: 2148–57.
Ghofrani HA, Voswinckel R, Reichenberger F, Olschewski H, Haredza P, Karadas B, et al. Differences in hemodynamic and oxygenation responses to three different phosphodiesterase-5 inhibitors in patients with pulmonary arterial hypertension: a randomized prospective study. J Am Coll Cardiol 2004; 44: 1488–96.
Kang KK, Ahn GJ, Sohn YS, Ahn BO, Kim WB . DA-8159, a potent cGMP phosphodiesterase inhibitor, attenuates monocrotaline-induced pulmonary hypertension in rats. Arch Pharm Res 2003; 26: 612–9.
Kang KK, Ahn GJ, Sohn YS, Ahn BO, Kim WB . DA-8159, a new PDE5 inhibitor, attenuates the development of compensatory right ventricular hypertrophy in a rat model of pulmonary hypertension. J Int Med Res 2003; 31: 517–28.
Dias-Junior CA, Vieira TF, Moreno H Jr, Evora PR, Tanus-Santos JE . Sildenafil selectively inhibits acute pulmonary embolism-induced pulmonary hypertension. Pulm Pharmacol Ther 2005; 18: 181–6.
Katz SD, Balidemaj K, Homma S, Wu H, Wang J, Maybaum S . Acute type 5 phosphodiesterase inhibition with sildenafil enhances flow-mediated vasodilation in patients with chronic heart failure. J Am Coll Cardiol 2000; 36: 845–51.
Halcox JP, Nour KR, Zalos G, Mincemoyer RA, Waclawiw M, Rivera CE, et al. The effect of sildenafil on human vascular function, platelet activation, and myocardial ischemia. J Am Coll Cardiol 2002; 40: 1232–40.
Desouza C, Parulkar A, Lumpkin D, Akers D, Fonseca VA . Acute and prolonged effects of sildenafil on brachial artery flow-mediated dilatation in type 2 diabetes. Diabetes Care 2002; 25: 1336–9.
Gori T, Sicuro S, Dragoni S, Donati G, Forconi S, Parker JD . Sildenafil prevents endothelial dysfunction induced by ischemia and reperfusion via opening of adenosine triphosphate-sensitive potassium channels: a human in vivo study. Circulation 2005; 111: 742–6.
Chen Y, Traverse JH, Hou M, Li Y, Du R, Bache RJ . Effect of PDE5 inhibition on coronary hemodynamics in pacing-induced heart failure. Am J Physiol Heart Circ Physiol 2003; 284: H1513–H1520.
Forfia PR, Lee M, Tunin RS, Mahmud M, Champion HC, Kass DA . Acute phosphodiesterase 5 inhibition mimics hemodynamic effects of B-type natriuretic peptide and potentiates B-type natriuretic peptide effects in failing but not normal canine heart. J Am Coll Cardiol 2007; 49: 1079–88.
Bocchi EA, Guimaraes G, Mocelin A, Bacal F, Bellotti G, Ramires JF . Sildenafil effects on exercise, neurohormonal activation, and erectile dysfunction in congestive heart failure: a double-blind, placebo-controlled, randomized study followed by a prospective treatment for erectile dysfunction. Circulation 2002; 106: 1097–103.
Hirata K, Adji A, Vlachopoulos C, O'Rourke MF . Effect of sildenafil on cardiac performance in patients with heart failure. Am J Cardiol 2005; 96: 1436–40.
Ferreira-Melo SE, Yugar-Toledo JC, Coelho OR, De Luca IM, Tanus-Santos JE, Hyslop S, et al. Sildenafil reduces cardiovascular remodeling associated with hypertensive cardiomyopathy in NOS inhibitor-treated rats. Eur J Pharmacol 2006; 542: 141–7.
Fisher PW, Salloum F, Das A, Hyder H, Kukreja RC . Phosphodiesterase-5 inhibition with sildenafil attenuates cardiomyocyte apoptosis and left ventricular dysfunction in a chronic model of doxorubicin cardiotoxicity. Circulation 2005; 111: 1601–10.
Khairallah M, Khairallah RJ, Young ME, Allen BG, Gillis MA, Danialou G, et al. Sildenafil and cardiomyocyte-specific cGMP signaling prevent cardiomyopathic changes associated with dystrophin deficiency. Proc Natl Acad Sci USA 2008; 105: 7028–33.
Stauffer JC, Ruiz V, Morard JD . Subaortic obstruction after sildenafil in a patient with hypertrophic cardiomyopathy. N Engl J Med 1999; 341: 700–1.
Fung E, Fiscus RR, Yim AP, Angelini GD, Arifi AA . The potential use of type-5 phosphodiesterase inhibitors in coronary artery bypass graft surgery. Chest 2005; 128: 3065–73.
Bode DC, Kanter JR, Brunton LL . Cellular distribution of phosphodiesterase isoforms in rat cardiac tissue. Circ Res 1991; 68: 1070–9.
Georget M, Mateo P, Vandecasteele G, Lipskaia L, Defer N, Hanoune J, et al. Cyclic AMP compartmentation due to increased cAMP-phosphodiesterase activity in transgenic mice with a cardiac-directed expression of the human adenylyl cyclase type 8 (AC8). FASEB J 2003; 17: 1380–91.
