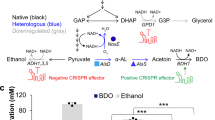
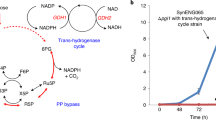
Abstract
Metabolism is a highly interconnected web of chemical reactions that power life. Though the stoichiometry of metabolism is well understood, the multidimensional aspects of metabolic regulation in time and space remain difficult to define, model and engineer. Complex metabolic conversions can be performed by multiple species working cooperatively and exchanging metabolites via structured networks of organisms and resources. Within cells, metabolism is spatially regulated via sequestration in subcellular compartments and through the assembly of multienzyme complexes. Metabolic engineering and synthetic biology have had success in engineering metabolism in the first and second dimensions, designing linear metabolic pathways and channeling metabolic flux. More recently, engineering of the third dimension has improved output of engineered pathways through isolation and organization of multicell and multienzyme complexes. This review highlights natural and synthetic examples of three-dimensional metabolism both inter- and intracellularly, offering tools and perspectives for biological design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gijzen, H.J. & Barugahare, M. Contribution of anaerobic protozoa and methanogens to hindgut metabolic activities of the American cockroach, Periplaneta americana. Appl. Environ. Microbiol. 58, 2565–2570 (1992).
van Hoek, A.H. et al. Multiple acquisition of methanogenic archaeal symbionts by anaerobic ciliates. Mol. Biol. Evol. 17, 251–258 (2000).
Akhmanova, A. et al. A hydrogenosome with a genome. Nature 396, 527–528 (1998).
Ushida, K. Symbiotic methanogens and rumen ciliates. in (Endo)symbiotic Methanogenic Archaea (ed. Hackstein, J.H.P.) 25–34 (Springer, 2011).
Boxma, B. et al. An anaerobic mitochondrion that produces hydrogen. Nature 434, 74–79 (2005).
Yeates, T.O., Crowley, C.S. & Tanaka, S. Bacterial microcompartment organelles: protein shell structure and evolution. Annu. Rev. Biophys. 9, 185–205 (2010).
MacLeod, F.A., Guiot, S. & Costerton, J. Layered structure of bacterial aggregates produced in an upflow anaerobic sludge bed and filter reactor. Appl. Environ. Microbiol. 56, 1598–1607 (1990).
Dueber, J.E. et al. Synthetic protein scaffolds provide modular control over metabolic flux. Nat. Biotechnol. 27, 753–759 (2009).
Lee, H., DeLoache, W.C. & Dueber, J.E. Spatial organization of enzymes for metabolic engineering. Metab. Eng. 14, 242–251 (2012).
Solomon, K.V. & Prather, K.L.J. The zero-sum game of pathway optimization: Emerging paradigms for tuning gene expression. Biotechnol. J. 6, 1064–1070 (2011).
Brenner, K., You, L. & Arnold, F. Engineering microbial consortia: a new frontier in synthetic biology. Trends Biotechnol. 26, 483–489 (2008).
Zuroff, T.R. & Curtis, W.R. Developing symbiotic consortia for lignocellulosic biofuel production. Appl. Microbiol. Biotechnol. 93, 1423–1435 (2012).
Shin, H.-D., McClendon, S., Vo, T. & Chen, R.R. Escherichia coli binary culture engineered for direct fermentation of hemicellulose to a biofuel. Appl. Environ. Microbiol. 76, 8150–8159 (2010).
Tsai, S.L., Oh, J., Singh, S., Chen, R. & Chen, W. Functional assembly of minicellulosomes on the Saccharomyces cerevisiae cell surface for cellulose hydrolysis and ethanol production. Appl. Environ. Microbiol. 75, 6087–6093 (2009).
Watanabe, K. Understanding substrate specificity of polyketide synthase modules by generating hybrid multimodular synthases. J. Biol. Chem. 278, 42020–42026 (2003).
Weeks, A., Lund, L. & Raushel, F.M. Tunneling of intermediates in enzyme-catalyzed reactions. Curr. Opin. Chem. Biol. 10, 465–472 (2006).
Bonacci, W. et al. Modularity of a carbon-fixing protein organelle. Proc. Natl. Acad. Sci. USA 109, 478–483 (2012).
Staunton, J. & Weissman, K.J. Polyketide biosynthesis: a millennium review. Nat. Prod. Rep. 18, 380–416 (2001).
