Abstract
Salmonella serovar Montevideo (SM) is frequently associated with human Salmonella infections and causes gastrointestinal disease, cases are common particularly among individuals who come in close contact with live poultry or poultry meat products. To characterize SM disease in chickens, the pathogenic traits and tissue predilections of the disease were investigated. Dissemination of fluorescent-tagged SM (JOL1575GFP) was monitored after oral and intramuscular mock infections of specific-pathogen-free chickens. The spleen was predominantly affected by intramuscular infection while the cecum, spleen, and minimally liver were affected by oral infection. No conspicuous illness was observed in infected birds, and histopathological examination showed minimal damage of the intestinal epithelium and splenic parenchyma though SM was readily isolated from these tissues. Levels of SM internalization by primary chicken peritoneal macrophages were similar to that of Salmonella Typhimurium. SM was more sensitive to chicken than rabbit serum complement killing. Internal egg contamination of SM mock infected layers also occurred at trace levels and lasted for a week after inoculation. This study also confirmed that SM infection in chickens is sub-clinical and asymptomatic, which suggests that latent asymptomatic carriers may excrete a large number of bacteria and transmit the pathogen by contaminating water or food sources.
Similar content being viewed by others
Introduction
Salmonellosis remains an important food-borne illness1. Recent years have seen a rise in human infections and outbreaks of a particular serotype, Salmonella Montevideo (SM), around the globe. It has been reported in the USA, Europe, Australia, and Asia including Korea and Japan2,3,4. In 2002, the first SM outbreaks were identified in Australia and New Zealand, and, since then, 68 infected individuals have been reported5. In 2010, an SM outbreak associated with a dietary food supplement was reported in Germany, with an unusual 15 SM infections being identified6. In the US, from 2009 to 2015, several SM outbreaks were reported, and the Centers for Disease Control and Prevention (CDC) recorded more than 500 cases during this period2,7,8. In several cases, the poultry sector was established as the source of SM. For instance, a multistate outbreak of human SM infections in 2012 in the US was linked to live poultry in backyard flocks9. In humans, non-typhoidal salmonellosis (NTS) is usually observed as localized entero-colitis, and the infected patients exhibit clinical signs of diarrhea, nausea, abdominal pain, mild fever, and chills2. Bacteremia due to multi-drug resistant SM was documented in a 3-year-old child3, and SM involvement has been reported in serous arthritis10, septic arthritis11, and acute myocarditis12. Cattle and sheep are commonly affected by SM infections, causing severe conditions, such as abortions, in sheep13. SM is readily isolated from cecum and cloacal swabs of infected chickens14. In most human SM infections, the likely source of the outbreak was live poultry or poultry products. While SM infection in humans is a gastro-intestinal disease, SM infection in chickens is not well documented. To date, it is unclear whether ‘salmonellosis’ is an appropriate term for chicken SM infection. Furthermore, contaminated eggs may transmit Salmonella to humans.
Our understanding of the pathogenic traits of NTS has markedly advanced over recent years through the investigation of host interaction with genetically modified mutant Salmonellae and through the development of suitable experimental models15. After oral entry, Salmonella actively invades intestinal epithelial cells, and the SPI-1-encoded type III secretion system (TTSS) provides the effector molecules required for tissue invasion16. Salmonella can survive in phagocytic and non-phagocytic cells. Intracellular survival requires a second TTSS that is encoded by SPI-217. Much of our knowledge on Salmonella pathogenesis came from mammalian host responses to Salmonella. Mice have been extensively used as a model for experimental infections. Most work in chicken models has focused on the mechanisms of enteric infection by Salmonella Typhimurium (ST) and enteric or egg infection by Salmonella Enteritidis18. Avian salmonellosis can be classified into three distinct phases, in a pattern similar to that of mammals. Invasion of the gastrointestinal tract marks the first phase of the disease. The second phase is the establishment of systemic infection mainly by intracellular infection of macrophages. In the third phase, infection is cleared by an immune response, the bird succumbs to the infection, or a subclinical carrier state develops18. However, differences between mammalian and avian host responses to Salmonella prevent accurate comparisons15. Little information is available on the pathogenesis of non-host specific NTS, such as SM. In the absence of a suitable model, investigation of SM infection in chickens is pragmatic.
