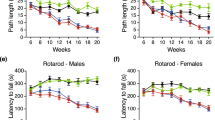
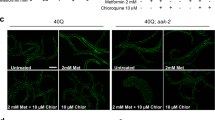
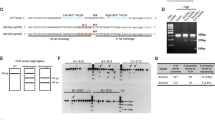
Abstract
Huntington's disease (HD) is an autosomal dominant neurodegenerative disorder, which is caused by an abnormal expansion of Cytosine Adenine Guanine (CAG) trinucleotide repeat in the gene making huntingtin (Htt). Despite intensive research efforts devoted to investigate molecular mechanisms of pathogenesis, effective therapy for this devastating disease is still not available at present. The development of various animal models of HD has offered alternative approaches in the study of HD molecular pathology. Many HD models, including chemical-induced models and genetic models, mimic some aspects of HD symptoms and pathology. To date, however, there is no ideal model which replicates all of the essential features of neuropathology and progressive motor and cognitive impairments of human HD. As a result, our understanding of molecular mechanisms of pathogenesis in HD is still limited. A new model is needed in order to uncover the pathogenesis and to develop novel therapies for HD. In this review we discussed usefulness and limitations of various animal and cellular models of HD in uncovering molecular mechanisms of pathogenesis and developing novel therapies for HD.
Similar content being viewed by others
Article PDF
References
The Huntington's Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell 1993; 72: 971–83.
Brinkman RR, Mezei MM, Theilmann J, Almqvist E, Hayden MR . The likelihood of being affected with Huntington disease by a particular age for a specific CAG size. Am J Hum Genet 1997; 60: 1202–10.
Beal MF . Aging, energy, and oxidation in neurodegenerative diseases. Ann Neurol 1995; 38: 357–66.
Cattaneo E . Dysfunction of wild type huntingtin in Huntingtondisease. J News Physiol Sci 2003; 18: 34–7.
Brouillet E, Condea F, Beal MF, Hantraye P . Replicating Huntington's disease phenotype in experimental animals. Prog Neurobiol 1999; 59: 427–68.
Foroud T, Gray J, Ivashina J, Conneally PM . Differences in duration of Huntington's disease based on age at onset. J Neurol Neurosurg Psychiatry 1999; 66: 52–6.
Leegwater J, Jang Ho K, Cha J . The paradigm of Huntington's disease: therapeutic opportunities in neurodegeneration. Am Soc Exp Neuro Ther 2004; 1: 128–38.
Vonsattel JP, Myers RH, Stevens TJ, Ferrante RJ, Bird ED, Richardson EP . Neuropathological classification of Huntington's disease. J Neuropathol Exp Neurol 1985; 44: 559–77.
Dawbarn D, De Quidt ME, Emson PC . Survival of basal ganglia neuropeptide Y-somatostatin neurones in Huntington's disease. Brain Res 1985; 340: 251–60.
Ferrante RJ, Kowall NW, Beal MF, Richardson EP Jr, Bird ED, Martin JB . Selective sparing of a class of striatal neurons in Huntington's disease. Science 1985; 230: 561–4.
Ferrante RJ, Kowall NW, Beal MF, Martin JB, Bird ED, Richardson EP . Morphologic and histochemical characteristics of a spared subset of striatal neurons in Huntington's disease. J Neuropathol Exp Neurol 1987; 46: 12–27.
Kowall NW, Ferrante RJ, Martin JB . Patterns of cell loss in Huntington's disease. Trends Neurosci 1987; 10: 24–9.
Cicchetti F, Parent A . Striatal interneurons in Huntington's disease: selective increase in the density of calretinin-immunoreactive medium-sized neurons. Mov Disord 1996; 11: 619–26.
Hedreen JC, Folstein SE . Early loss of neostriatal neurons in Huntington's disease. J Neuropathol Exp Neurol 1995; 54: 105–20.
Martin JB . Molecular basis of the neurodegenerative disorders. N Engl J Med 1999; 340: 1970–80.
Ho LW, Carmichael J, Swartz J, Wyttenbach A, Rankin J, Rubinsztein DC . The molecular biology of Huntington's disease. Pyschol Med 2001; 31: 3–14.
