Abstract
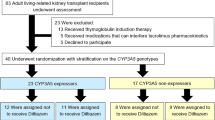
The impact of CYP3A5*3, a CYP3A5 nonexpresser genotype, on inhibitory effects of diltiazem on tacrolimus metabolism has not been assessed. In retrospective study, when coadministered with diltiazem, mean increments in dose-adjusted C0D7, Cmax and AUC0–12 h for tacrolimus were larger in CYP3A5 expressers than in CYP3A5 nonexpressers (48.7 vs 3.7%, 31.7 vs 17.2% and 38.2 vs 18.5%, respectively). Subsequently, a prospective study was carried out, patients were randomized to algorithm-predicted dosing or standard dosing. For CYP3A5 expressers, an algorithm guided by CYP3A5 and diltiazem significantly reduced tacrolimus maintenance dosage (P=0.009) and improved the accuracy of tacrolimus initial dose, resulting in reduction in out-of-range C0 after initial dose (P=0.002) and dose adjustments (P=0.004). However, for CYP3A5 nonexpressers, primary end points were not achieved, and tacrolimus-sparing effect of diltiazem was not remarkable. Our study results show that CYP3A5 genotype-guided tacrolimus–diltiazem combination is a promising therapy in renal transplant recipients in the early postoperative stage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Venkataramanan R, Swaminathan A, Prasad T, Jain A, Zuckerman S, Warty V et al. Clinical pharmacokinetics of tacrolimus. Clin Pharmacokinet 1995; 29: 404–430.
Staatz CE, Tett SE . Clinical pharmacokinetics and pharmacodynamics of tacrolimus in solid organ transplantation. Clin Pharmacokinet 2004; 43: 623–653.
Jones DR, Gorski JC, Hamman MA, Mayhew BS, Rider S, Hall SD . Diltiazem inhibition of cytochrome P-450 3A activity is due to metabolite intermediate complex formation. J Pharmacol Exp Ther 1999; 290: 1116–1125.
Hebert MF, Lam AY . Diltiazem increases tacrolimus concentrations. Ann Pharmacother 1999; 33: 680–682.
Jones TE, Morris RG . Pharmacokinetic interaction between tacrolimus and diltiazem: dose-response relationship in kidney and liver transplant recipients. Clin Pharmacokinet 2002; 41: 381–388.
Kuypers DR, de Jonge H, Naesens M, Vanrenterghem Y . Effects of CYP3A5 and MDR1 single nucleotide polymorphisms on drug interactions between tacrolimus and fluconazole in renal allograft recipients. Pharmacogenet Genomics 2008; 18: 861–868.
Kalliokoski A, Backman JT, Kurkinen KJ, Neuvonen PJ, Niemi M . Effects of gemfibrozil and atorvastatin on the pharmacokinetics of repaglinide in relation to SLCO1B1 polymorphism. Clin Pharmacol Ther 2008; 84: 488–496.
Uno T, Shimizu M, Yasui-Furukori N, Sugawara K, Tateishi T . Different effects of fluvoxamine on rabeprazole pharmacokinetics in relation to CYP2C19 genotype status. Br J Clin Pharmacol 2006; 61: 309–314.
Lim KS, Cho JY, Jang IJ, Kim BH, Kim J, Jeon JY et al. Pharmacokinetic interaction of flecainide and paroxetine in relation to the CYP2D6*10 allele in healthy Korean subjects. Br J Clin Pharmacol 2008; 66: 660–666.
Fan L, Zhang W, Guo D, Tan ZR, Xu P, Li Q et al. The effect of herbal medicine baicalin on pharmacokinetics of rosuvastatin, substrate of organic anion-transporting polypeptide 1B1. Clin Pharmacol Ther 2008; 83: 471–476.
Chandel N, Aggarwal PK, Minz M, Sakhuja V, Kohli KK, Jha V . CYP3A5*1/*3 genotype influences the blood concentration of tacrolimus in response to metabolic inhibition by ketoconazole. Pharmacogenet Genomics 2009; 19: 458–463.
Yasui-Furukori N, Saito M, Uno T, Takahata T, Sugawara K, Tateishi T . Effects of fluvoxamine on lansoprazole pharmacokinetics in relation to CYP2C19 genotypes. J Clin Pharmacol 2004; 44: 1223–1229.
