Abstract
Data sources
Medline, Embase, the Cochrane Central Register of Controlled Trials (CENTRAL), LILACS (Latin American and Caribbean Health Sciences), CNKI (China National Knowledge Infrastructure), Wanfang (China) and the South African Department of Health databases were searched. In addition the archives at the WHO Collaborating Centre for Nutrition and Oral Health at Newcastle University and reference lists of reviews were searched and experts contacted for further relevant papers.
Study selection
Intervention, cohort population or cross sectional studies were considered where there was any intervention to alter the intake of sugar or the intake of sugars or change of sugar intake was monitored. The main outcome was caries incidence. The GRADE (Grading of Recommendations Assessment Development and Evaluation) system was used to assess study quality.
Data extraction and synthesis
Separate data abstraction forms were designed for RCTs and observational studies and meta-analyses were to be conducted where possible.
Results
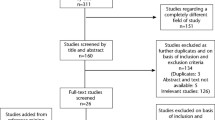
Sixty-five papers reporting 55 studies were included. No RCTs were identified. Three studies (four papers) were interventional, eight (12 papers) were prospective cohort, 20 (25 papers) were population based and 24 (25 papers) were cross-sectional. Data variability limited meta-analysis. Of the studies, 42 out of 50 of those in children and five out of five in adults reported at least one positive association between sugars and caries.
Five of the eight cohort studies allowed comparison of dental caries development when sugars consumption was equivalent to a level < 10% E (Energy) or > 10% E. All eight studies found higher caries with sugars intake > 10% E compared with < 10% E.
Conclusions
This in-depth systematic review shows consistent evidence of moderate quality supporting a relationship between the amount of sugars consumed and dental caries development. There is evidence of moderate quality to show that dental caries is lower when free-sugars intake is < 10% E. Dental caries progresses with age, and the effects of sugars on the dentition are lifelong. Even low levels of caries in childhood are of significance to levels of caries throughout the life course. Analysis of the data suggests that there may be benefit in limiting sugars to < 5% E to minimise the risk of dental caries throughout the life course.
Similar content being viewed by others
Commentary
In 2010, the World Health Organisation issued their Guideline Development Process1 protocol and as part of their activity, commissioned two systematic reviews: one concerning the evidence for the relationship between ‘dietary sugars and body weight’ by Te Morenga et al2 and the other, namely: ‘the effects of sugars on dental caries’ by Moynihan and Kelly3. At the outset of this commentary, the reader should consider the need to study these two WHO commissioned reviews in tandem. This is of central importance for two reasons: first, the evidence of the effects of free sugars upon bodily disease should be conceptualized within a common risk factor approach and secondly, to permit the translation of this evidence into policy and practice. For the purposes of this invited review, my comments will be restricted to the work of Moynihan and Kelly3 (2013). The commentary will examine the quality of evidence presented and provide support for Moynihan and Kelly's3 recommendation that free sugars should be restricted to less than 5% E.4
The questions raised by the authors were simple and distinct, and based upon the WHO Nutrition Guidance Expert Advisory Group's questions. The authors, therefore, questioned the evidence on the effect of reducing or increasing sugar consumption upon dental caries prevalence and the evidence for a threshold of sugar intakes, with regard to dental caries experience. In order to answer their questions, Moynihan and Kelly first defined ‘sugars’ by providing the reader with a wide definition of dietary sugars to include: ‘total sugars’, ‘non-milk extrinsic sugars’, ‘free sugars’, ‘added sugars’ etc, expressed as ‘g or kg/day or/year’ or as ‘percentage E’. From this they provided a composite measure of dietary sugars which they conceptualised as ‘total sugars’. The outcome variable, dental caries, included measures of prevalence, incidence and/or severity (eg DMFT, deft etc) as well as comparisons between no caries and caries or between high and low caries experience.