Tobin AJ, Signer ER . Huntington's disease: the challenge for cellbiologists. Trends Cell Biol 2000; 10: 531–6.
Cudkowicz M, Kowall NW . Degeneration of pyramidal projection neurons in Huntington's disease cortex. Ann Neurol 1990; 27: 200–4.
Aronin N, Chase K, Young C, Sapp E, Schwarz C, Matta N, et al. CAG expansion affects the expression of mutant huntingtin in the Huntington's disease brain. Neuron 1995; 15: 1193–201.
Martindale D, Hackam A, Wieczorek A, Ellerby L, Wellington C, McCutcheon K, et al. Length of huntingtin and its polyglutamine tract influences localization and frequency of intracellular aggregates. Nat Genet 1998; 18: 150–4.
Kegel KB, Meloni AR, Yi Y, Kim KY, Doyle E, Cuiffo BG, et al. Huntingtin is present in the nucleus, interacts with the transcriptional corepressor C-terminal binding protein, and represses transcription. J Biol Chem 2002; 277: 7466–76.
Sugars KL, Rubinsztein DC . Transcriptional abnormalities in Huntington disease. Trends Genet 2003; 19: 233–8.
DiFiglia M, Sapp E, Chase K, Schwarz C, Meloni A, Young C, et al. Huntingtin is a cytoplasmic protein associated with vesiclesin human and rat brain neurons. Neuron 1995; 14: 1075–81.
White JK, Auerbach W, Duyao MP, Vonsattel JP, Gusella JF, Joyner AL, et al. Huntingtin is required for neurogenesis and is not impaired by the Huntington's disease CAG expansion. Nat Genet 1997; 17: 404–10.
Goldberg YP, Nicholson DW, Rasper DM, Kalchman MA, Koide HB, Graham RK, et al. Cleavage of huntingtin by apopain, aproapoptotic cysteine protease, is modulated by the polyglutamine tract. Nat Genet 1996; 13: 442–9.
Wellington CL, Ellerby LM, Hackam AS, Margolis RL, Trifiro MA, Singaraja R, et al. Caspase cleavage of gene products associated with triplet expansion disorders generates truncated fragments containing the polyglutamine tract. J Biol Chem 1998; 273: 9158–67.
Goffredo D, Rigamonti D, Tartari M, De Micheli A, Verderio C, Matteoli M, et al. Calcium-dependent cleavage of endogenous wild-type huntingtin in primary cortical neurons. J Biol Chem 2002; 277: 39 594–8.
Gafni J, Ellerby LM . Calpain activation in Huntington's disease. J Neurosci 2002; 22: 4842–9.
Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 1997; 90: 537–48.
DiFiglia M, Sapp E, Chase O, Davies SW, Bates GP, Vonsattel JP, et al. Aggregation of Huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 1997; 277: 1990–3.
Scherzinger E, Lurz R, Turmaine M, Maggiarini L, Hollenbach B, Hasenbank R, et al. Huntingtin-encoded polyglutamine expansions form amyloid-like protein aggregates in vitro and in vivo. Cell 1997; 90: 549–58.
Qin ZH, Gu ZL . Huntingtin processing in pathogenesis of Huntington disease. Acta Pharmacol Sin 2004; 25: 1243–9.
Ravikumar B, Duden R, Rubinsztein DC . Aggregate-prone proteins with polyglutamine and polyalanine expansions are degraded by autophagy. Hum Mol Genet 2002; 11: 1107–17.
McGeer EG, McGeer PL . Duplication of bio-chemical changes of Huntington's chorea by intrastriatal injections of glutamic and kainic acid. Nature 1976; 263: 517–9.
Beal MF, Marshall PE, Burd GD, Landis DMD, Martin JBB . Excitotoxin lesions do not mimic the alteration of somatostatin in Huntington disease. Brain Res 1985; 361: 135–45.
Beal MF, Kowall NW, Ellison DW, Swartz KJ, Mc Garvey U, Bird ED, et al. Replication of the neurochemical characteristics of Huntington's disease by quinolinic acid. Nature 1986; 321: 168–71.
Roberts R, Ahn A, Swartz KJ, Beal MF, DiFiglia M . Intrastriatal injections of quinolinic acid or kainic acid: differential patterns of cell survival and effects of data analysis on outcome. Exp Neurol 1993; 124: 274–82.