Thattai, M., Burak, Y. & Shraiman, B.I. The origins of specificity in polyketide synthase protein interactions. PLoS Comput. Biol. 3, 1827–1835 (2007).
Kim, H.J., Du, W. & Ismagilov, R.F. Complex function by design using spatially pre-structured synthetic microbial communities: degradation of pentachlorophenol in the presence of Hg(II). Integr. Biol. (Camb.) 3, 126–133 (2011).
Goyal, G., Tsai, S.-L., Madan, B., DaSilva, N.A. & Chen, W. Simultaneous cell growth and ethanol production from cellulose by an engineered yeast consortium displaying a functional mini-cellulosome. Microb. Cell Fact. 10, 89 (2011).
Vogels, G.D., Hoppe, W.F. & Stumm, C.K. Association of methanogenic bacteria with rumen ciliates. Appl. Environ. Microbiol. 40, 608–612 (1980).
Lee, M.J., Schreurs, P.J., Messer, A.C. & Zinder, S.H. Association of methanogenic bacteria with flagellated protozoa from a termite hindgut. Curr. Microbiol. 15, 337–341 (1987).
Gonzalez-Gil, G. et al. Cluster structure of anaerobic aggregates of an expanded granular sludge bed reactor. Appl. Environ. Microbiol. 67, 3683–3692 (2001).
Liu, W.T., Chan, O.C. & Fang, H.H.P. Characterization of microbial community in granular sludge treating brewery wastewater. Water Res. 36, 1767–1775 (2002).
Sekiguchi, Y., Kamagata, Y., Nakamura, K., Ohashi, A. & Harada, H. Fluorescence in situ hybridization using 16S rRNA-targeted oligonucleotides reveals localization of methanogens and selected uncultured bacteria in mesophilic and thermophilic sludge granules. Appl. Environ. Microbiol. 65, 1280–1288 (1999).
Boone, D.R. & Bryant, M.P. Propionate-degrading bacterium, Syntrophobacter wolinii sp. nov. gen. nov., from methanogenic ecosystems. Appl. Environ. Microbiol. 40, 626–632 (1980).
Dwyer, D.F., Weeg-Aerssens, E., Shelton, D.R. & Tiedje, J.M. Bioenergetic conditions of butyrate metabolism by a syntrophic, anaerobic bacterium in coculture with hydrogen-oxidizing methanogenic and sulfidogenic bacteria. Appl. Environ. Microbiol. 54, 1354–1359 (1988).
Raven, J.A., Cockell, C.S. & De La Rocha, C.L. The evolution of inorganic carbon concentrating mechanisms in photosynthesis. Philos. Trans. R. Lond. B. Biol. Sci. 363, 2641–2650 (2008).
Kumar, K., Mella-Herrera, R.A. & Golden, J.W. Cyanobacterial heterocysts. Cold Spring Harb. Perspect. Biol. 2, a000315 (2010).
Meeks, J.C. & Elhai, J. Regulation of cellular differentiation in filamentous Cyanobacteria in free-living and plant-associated symbiotic growth states. Microbiol. Mol. Biol. Rev. 66, 94–121 (2002).
Stevenson, B.S. et al. Hoeflea anabaenae sp. nov., an epiphytic symbiont that attaches to the heterocysts of a strain of Anabaena. Int. J. Syst. Evol. Microbiol. 61, 2439–2444 (2011).
Phelan, V.V., Liu, W.-T., Pogliano, K. & Dorrestein, P.C. Microbial metabolic exchange—the chemotype-to-phenotype link. Nat. Chem. Biol. 8, 26–35 (2011).
Basu, S., Gerchman, Y., Collins, C.H., Arnold, F.H. & Weiss, R. A synthetic multicellular system for programmed pattern formation. Nature 434, 1130–1134 (2005).
You, L., Cox, R.S., Weiss, R. & Arnold, F.H. Programmed population control by cell-cell communication and regulated killing. Nature 428, 868–871 (2004).
Markx, G.H., Andrews, J.S. & Mason, V.P. Towards microbial tissue engineering? Trends Biotechnol. 22, 417–422 (2004).
Lanthier, M., Tartakovsky, B., Villemur, R., DeLuca, G. & Guiot, S.R. Microstructure of anaerobic granules bioaugmented with Desulfitobacterium frappieri PCP-1. Appl. Environ. Microbiol. 68, 4035–4043 (2002).