In vivo and in vitro insights into the pathobiology of SM infection in chicken may help to devise effective control and treatment strategies. To this aim, we investigated for the first time, the pathobiology of SM infection in chickens, dissemination in chicken organs and eggs, and the pathological implications of experimental infection with wild-type SM in specific pathogen free (SPF) chickens. We also investigated cellular SM uptake by chicken peritoneal macrophages, in vivo invasion levels by using rabbit and chicken intestinal loops, serum complement killing sensitivity, and the histopathology and immunohistochemistry of infected organs.
Results
Salmonella Montevideo is more sensitive to chicken complement than rabbit complement
To evaluate the sensitivity of SM strains to serum complement killing, JOL1575, JOL1577, and reference ST strains were exposed to chicken and rabbit serum complements. The principle behind the test was to determine the sensitivity of the bacteria to complement-mediated bacterial lysis. Fresh serum contains an active complement system that lysed susceptible organisms, but heat treatment for 30 min at 56 °C inactivated the serum complements leading to decomplementation. The effect of bacterial cell lysis by serum complement killing was measured in CFU. SM was more susceptible to complement lysis by chicken serum than was ST (Fig. 1). However, SM was relatively resistant to complement killing by rabbit serum.
S. Montevideo serotype susceptibility to complement killing was assessed with chicken and rabbit complements. One hour after incubation with complement, viable bacteria were determined by plating on BGA plates. CFU of bacterial strains are represented as percent reduction. (A) SM strains were more susceptible to chicken serum than was the reference ST strain. (B) SM was relatively resistant to rabbit complement. *P ≤ 0.05; error bars indicate the SEM.
Salmonella Montevideo invades chicken intestinal epithelium in a similar pattern to Salmonella Typhimurium
An intestinal loop assay was performed to ascertain the degree of invasiveness of the strains to the intestinal epithelium in vivo. The invasiveness of wild-type SM JOL1575, JOL1577, and JOL401 was compared to evaluate the ability of the model to demonstrate differences between strains. The degree of intestinal epithelial invasion by JOL1575 and JOL1577 was highly similar to the reference strain, JOL401. The competence of SM strains in invading the epithelium of chicken intestines was equivalent to that of ST (Fig. 2A). The rabbit intestinal loop model did not show significant differences between the SM and ST strains in terms of intestinal- bacterial uptake. The average log10 CFU of bacteria recovered from the chicken intestinal loop was 7.57, 7.66, and 7.77 from loops dosed with SM JOL1575, SM JOL1577, and ST, respectively. The mean uptake of the rabbit intestinal loop was 2.46, 2.88, and 3.04 from loops dosed with SM JOL1575, SM JOL1577, and ST, respectively (Fig. 2A).
The ability of SM strains to invade host tissues and cells was investigated with intestinal loop assay and macrophage uptake assay. (A) Ligated chicken and rabbit intestinal loops were inoculated with 1 × 108CFU of JOL1575, JOL1577, and JOL401 strains. Intracellular bacteria were harvested by cell lysis and plated on BGA plates. Bacterial CFU were obtained for each loop, and the mean log10 CFU/cm.sq ± SEM was determined. The invasion capability did not differ significantly among strains in the chicken or rabbit intestinal models. However, each strain had diminished invasion capability in the rabbit model as compared to the chicken. (B) In vitro cellular uptake was investigated by using chicken intraperitoneal macrophages. A total 1 × 105 cells were infected with JOL1575, JOL1577, or JOL401 at an MOI of 10:1. Extracellular bacteria were killed with gentamicin antibiotics incubation. Internalized bacteria were harvested by cell lysis 2 hr after inoculation, and total CFU per well were determined. SM strains and ST did not differ significantly on the macrophage uptake assay. Error bars indicate the SEM.
To examine the in vitro cellular invasiveness of the strains, macrophage uptake was assessed in chicken primary intraperitoneal macrophages. Both serotypes were efficiently internalized by chicken peritoneal macrophages. No significant difference was observed between JOL1575 and JOL401 in their propensity to taken up by macrophages (Fig. 2B).