Qin ZH, Wang YM, Chase TN . Stimulation of N-methyl-D-aspartate receptors induces apoptosis in rat brain. Brain Res 1996; 725: 166–76.
Qin ZH, Wang YM, Nakai M, Chase TN . Nuclear factor-kB contributes to excitotoxin-induced apoptosis in rat striatum. Mol Pharmacol 1998; 53: 33–42.
Liang ZQ, Wang XX, Wang YM, Chuang DW, DiFiglia M, Thomas N, et al. Susceptibility of striatal neurons to excitotoxic injury correlates with basal levels of Bcl-2 and the induction of P53 and c-Myc immunoreactivity. Neurobiol Disease 2005; 20: 562–73.
Sanberg PR, Coyle JT . Scientific approaches to Huntington's disease. Crit Rev Clin Neurobiol 1984; 1: 1–44.
Qin ZH, Wang J, Gu ZL . Development of novel therapies for Huntington's disease: hope and challenge. Acta Pharmacol Sin 2005; 26: 129–42.
Ludolph AC, He F, Spencer PS, Hammerstad J, Sabri M . 3–Nitroproprionic acid-exogenous animal neurotoxin and possible human striatal toxin. Can J Neurol Sci 1991; 18: 492–8.
Beal MF, Kowall NW, Ellison DW, Swartz KJ, Mc Garvey U, Bird ED, et al. Replication of the neurochemical characteristics of Huntington's disease by quinolinic acid. Nature 1986; 321: 168–71.
Hamilton BF, Gould DH . Nature and distribution of brain lesionsin rats intoxicated with 3-nitropropionic acid: a type of hypoxic(energy deficient) brain damage. Acta Neuropathol 1987; 72: 286–97.
Guyot MC, Hantraye P, Dolan R, Palfi S, Maziere M, Brouillet E . Quantifiable bradykinesia, gait abnormalities and Huntington's disease-like striatal lesions in rats chronically treated with 3-nitropropionic acid. Neuroscience 1997; 79: 45–56.
Beal MF, Brouillet E, Jenkins B, Ferrante RJ, Kowall NW, Miller JM, et al. Neurochemical and histological characterization of the striatal excitotoxic lesions produced by the mitochondrialtoxins 3-nitropropionic acid. J Neurosci 1993; 13: 1481–92.
Brouillet E, Guyot MC, Mittoux V, Altairac S, Conde F, Palfi S, et al. Partial inhibition of brain succinate deshydrogenase by 3-nitropropionic acid is sufficient to initiate striatal degenerationin rat. J Neurochem 1998; 70: 794–805.
Borlongan CV, Koutousis TK, Freeman TB, Cahill DW, Sanberg PR . Behavioral pathology induced by repreated systemic injections of 3-nitropropionic acid mimics the motoric symptoms of Huntington's disease. Brain Res 1995; 697: 254–7.
Borlongan CV, Koutousis TK, Randall TS, Freeman TB, Cahill DW, Sanberg PR . Systemic 3-nitropropionic acid: behavioral deficits and striatal damage in adult rats. Brain Res Bull 1995; 36: 549–56.
Brouillet E, Jenkins B, Hyman B, Ferrante RJ, Kowall NW, Srivastava R, et al. Age-dependent vulnerability of the striatum to the mitochondrial toxin 3-nitropropionic acid. J Neurochem 1993; 60: 356–9.
Borlongan CV, Koutouzis TK, Sanberg PR . 3-Nitropropionic acid animal model and Huntington's disease. Neurosci Biobehav Rev 1997; 21: 289–93.
Mettler FA . Choreoathetosis and striopallidonigral necrosis due to sodium azide. Exp Neurol 1972; 34: 291–308.
Borlongan CV, Koutouzis TK, Cahill DW, Freeman TB, Sanberg PR . Behavioral pathology induced by repeated systemic injections of 3-nitropropionic acid mimics the motoric symptoms of Huntington's disease. Brain Res 1995; 697: 254–7.
Stober T, Wussow W, Schimrigk K . Bicaudate diameter – the most specific and simplified CT parameter in the diagnosis of Huntington's disease. Neuroradiology 1984; 26: 25–8.