Summers, Z.M. et al. Direct exchange of electrons within aggregates of an evolved syntrophic coculture of anaerobic bacteria. Science 330, 1413–1415 (2010).
Shapiro, J.A. Thinking about bacterial populations as multicellular organisms. Annu. Rev. Microbiol. 52, 81–104 (1998).
Shou, W., Ram, S. & Vilar, J.M.G. Synthetic cooperation in engineered yeast populations. Proc. Natl. Acad. Sci. USA 104, 1877–1882 (2007).
Wintermute, E.H. & Silver, P.A. Emergent cooperation in microbial metabolism. Mol. Syst. Biol. 6, 407 (2010).
Kim, H.J., Boedicker, J.Q., Choi, J.W. & Ismagilov, R.F. Defined spatial structure stabilizes a synthetic multispecies bacterial community. Proc. Natl. Acad. Sci. USA 105, 18188–18193 (2008).
Kerr, B., Riley, M. & Feldman, M. Local dispersal promotes biodiversity in a real-life game of rock-paper-scissors. Nature 418, 171–174 (2002).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Tvede, M. & Rask-Madsen, J. Bacteriotherapy for chronic relapsing Clostridium difficile diarrhoea in six patients. Lancet 1, 1156–1160 (1989).
Shong, J., Jimenez Diaz, M.R. & Collins, C.H. Towards synthetic microbial consortia for bioprocessing. Curr. Opin. Biotechnol. published online, doi:10.1016/j.copbio.2012.02.001 (1 March 2012).
Riesenfeld, C.S., Schloss, P.D. & Handelsman, J. Metagenomics: genomic analysis of microbial communities. Annu. Rev. Genet. 38, 525–552 (2004).
Daniel, R. The metagenomics of soil. Nat. Rev. Microbiol. 3, 470–478 (2005).
Turnbaugh, P.J. et al. The human microbiome project. Nature 449, 804–810 (2007).
Choudhary, S. & Schmidt-Dannert, C. Applications of quorum sensing in biotechnology. Appl. Microbiol. Biotechnol. 86, 1267–1279 (2010).
Aldaye, F.A., Senapedis, W.T., Silver, P.A. & Way, J.C. A structurally tunable DNA-based extracellular matrix. J. Am. Chem. Soc. 132, 14727–14729 (2010).
Embley, T.M. & Martin, W. Eukaryotic evolution, changes and challenges. Nature 440, 623–630 (2006).
Howe, C.J., Barbrook, A.C., Nisbet, R.E.R., Lockhart, P.J. & Larkum, A.W.D. The origin of plastids. Phil. Trans. R. Soc. Lond. B 363, 2675–2685 (2008).
van der Klei, I.J., Yurimoto, H., Sakai, Y. & Veenhuis, M. The significance of peroxisomes in methanol metabolism in methylotrophic yeast. Biochim. Biophys. Acta 1763, 1453–1462 (2006).
Yurimoto, H., Oku, M. & Sakai, Y. Yeast methylotrophy: metabolism, gene regulation and peroxisome homeostasis. Int. J. Microbiol. 2011, 101298 (2011).
van der Klei, I.J., Harder, W. & Veenhuis, M. Biosynthesis and assembly of alcohol oxidase, a peroxisomal matrix protein in methylotrophic yeasts: a review. Yeast 7, 195–209 (1991).
Ozimek, P., Veenhuis, M. & van der Klei, I.J. Alcohol oxidase: a complex peroxisomal, oligomeric flavoprotein. FEMS Yeast Res. 5, 975–983 (2005).
Vonck, J. & van Bruggen, E.F. Architecture of peroxisomal alcohol oxidase crystals from the methylotrophic yeast Hansenula polymorpha as deduced by electron microscopy. J. Bacteriol. 174, 5391–5399 (1992).
Roggenkamp, R. Targeting signals for protein import into peroxisomes. Cell Biochem. Funct. 10, 193–199 (1992).
Bayer, T.S. et al. Synthesis of methyl halides from biomass using engineered microbes. J. Am. Chem. Soc. 131, 6508–6515 (2009).
Lin, J.-P. et al. An effective strategy for the co-production of S-adenosyl-L-methionine and glutathione by fed-batch fermentation. Biochem. Eng. J. 21, 19–25 (2004).