Miura M, Tada H, Yasui-Furukori N, Uno T, Sugawara K, Tateishi T et al. Enantioselective disposition of lansoprazole in relation to CYP2C19 genotypes in the presence of fluvoxamine. Br J Clin Pharmacol 2005; 60: 61–68.
Yasui-Furukori N, Takahata T, Nakagami T, Yoshiya G, Inoue Y, Kaneko S et al. Different inhibitory effect of fluvoxamine on omeprazole metabolism between CYP2C19 genotypes. Br J Clin Pharmacol 2004; 57: 487–494.
Brosen K, Hansen JG, Nielsen KK, Sindrup SH, Gram LF . Inhibition by paroxetine of desipramine metabolism in extensive but not in poor metabolizers of sparteine. Eur J Clin Pharmacol 1993; 44: 349–355.
Lessard E, Yessine MA, Hamelin BA, O’Hara G, LeBlanc J, Turgeon J . Influence of CYP2D6 activity on the disposition and cardiovascular toxicity of the antidepressant agent venlafaxine in humans. Pharmacogenetics 1999; 9: 435–443.
Hustert E, Haberl M, Burk O, Wolbold R, He YQ, Klein K et al. The genetic determinants of the CYP3A5 polymorphism. Pharmacogenetics 2001; 11: 773–779.
Li JL, Wang XD, Wang CX, Fu Q, Liu LS, Huang M et al. Rapid and simultaneous determination of tacrolimus (FK506) and diltiazem in human whole blood by liquid chromatography–tandem mass spectrometry: application to a clinical drug–drug interaction study. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 867: 111–118.
Loparev VN, Cartas MA, Monken CE, Velpandi A, Srinivasan A . An efficient and simple method of DNA extraction from whole blood and cell lines to identify infectious agents. J Virol Methods 1991; 34: 105–112.
Wang Y, Wang C, Li J, Wang X, Zhu G, Chen X et al. Effect of genetic polymorphisms of CYP3A5 and MDR1 on cyclosporine concentration during the early stage after renal transplantation in Chinese patients co-treated with diltiazem. Eur J Clin Pharmacol 2009; 65: 239–247.
Renders L, Frisman M, Ufer M, Mosyagin I, Haenisch S, Ott U et al. CYP3A5 genotype markedly influences the pharmacokinetics of tacrolimus and sirolimus in kidney transplant recipients. Clin Pharmacol Ther 2007; 81: 228–234.
Haufroid V, Mourad M, Van Kerckhove V, Wawrzyniak J, De Meyer M, Eddour DC et al. The effect of CYP3A5 and MDR1 (ABCB1) polymorphisms on cyclosporine and tacrolimus dose requirements and trough blood levels in stable renal transplant patients. Pharmacogenetics 2004; 14: 147–154.
Hesselink DA, van Schaik RH, van der Heiden IP, van der Werf M, Gregoor PJ, Lindemans J et al. Genetic polymorphisms of the CYP3A4, CYP3A5, and MDR-1 genes and pharmacokinetics of the calcineurin inhibitors cyclosporine and tacrolimus. Clin Pharmacol Ther 2003; 74: 245–254.
Haufroid V, Wallemacq P, VanKerckhove V, Elens L, De Meyer M, Eddour DC et al. CYP3A5 and ABCB1 polymorphisms and tacrolimus pharmacokinetics in renal transplant candidates: guidelines from an experimental study. Am J Transplant 2006; 6: 2706–2713.
Loh PT, Lou HX, Zhao Y, Chin YM, Vathsala A . Significant impact of gene polymorphisms on tacrolimus but not cyclosporine dosing in Asian renal transplant recipients. Transplant Proc 2008; 40: 1690–1695.
Rodicio JL, Morales JM, Alcazar JM, Ruilope LM . Calcium antagonists and renal protection. J Hypertens Suppl 1993; 11: S49–S53.
Kumano K, Endo T, Koshiba K . [Functional and morphological changes in rat kidney induced by FK506 and its reversal by various vasodilators]. Nippon Hinyokika Gakkai Zasshi 1992; 83: 650–657.