Hantraye P, Riche D, Maziere M, Isacson O . An experimental primate model of Huntington's disease: anatomical and behavioural studies of unilateral excitotoxic lesions of the caudate-putamen in the baboon. Exp Neurol 1990; 108: 91–104.
Leavitt BR, Raamsdonk JM, Shehadeh J, Fernandes H, Murphy Z, Graham RK, et al. Wild-type huntingtin protects neurons from excitotoxicity. J Neurochem 2006; 96: 1121–9.
Hickey MA, Chesselet MF . The use of transgenic and knock-in mice to study Huntington's disease. Cytogenet Genome Res 2003; 100: 276–86.
Sathasivam K, Hobbs C, Mangiarini L, Mahal A, Turmaine M, Doherty P, et al. Transgenic models of Huntington's disease. Phil Trans R Soc Lond B 1999; 354: 963–9.
Mangiarini L, Sathasivam K, Seller M, Cozens B, Harper A, Hetherington C, et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 1996; 87: 493–506.
Hurlbert MS, Zhou W, Wasmeier C, Kaddis FG, Hutton JC, Freed CR . Mice transgenic for an expanded CAG repeat in the Huntington's disease gene develop diabetes. Diabetes 1999; 48: 649–51.
Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 1997; 90: 537–48.
Turmaine M, Raza A, Mahal A, Mangiarini L, Bates GP, Davies SW . Nonapoptotic neurodegeneration in a transgenic mousemodel of Huntington's disease. Proc Natl Acad Sci USA 2000; 97: 8093–7.
Hersch SM, Ferrante RJ . Translating therapies for Huntington's disease from genetic animal models to clinical trials. NeuroRx 2004; 3: 298–603.
Ross CA . Intranuclear neuronal inclusions: a common pathogenic mechanism for glutamine-repeat neurodegenerative diseases?. Neuron 1997; 19: 1147–50.
DiFiglia M, Sapp E, Chase KO, Davies SW, Bates GP, Vonsattel JP, et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 1997; 277: 1990–3.
Becher MW, Kotzuk JA, Sharp AH, Davies SW, Bates GP, Price DL, et al. Intranuclear neuronal inclusions in Huntington's disease and dentatorubral and pallidoluysian atrophy: correlation between the density of inclusions and IT15 CAG triplet repeatlength. Neurobiol Dis 1998; 4: 387–97.
Gourfinkel-An I, Cancel G, Faucheux B, Hauw JJ, Trottier Y, et al. Neuronal distribution of intranuclear inclusions in Huntington's disease with adult onset. Neuro Report 1998; 9: 1823–6.
Goldberg YP, Kalchman MA, Metzler M, Nasir J, Zeisler J, Graham R, et al. Absence of disease phenotype and intergenerational stability of the CAG repeat in transgenic mice expressing the human Huntington disease transcript. Hum Mol Genet 1996; 5: 177–85.
Reddy PH, Williams M, Charles V, Garrett L, Pike-Buchanan L, Whetsell WO Jr, et al. Behavioural abnormalities and selective neuronal loss in HD transgenic mice expressing mutated full-length HD cDNA. Nat Genet 1998; 20: 198–202.
Hodgson JG, Agopyan N, Gutekunst CA, Leavitt BR, LePiane F, Singaraja R, et al. A YAC mouse model for Huntington's disease with full-length mutant huntingtin, cytoplasmic toxicity, and selective striatal neurodegeneration. Neuron 1999; 23: 181–92.
Jeremy M, Raamsdonk V, Murphy Z, Slow EJ, Leavitt BR, Hayden MR . Selective degeneration and nuclear localization of mutant huntingtin in the YAC128 mouse model of Huntington disease. Hum Mol Genet 2005; 24: 3823–35.
Tanaka Y, Igarashi S, Nakamura M, Gafni J, Torcassi C, Schilling G, et al. Progressive phenotype and nuclear accumulation of anamino-terminal cleavage fragment in a transgenic mouse model with inducible expression of full-length mutant huntingtin. Neurobiol 2005; 14: 1–11.