The authors' search strategy, details of which are shown above, permitted 65 papers, detailing the findings of 55 studies, to emerge. The years of publication ranged from 19505 to 2010.6 Their search did not find any randomised controlled trials and the included non-randomised controlled trials, fundamentally flawed, were excluded from further analysis. Therefore, the study designs were population; cross-sectional studies; longitudinal cohort and cohort studies in adults and/or children. These were subjected to the GRADE assessment for quality. From the analysis of the available evidence, the authors stated that, ‘the evidence for an effect on dental caries of increasing or decreasing sugars intake as “moderate” for both children and adults’ and proposed that the population studies, ‘support the dose-effect’.3 However, including population studies raises the spectre of bias, due to ‘differences in prognosis in exposed and unexposed populations’ and particularly if the data were gathered over different time periods.7 All of the included studies were published in the years between 1950 and 2010. Would it be possible that the known changes in dietary habits, together with the ready availability of free sugars during the intervening 60 years, had an unaccountable effect on caries progression? Some evidence to support this proposition may be found in Sheiham and James.4 The role of prognostic factors relating to ‘fluoride exposure’ was commented upon by Moynihan and Kelly3 and it may be suggested that it was due to a lack of available data associated with more recent dietary changes that meant it was not possible for the authors to comment upon the effect of such key prognostic variables upon caries progression. Thus the lack of additional dietary data supports the authors' view that the evidence from population studies was of a ‘very low’ quality. Despite the moderate to low quality of the overall evidence, the limited meta-analysis suggested a ‘large effect size of sugars intake on dental caries’ and the recommendation to reduce sugars intake to less than 10kgs/person/year or less than 5% E was made.3
The significance of reducing sugar consumption to less than 5% E and the potential health ramifications this will have for populations across the world is undeniable. The question that arises, however, is how can the findings based upon evidence of a very low to moderate quality be used to inform policy and practice? To answer this question, it is necessary to return to Moynihan and Kelly's review and to read Sheiham and James' paper. Moynihan and Kelly have stated that ‘the effects of sugar on the dentition are lifelong’ while Sheiham and James have concluded that when sugar consumption contributes to 10% E there is ‘a costly burden of disease’. The question for governments is not about the quality of evidence but how they use this evidence to produce multi-sectorial policies that will promote health and reduce social inequality. It is the attention to detail in this review and the wealth of the evidence, irrespective of quality, that will act as a catalyst for governments to recommend a reduction in sugar intakes to less than 5% E and to support Sheiham and James' call for a value of less than 3% E. For governments to disregard the conclusions of Moynihan and Kelly's review to reduce sugar intakes ‘to less than 5% E to minimize dental caries throughout the life-course’, on account of the quality of the evidence, is ‘to throw the baby out with the bathwater’ and to subject future populations to a lifetime of dental caries as the consequence of increased sugar intakes.
References
World Health Organisation. Handbook for guideline development. Geneva. WHO. 2010: [Electronic version] http://apps.who.int/iris/bitstream/10665/75146/1/9789241548441_eng.pdf [Accessed 30th October 2014]
Te Morenga L, Mallard S, Mann J . Dietary sugars and body weight: systematic review and meta-analyses of randomised controlled trials and cohort studies. BMJ 2012; 346: e7492.
Moynihan P . Kelly S: Effect on caries of restricting sugars intake: systematic review to update WHO guidelines. J Dent Res 2014; 93: 8–18.
Sheiham A, James WP . A reappraisal of the quantitative relationship between sugar intake and dental caries: the need for new criteria for developing goals for sugar intake. BMC Public Health 2014; 14: 863.
Schulerud A: Dental caries and nutrition during wartime in Norway. Oslo. Fabritius and Sonners Trykkeri. 1950
Masson LF, Blackburn A, Sheehy C, et al. Sugar intake and dental decay: results from a national survey of children in Scotland. Br J Nutr 2010; 104: 1555–1564.
Guyatt GH, Oxman AD, Vist G, et al. GRADE guidelines: 4. Rating the quality of evidence--study limitations (risk of bias). J Clin Epidemiol 2011; 64: 407–415 doi: 10.1016/j.jclinepi.2010.07.017
Author information
Authors and Affiliations
Additional information
Address for correspondence: Paula Moynihan, WHO Collaborating Centre for Nutrition and Oral Health, Centre for Oral Health Research, Institute for Ageing and Health, Newcastle University, UK. E-mail paula.moynihan@ncl.ac.uk
Moynihan PJ, Kelly SA. Effect on caries of restricting sugars intake: systematic review to inform WHO guidelines. J Dent Res 2014; 93: 8–18.
Rights and permissions
About this article
Cite this article
Freeman, R. Moderate evidence support a relationship between sugar intake and dental caries. Evid Based Dent 15, 98–99 (2014). https://doi.org/10.1038/sj.ebd.6401055
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ebd.6401055
This article is cited by
-
20 years - 20 highlights
Evidence-Based Dentistry (2019)
-
Are restrictive guidelines for added sugars science based?
Nutrition Journal (2015)