Agapakis, C.M. et al. Towards a synthetic chloroplast. PLoS ONE 6, e18877 (2011).
Savage, D.F., Afonso, B., Chen, A.H. & Silver, P.A. Spatially ordered dynamics of the bacterial carbon fixation machinery. Science 327, 1258–1261 (2010).
Iancu, C.V. et al. The structure of isolated Synechococcus strain WH8102 carboxysomes as revealed by electron cryotomography. J. Mol. Biol. 372, 764–773 (2007).
So, A.K.-C. et al. A novel evolutionary lineage of carbonic anhydrase (e class) is a component of the carboxysome shell. J. Bacteriol. 186, 623–630 (2004).
Price, G.D. & Badger, M.R. Expression of human carbonic anhydrase in the cyanobacterium Synechococcus PCC7942 creates a high CO2-requiring phenotype: evidence for a central role for carboxysomes in the CO2 concentrating mechanism. Plant Physiol. 91, 505–513 (1989).
Klein, M.G. et al. Identification and structural analysis of a novel carboxysome shell protein with implications for metabolite transport. J. Mol. Biol. 392, 319–333 (2009).
Cheng, S. & Bobik, T.A. Characterization of the PduS cobalamin reductase of Salmonella enterica and its role in the Pdu microcompartment. J. Bacteriol. 192, 5071–5080 (2010).
Cheng, S., Liu, Y., Crowley, C.S., Yeates, T.O. & Bobik, T.A. Bacterial microcompartments: their properties and paradoxes. Bioessays 30, 1084–1095 (2008).
Fan, C. et al. Short N-terminal sequences package proteins into bacterial microcompartments. Proc. Natl. Acad. Sci. USA 107, 7509–7514 (2010).
Parsons, J.B. et al. Biochemical and structural insights into bacterial organelle form and biogenesis. J. Biol. Chem. 283, 14366–14375 (2008).
Doblin, M.S., Kurek, I., Jacob-Wilk, D. & Delmer, D.P. Cellulose biosynthesis in plants: from genes to rosettes. Plant Cell Physiol. 43, 1407–1420 (2002).
Ding, S.-Y. & Himmel, M.E. The maize primary cell wall microfibril: a new model derived from direct visualization. J. Agric. Food Chem. 54, 597–606 (2006).
Fontes, C.M.G.A. & Gilbert, H.J. Cellulosomes: highly efficient nanomachines designed to deconstruct plant cell wall complex carbohydrates. Annu. Rev. Biochem. 79, 655–681 (2010).
Fischbach, M. & Voigt, C.A. Prokaryotic gene clusters: a rich toolbox for synthetic biology. Biotechnol. J. 5, 1277–1296 (2010).
Sabathé, F. & Soucaille, P. Characterization of the CipA scaffolding protein and in vivo production of a minicellulosome in Clostridium acetobutylicum. J. Bacteriol. 185, 1092–1096 (2003).
Lilly, M., Fierobe, H.-P., van Zyl, W.H. & Volschenk, H. Heterologous expression of a Clostridium minicellulosome in Saccharomyces cerevisiae. FEMS Yeast Res. 9, 1236–1249 (2009).
Anderson, T.D. et al. Assembly of minicellulosomes on the surface of Bacillus subtilis. Appl. Environ. Microbiol. 77, 4849–4858 (2011).
Moraïs, S. et al. Contribution of a xylan-binding module to the degradation of a complex cellulosic substrate by designer cellulosomes. Appl. Environ. Microbiol. 76, 3787–3796 (2010).
Moraïs, S. et al. Cellulase-xylanase synergy in designer cellulosomes for enhanced degradation of a complex cellulosic substrate. MBio 1, e00285–e00210 (2010).
Smith, S. & Tsai, S.-C. The type I fatty acid and polyketide synthases: a tale of two megasynthases. Nat. Prod. Rep. 24, 1041 (2007).
Weissman, K.J. & Müller, R. Protein-protein interactions in multienzyme megasynthetases. ChemBioChem 9, 826–848 (2008).
McDaniel, R. et al. Multiple genetic modifications of the erythromycin polyketide synthase to produce a library of novel “unnatural” natural products. Proc. Natl. Acad. Sci. USA 96, 1846–1851 (1999).