Shelbourne PF, Killeen N, Hevner RF, Johnston HM, Tecott L, Lewandoski M, et al. A Huntington's disease CAG expansion at the murine Hdh locus is unstable and associated with behavioural abnormalities in mice. Hum Mol Genet 1999; 8: 763–74.
Lin CH, Tallaksen-Greene S, Chien WM, Cearley JA, Jackson WS, Crouse AB, et al. Neurological abnormalities in a knock-in mouse model of Huntington's disease. Hum Mol Genet 2001; 10: 137–44.
Menalled LB, Sison JD, Wu Y, Olivieri M, Li XJ, Li H, et al. Early motor dysfunction and striosomal distribution of huntingtin microaggregates in Huntington's disease knock-in mice. J Neurosci 2002; 22: 8266–76.
Kirkwood SC, Su JL, Conneally P, Foroud T . Progression of symptoms in the early and middle stages of Huntington disease. Arch Neurol 2001; 58: 273–8.
Menalled LB, Chesselet MF . Mouse models of Huntington's disease. Trends Pharmacol Sci 2002; 23: 32–9.
Kennedy L, Shelbourne PF . Dramatic mutation instability in HD mouse striatum: does polyglutamine load contribute to cell-specific vulnerability in Huntington's disease?. Hum Mol Genet 2000; 9: 2539–44.
Duyao MP, Auerbach AB, Ryan A, Persichetti F, Barnes GT, McNeil SM, et al. Inactivation of the mouse Huntington's disease gene homolog Hdh. Science 1995; 269: 407–10.
Nasir J, Floresco SB, O'Kusky JR, Diewert VM, Richman JM, Zeisler J, et al. Targeted disruption of the Huntington's diseasegene results in embryonic lethality and behavioral and morphological changes in heterozygotes. Cell 1995; 81: 811–23.
Zeitlin S, Liu JP, Chapman DL, Papaioannou VE, Efstratiadis A . Increased apoptosis and early embryonic lethality in micenullizygous for the Huntington's disease gene homologue. Nat Genet 1995; 11: 155–63.
White JK, Auerbach W, Duyao MP, Vonsattel JP, Gusella JF, Joyner AL, et al. Huntingtin is required for neurogenesis and is not impaired by the Huntington's disease CAG expansion. Nat Genet 1997; 17: 404–10.
Clabough EB, Zeitlin SO . Deletion of the triplet repeat encoding polyglutamine within the mouse Huntington's disease gene results in subtle behavioral/motor phenotypes in vivo and elevated levels of ATP with cellular senescence in vitro. Hum Mol Genet 2006; 15: 607–23.
Senut MC, Suhr ST, Kaspar B, Gage FH . Intraneuronal aggregate formation and cell death after viral expression of expanded polyglutamine tracts in the adult rat brain. J Neurosci 2000; 20: 219–29.
Morley JF, Brignull HR, Weyers JJ, Morimoto RI . The threshold for polyglutamine-expansion protein aggregation and cellular toxicity is dynamic and influenced by aging in Caenorhabditis elegans. Proc Natl Acad Sci USA 2002; 99: 10417–22.
de Almeida LP, Ross CA, Zala D, Aebischer P, Deglon N . Lentiviral-mediated delivery of mutant huntingtin in the striatum of rats induces a selective neuropathology modulated by polyglutamine repeat size huntingtin expression levels and protein length. J Neurosci 2002; 22: 3473–83.
Rubin GM, Yandell MD, Wortman JR, Gabor Miklos GL, Nelson CR, et al. Comparative genomics of the eukaryotes. Science 2000; 287: 2204–15.
Reiter LT, Potocki L, Chien S, Gribskov M, Bier E . A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res 2001; 11: 1114–25.
Li Z, Karlovich CA, Fish MP, Scott MP, Myers RM . A putativedrosophila homolog of the Huntington's disease gene. Hum Mol Genet 1999; 8: 1807–15.
Marsh JL, Pallos J, Thompson LM . Fly models of Huntington's disease. Hum Mol Genet 2003; 12: R187–R193.
Brand AH, Perrimon N . Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 1993; 118: 401–15.
Brand AH, Dormand EL . The GAL4 system as a tool for unravelling the mysteries of the Drosophila nervous system. Curr Opin Neurobiol 1995; 5: 572–8.