Wu SN, Li HF, Chiang HT . Vinpocetine-induced stimulation of calcium-activated potassium currents in rat pituitary GH3 cells. Biochem Pharmacol 2001; 61: 877–92.
Lugnier C, Schoeffter P, Le Bec A, Strouthou E, Stoclet JC . Selective inhibition of cyclic nucleotide phosphodiesterases of human, bovine and rat aorta. Biochem Pharmacol 1986; 35: 1743–51.
Snyder PB, Esselstyn JM, Loughney K, Wolda SL, Florio VA . The role of cyclic nucleotide phosphodiesterases in the regulation of adipocyte lipolysis. J Lipid Res 2005; 46: 494–503.
Mongillo M, Tocchetti CG, Terrin A, Lissandron V, Cheung YF, Dostmann WR, et al. Compartmentalized phosphodiesterase-2 activity blunts beta-adrenergic cardiac inotropy via an NO/cGMP-dependent pathway. Circ Res 2006; 98: 226–34.
Rivet-Bastide M, Vandecasteele G, Hatem S, Verde I, Benardeau A, Mercadier JJ, et al. cGMP-stimulated cyclic nucleotide phosphodiesterase regulates the basal calcium current in human atrial myocytes. J Clin Invest 1997; 99: 2710–8.
Podzuweit T, Nennstiel P, Muller A . Isozyme selective inhibition of cGMP-stimulated cyclic nucleotide phosphodiesterases by erythro-9-(2-hydroxy-3-nonyl) adenine. Cell Signal 1995; 7: 733–8.
Boess FG, Hendrix M, van der Staay FJ, Erb C, Schreiber R, van Staveren W, et al. Inhibition of phosphodiesterase 2 increases neuronal cGMP, synaptic plasticity and memory performance. Neuropharmacology 2004; 47: 1081–92.
Seybold J, Thomas D, Witzenrath M, Boral S, Hocke AC, Burger A, et al. Tumor necrosis factor-alpha-dependent expression of phosphodiesterase 2: role in endothelial hyperpermeability. Blood 2005; 105: 3569–76.
Chambers RJ, Abrams K, Garceau NY, Kamath AV, Manley CM, Lilley SC, et al. A new chemical tool for exploring the physiological function of the PDE2 isozyme. Bioorg Med Chem Lett 2006; 16: 307–10.
Sudo T, Tachibana K, Toga K, Tochizawa S, Inoue Y, Kimura Y, et al. Potent effects of novel anti-platelet aggregatory cilostamide analogues on recombinant cyclic nucleotide phosphodiesterase isozyme activity. Biochem Pharmacol 2000; 59: 347–56.
Szilagyi S, Pollesello P, Levijoki J, Haikala H, Bak I, Tosaki A, et al. Two inotropes with different mechanisms of action: contractile, PDE-inhibitory and direct myofibrillar effects of levosimendan and enoximone. J Cardiovasc Pharmacol 2005; 46: 369–76.
Brunkhorst D, der Leyen H, Meyer W, Nigbur R, Schmidt-Schumacher C, Scholz H . Relation of positive inotropic and chronotropic effects of pimobendan, UD-CG 212 Cl, milrinone and other phosphodiesterase inhibitors to phosphodiesterase III inhibition in guinea-pig heart. Naunyn Schmiedebergs Arch Pharmacol 1989; 339: 575–83.
Salloum FN, Das A, Thomas CS, Yin C, Kukreja RC . Adenosine A1 receptor mediates delayed cardioprotective effect of sildenafil in mouse. J Mol Cell Cardiol 2007; 43: 545–51.
Acknowledgements
This work was supported by the American Heart Association National Center (grant No 0530157N).
We are grateful to Prof Rakesh C KUKREJA and other colleagues at Virginia Commonwealth University for their inspiration and collaboration in the pioneered studies on cardioprotection by PDE5 inhibitors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, Y., Xi, L. Pivotal effects of phosphodiesterase inhibitors on myocyte contractility and viability in normal and ischemic hearts. Acta Pharmacol Sin 30, 1–24 (2009). https://doi.org/10.1038/aps.2008.1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2008.1
Keywords
This article is cited by
-
The phosphodiesterase 5 inhibitor sildenafil decreases the proinflammatory chemokine IL-8 in diabetic cardiomyopathy: in vivo and in vitro evidence
Journal of Endocrinological Investigation (2019)
-
miR-125a, miR-139 and miR-324 contribute to Urocortin protection against myocardial ischemia-reperfusion injury
Scientific Reports (2017)
-
Molecular inotropy mediated by cardiac miR-based PDE4D/PRKAR1α/phosphoprotein signaling
Scientific Reports (2016)
-
Phosphodiesterase Type 5 Inhibitor Sildenafil Decreases the Proinflammatory Chemokine CXCL10 in Human Cardiomyocytes and in Subjects with Diabetic Cardiomyopathy
Inflammation (2016)
-
Phosphodiesterase type 5 inhibitors: back and forward from cardiac indications
Journal of Endocrinological Investigation (2016)