Menzella, H.G. et al. Combinatorial polyketide biosynthesis by de novo design and rearrangement of modular polyketide synthase genes. Nat. Biotechnol. 23, 1171–1176 (2005).
Tang, L., Fu, H. & McDaniel, R. Formation of functional heterologous complexes using subunits from the picromycin, erythromycin and oleandomycin polyketide synthases. Chem. Biol. 7, 77–84 (2000).
Gokhale, R.S., Tsuji, S.Y., Cane, D.E. & Khosla, C. Dissecting and exploiting intermodular communication in polyketide synthases. Science 284, 482–485 (1999).
Skerker, J.M. et al. Rewiring the specificity of two-component signal transduction systems. Cell 133, 1043–1054 (2008).
Park, S.-H., Zarrinpar, A. & Lim, W.A. Rewiring MAP kinase pathways using alternative scaffold assembly mechanisms. Science 299, 1061–1064 (2003).
Bashor, C.J., Helman, N.C., Yan, S. & Lim, W.A. Using engineered scaffold interactions to reshape MAP kinase pathway signaling dynamics. Science 319, 1539–1543 (2008).
Bhattacharyya, R.P., Reményi, A., Yeh, B.J. & Lim, W.A. Domains, motifs, and scaffolds: the role of modular interactions in the evolution and wiring of cell signaling circuits. Annu. Rev. Biochem. 75, 655–680 (2006).
Page, C.C., Moser, C.C., Chen, X. & Dutton, P.L. Natural engineering principles of electron tunnelling in biological oxidation-reduction. Nature 402, 47–52 (1999).
Bretschger, O. et al. Current production and metal oxide reduction by Shewanella oneidensis MR-1 wild type and mutants. Appl. Environ. Microbiol. 73, 7003–7012 (2007).
Urlacher, V.B. & Eiben, S. Cytochrome P450 monooxygenases: perspectives for synthetic application. Trends Biotechnol. 24, 324–330 (2006).
Shiota, N., Kodama, S., Inui, H. & Ohkawa, H. Expression of human cytochromes P450 1A1 and P450 1A2 as fused enzymes with yeast NADPH-cytochrome P450 oxidoreductase in transgenic tobacco plants. Biosci. Biotechnol. Biochem. 64, 2025–2033 (2000).
Dodhia, V.R., Fantuzzi, A. & Gilardi, G. Engineering human cytochrome P450 enzymes into catalytically self-sufficient chimeras using molecular Lego. J. Biol. Inorg. Chem. 11, 903–916 (2006).
Aliverti, A. & Zanetti, G. A three-domain iron-sulfur flavoprotein obtained through gene fusion of ferredoxin and ferredoxin-NADP+ reductase from spinach leaves. Biochemistry 36, 14771–14777 (1997).
Agapakis, C.M. et al. Insulation of a synthetic hydrogen metabolism circuit in bacteria. J. Biol. Eng. 4, 3 (2010).
Ihara, M. et al. Light-driven hydrogen production by a hybrid complex of a [NiFe]-hydrogenase and the cyanobacterial photosystem I. Photochem. Photobiol. 82, 676–682 (2006).
Lubner, C.E. et al. Solar hydrogen-producing bionanodevice outperforms natural photosynthesis. Proc. Natl. Acad. Sci. USA 108, 20988–20991 (2011).
Delebecque, C.J., Lindner, A.B., Silver, P.A. & Aldaye, F.A. Organization of intracellular reactions with rationally designed RNA assemblies. Science 333, 470–474 (2011).
Conrado, R.J. et al. DNA-guided assembly of biosynthetic pathways promotes improved catalytic efficiency. Nucleic Acids Res. 40, 1879–1889 (2012).
Acknowledgements
The authors would like to thank J. Hackstein, Z. Summers, D. Lovley, D. Savage and B. Afonso for the use of images. P.M.B. is supported by fellowships from the Harvard University Center for the Environment and the US National Science Foundation Synthetic Biology Engineering Research Center. P.A.S. acknowledges support from the Radcliffe Institute of Advanced Study, the Wyss Institute for Biologically Inspired Engineering, the Department of Defense Army Research Office and the Department of Energy Advanced Research Projects Agency-Energy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Agapakis, C., Boyle, P. & Silver, P. Natural strategies for the spatial optimization of metabolism in synthetic biology. Nat Chem Biol 8, 527–535 (2012). https://doi.org/10.1038/nchembio.975
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.975