Steffan JS, Bodai L, Pallos J, Poelman M, McCampbell A, Apostol BL, et al. Histone deacetylase inhibitors arrest polyglutamine-dependent neurodegeneration in Drosophila. Nature 2001; 413: 739–43.
Jackson GR, Salecker I, Dong X, Yao X, Arnheim N, Faber PW, et al. Polyglutamine-expanded human huntingtin transgenes induce degeneration of Drosophila Bonini, NM. Suppression of polyglutamine-mediated neurodegene-photoreceptor neurons. Neuron 1999; 21: 633–42.
Parker JA, Connolly JB, Wellington C, Hayden M, Dausset J, Neri C . Expanded polyglutamines in Caenorhabditis elegans cause axonal abnormalities and severe dysfunction of PLM mechano-sensory neurons without cell death. Proc Natl Acad Sci USA 2001; 98: 13 318–23.
Chalfie M . The mec-3 gene of Caenorhabditis elegans requiresits own product for maintained expression and is expressed in three neuronal cell types. Genes Dev 1989; 3: 1823–33.
The C elegans Sequencing Consortium. Genome sequence of the nematode C elegans: a platform for investigating biology. Science 1998; 282: 2012–8.
Faber PW, Alter JR, MacDonald ME, Hart AC . Polyglutamine-mediated dysfunction and apoptotic death of a Caenorhabditis elegans sensory neuron. Proc Natl Acad Sci USA 1999; 96: 179–84.
Marsh JL, Walker H, Theisen H, Zhu YZ, Fielder T, Purcell J, et al. Expanded polyglutamine peptides alone are intrinsicallycytotoxic and cause neurodegeneration in Drosophila. Hum Mol Genet 2000; 9: 13–25.
Moore H, Greenwell PW, Liu CP, Arnheim N, Petes TD . Triplet repeats form secondary structures that escape DNA repair in yeast. Proc Natl Acad Sci USA 1999; 96: 1504–9.
Schweitzer JK, Livingston DM . Destabilization of CAG tri-nucleotide repeat tracts by mismatch repair mutations in yeast. Hum Mol Genet 1997; 6: 349–55.
Kazantsev A, Preisinger E, Dranovsky A, Goldgaber D, Housman D . Insoluble detergent-resistant aggregates form between pathological and nonpathological lengths of polyglutamine in mammalian cells. Proc Natl Acad Sci USA 1999; 96: 11404–9.
Krobitsch S, Lindquist S . Aggregation of huntingtin in yeastvaries with the length of the polyglutamine expansion and theexpression of chaperone proteins. Proc Natl Acad Sci USA 2000; 97: 1589–94.
Burke JR, Enghild JJ, Martin ME, Jou YS, Myers RM, Roses AD, et al. Huntingtin and DRPLA proteins selectively interact with the enzyme GAPDH. Nat Med 1996; 2: 347–50.
Li SH, Hosseini SH, Gutekunst CA, Hersch SM, Ferrante RJ, Li XJ . A human HAP1 homologue. Cloning, expression, and interaction with huntingtin. J Biol Chem 1998; 273: 19 220–7.
Kim M, Lee HS, LaForet G, McIntyre C, Martin EJ, Chang P, et al. Mutant huntingtin expression in clonal striatal cells: dissociation of inclusion formation and neuronal survival by caspase inhibition. J Neurosci 1999; 19: 964–73.
Rigamonti D, Bauer JH, De-Fraja C, Conti L, Sipione S, Sciorati C, et al. Wild-type huntingtin protects from apoptosis upstream of caspase-3. J Neurosci 2000; 20: 3705–13.
Sipione S, Rigamonti D, Valenza M, Zuccato C, Conti L, Pritchard J, et al. Early transcriptional profiles in huntingtin-inducible striatal cells by microarray analyses. Hum Mol Genet 2002; 17: 1953–65.
Wyttenbach A, Swartz J, Kita H, Thykjaer T, Carmichael J, Bradley J, et al. Polyglutamine expansions cause decreased CRE-mediated transcription and early gene expression changesprior to cell death in an inducible cell model of Huntington's disease. Hum Mol Genet 2001; 10: 1829–45.