Bensoussan C, Delaforge M, Mansuy D . Particular ability of cytochromes P450 3A to form inhibitory P450-iron-metabolite complexes upon metabolic oxidation of aminodrugs. Biochem Pharmacol 1995; 49: 591–602.
Yamaori S, Yamazaki H, Iwano S, Kiyotani K, Matsumura K, Honda G et al. CYP3A5 contributes significantly to CYP3A-mediated drug oxidations in liver microsomes from Japanese subjects. Drug Metab Pharmacokinet 2004; 19: 120–129.
Uesugi M, Masuda S, Katsura T, Oike F, Takada Y, Inui K . Effect of intestinal CYP3A5 on postoperative tacrolimus trough levels in living-donor liver transplant recipients. Pharmacogenet Genomics 2006; 16: 119–127.
Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet 2001; 27: 383–391.
Canaparo R, Finnstrom N, Serpe L, Nordmark A, Muntoni E, Eandi M et al. Expression of CYP3A isoforms and P-glycoprotein in human stomach, jejunum and ileum. Clin Exp Pharmacol Physiol 2007; 34: 1138–1144.
von Richter O, Burk O, Fromm MF, Thon KP, Eichelbaum M, Kivisto KT . Cytochrome P450 3A4 and P-glycoprotein expression in human small intestinal enterocytes and hepatocytes: a comparative analysis in paired tissue specimens. Clin Pharmacol Ther 2004; 75: 172–183.
Kliewer SA . The nuclear pregnane X receptor regulates xenobiotic detoxification. J Nutr 2003; 133: 2444S–2447S.
Zhang J, Kuehl P, Green ED, Touchman JW, Watkins PB, Daly A et al. The human pregnane X receptor: genomic structure and identification and functional characterization of natural allelic variants. Pharmacogenetics 2001; 11: 555–572.
Lamba J, Lamba V, Strom S, Venkataramanan R, Schuetz E . Novel single nucleotide polymorphisms in the promoter and intron 1 of human pregnane X receptor/NR1I2 and their association with CYP3A4 expression. Drug Metab Dispos 2008; 36: 169–181.
Jusko WJ, Piekoszewski W, Klintmalm GB, Shaefer MS, Hebert MF, Piergies AA et al. Pharmacokinetics of tacrolimus in liver transplant patients. Clin Pharmacol Ther 1995; 57: 281–290.
Acknowledgements
We acknowledge the financial support provided by the National Major Projects for science and technology development from Science and Technology Ministry of China (no. 2009ZX09304-003), National Natural Science Foundations of China (nos. 30873124 and 30873125) and Science and Technology Planning Project of Guangdong Province (no 2007B031511001). The authors would like to thank Dr Qibiao Su (Department of Pharmacokinetics & Pharmacodynamics, The South China Center for Innovative Pharmaceuticals) for the language editing of the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Li, JL., Wang, XD., Chen, SY. et al. Effects of diltiazem on pharmacokinetics of tacrolimus in relation to CYP3A5 genotype status in renal recipients: from retrospective to prospective. Pharmacogenomics J 11, 300–306 (2011). https://doi.org/10.1038/tpj.2010.42
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2010.42
Keywords
This article is cited by
-
The effect of the very low dosage diltiazem on tacrolimus exposure very early after kidney transplantation: a randomized controlled trial
Scientific Reports (2022)
-
Diltiazem on tacrolimus exposure and dose sparing in Chinese pediatric primary nephrotic syndrome: impact of CYP3A4, CYP3A5, ABCB1, and SLCO1B3 polymorphisms
European Journal of Clinical Pharmacology (2021)
-
Kidney podocyte-associated gene polymorphisms affect tacrolimus concentration in pediatric patients with refractory nephrotic syndrome
The Pharmacogenomics Journal (2020)
-
Diltiazem used as a tacrolimus-sparing agent for treatment of pediatric patients with refractory nephrotic syndrome: a case report and retrospective analysis
European Journal of Clinical Pharmacology (2019)
-
Conversion from Twice-Daily Prograf® to Once-Daily Advagraf® in Multi-ethnic Asian Adult Renal Transplant Recipients With or Without Concomitant Use of Diltiazem: Impact of CYP3A5 and MDR1 Genetic Polymorphisms on Tacrolimus Exposure
European Journal of Drug Metabolism and Pharmacokinetics (2019)