Qin ZH, Wang Y, Kegel KB, Kazantsev A, Apostol BL, Thompson LM, et al. Autophagy regulates the processing of aminoterminal huntingtin fragments. Hum Mol Genet 2003; 24: 3231–44.
Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, et al. Inhibition of mTOR induces autophagy and reduces toxicityof polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 2004; 36: 585–95.
Hersch SM, Ferrante RJ . Translating therapies for Huntington's disease from genetic animal models to clinical trials. Neuro Rx 2004; 1: 298–306.
Young AB . Huntingtin in health and disease. J Clin Invest 2003; 111: 299–302.
Sittler A, Lurz R, Lueder G, Priller J, Lehrach H, Hayer-Hartl MK, et al. Geldanamycin activates a heat shock response and inhibits huntingtin aggregation in a cell culture model of Huntington's disease. Hum Mol Genet 2001; 10: 1307–15.
Heemskerk J, Tobin AJ, Bain LJ . Teaching old drugs new tricks. Meeting of the neurodegeneration drug screening consortium. Trends Neurosci 2002; 25: 494–6.
Heiser V, Scherzinger E, Boeddrich A, Nordhoff E, Lurz R . Schugardt N, et al. Inhibition of huntingtin fibrillogenesis by specific antibodies and small molecules: implications for Huntington's disease therapy. Proc Natl Acad Sci USA 2000; 97: 6739–44.
Apostol BL, Kazantsev A, Raffioni S, Illes K, Pallos J, Bodai L, et al. A cell-based assay for aggregation inhibitors as therapeutics of polyglutamine-repeat disease and validation in Drosophila. Proc Natl Acad Sci USA 2003; 100: 5950–5.
Hickey MA, Gallant K, Gross GG, Levine MS, Chesselet MF . Early behavioral deficits in R6/2 mice suitable for use in pre-clinical drug testing. Neurobiol Dis 2005; 20: 1–11.
Kazantsev A, Walker H, Slepko N, Bear JE, Preisinger E, Steffan JS, et al. A bivalent Huntingtin binding peptide suppresses polyglutamine aggregation and pathogenesis in Drosophila. NatGenet 2002; 30: 367–76.
Apostol BL, Kazantsev A, Raffioni S, Illes K, Pallos J, Bodai L, et al. A cell-based assay for aggregation inhibitors as therapeutics of polyglutamine-repeat disease and validation in Drosophila. Proc Natl Acad Sci USA 2003; 100: 5950–5.
Dedeoglu A, Kubilus JK, Jeitner TM, Matson SA, Bogdanov M, Kowall NW, et al. Therapeutic effects of cystamine in a murine model of Huntington's disease. J Neurosci 2002; 22: 8942–50.
Karpuj MV, Becher MW, Springer JE, Chabas D, Youssef S, Pedotti R, et al. Prolonged survival and decreased abnormal movements in transgenic model of Huntington disease, with administration of the transglutaminase inhibitor cystamine. Nat Med 2002; 8: 143–9.
Sanchez I, Mahlke C, Yuan J . Pivotal role of oligomerizationin expanded polyglutamine neurodegenerative disorders. Nature 2003; 421: 373–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30370506) and by the Specialized Research Fund for the Doctoral Program of Higher Education (No 20050285017).
Rights and permissions
About this article
Cite this article
Wang, Lh., Qin, Zh. Animal models of Huntington's disease: implications in uncovering pathogenic mechanisms and developing therapies. Acta Pharmacol Sin 27, 1287–1302 (2006). https://doi.org/10.1111/j.1745-7254.2006.00410.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00410.x
Keywords
This article is cited by
-
An Overview of the Pathophysiological Mechanisms of 3-Nitropropionic Acid (3-NPA) as a Neurotoxin in a Huntington's Disease Model and Its Relevance to Drug Discovery and Development
Neurochemical Research (2023)
-
Mouse models of neurodegenerative disease: preclinical imaging and neurovascular component
Brain Imaging and Behavior (2018)
-
Animal models for metabolic, neuromuscular and ophthalmological rare diseases
Nature Reviews Drug Discovery (2013)
-
Modeling mitochondrial dysfunctions in the brain: from mice to men
Journal of Inherited Metabolic Disease (2012)