In vivo tracking of JOL1575GFP dissemination in chickens
Visceral chicken organs affected by inoculation with wild type SM were monitored and tracked with strain JOL1575GFP expressing GFPuV (Supplementary Fig. 1). At 48 hr after inoculation, the spleen and cecum were the main sites of SM bacterial localization. Fluorescent signals from the SM-GFP strains were also detected in the liver, although at a lower intensity than in the spleen or intestinal epithelial tissues. The duodenum and jejunum also revealed infection (Fig. 3). GFP signals were not observed in the crop, lungs, or proventriculus. Reproductive tissues did not show any signs of SM localization or infection. Oviduct and ovarian tissues did not show a JOL1575GFP signal upon fluorescence microscopic examination. Ex vivo whole-organ fluorescence images were also acquired, with representative spleens showing fluorescence (Supplementary Fig. 2). No SM-GFP-associated fluorescence signals were observed in organs of control uninoculated birds.
SM dissemination was tracked with a GFP-tagged SM strain at 48 hr after mock infection. Immunologically mature SPF chickens were inoculated with JOL1575GFP expressing GFPuV which is having a peak excitation at 395 nm and a peak emission at 509 nm. Uninoculated chickens were used as a control to differentiate autofluorescent tissues. Thin tissue biopsy sections were collected from representative organs and observed under a fluorescent microscope. Infection foci were observed in the liver, small intestines, spleen, and cecum of infected birds. The organs of control uninoculated birds had no SM-GFP fluorescence signal. Black arrow heads indicate fluorescent signals emitted by SM-GFP in infected tissues.
Salmonella Montevideo infection showed minimal pathological lesions in chickens
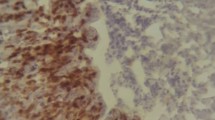
To gain insight into SM interaction with chicken organs, tissue samples from mock-infected chickens were investigated by using histopathological sections and immunohistochemical analysis. Tissue samples were collected at 20 days after inoculation to allow SM infection to establish in permissive tissues and allow the initial host inflammatory response to subside. No conspicuous tissue damage was observed in histopathological samples of the intestines 20 days after infection (Fig. 4). Although SM bacteria were detected with specific anti-SM antibodies in the villi of the intestine, spleen, and cecum, no disruptive or characteristic damage on the epithelia was observed (Fig. 5). The splenic parenchyma also did not have any conspicuous abnormality, such as lymphocyte depletion or giant cell infiltration. We also investigated the possible involvement of the reproductive tract in internal egg contamination. Immunostaining and histopathology of uterus sections from SM-infected layer chickens did not reveal the presence of SM (Fig. 5). Strains JOL1575 and JOL1577 had no variations in pathological lesions.
Brown nick layers were mock infected with wild-type SM strains. Twenty days after inoculation and establishment of SM infection, birds were euthanized, tissue samples were collected from the small intestine, spleen, cecum, uterus and subjected to H&E-based histopathology. Representative H&E images of organs from mock-infected chickens are shown. On the SM-image panel, no prominent evidence of pathological lesions, such as necrosis, disruption of villi were observed.
Brown nick layers were mock infected with wild type JOL1575 and JOL1577 strains. Twenty days after inoculation, tissue sections were collected from the liver, intestines, cecum, uterus, and spleen and subjected to IHC analysis. Representative IHC images of chicken tissue sections 20 days after infection with a chicken anti-Salmonella polyclonal antibody. Salmonella was detected in the apical portion of the intestinal villi, in the splenic parenchyma and caecal mucosa. Sections were visualized with DAB chromogenic substrate and hematoxylin counterstain. Black arrowheads indicate reactive signals specific for Salmonellae on the tissue sections.
Salmonella Montevideo causes internal contamination of hen eggs
The degree of internal egg contamination by SM was studied in immunologically mature brown nick layers, which were monitored for four weeks after inoculation. Immediately after inoculation with wild-type SM, egg production declined drastically (Table 1). The mean egg production was 50.75 ± 0.74, 48.5 ± 1.94, and 46.25 ± 2.09 for control, JOL1575-inoculated, and JOL1577-inoculated groups, respectively. The egg production rate for 4 weeks after inoculation was 72.50%, 69.29%, and 66.07% for the control, JOL1575-inoculated, and JOL1577-inoculated groups, respectively. Of the internal egg contents 1.66% and 2.25% tested positive in the JOL1575 and JOL1577 groups, respectively, in the first week after inoculation (Table 1). No further egg internal contamination was observed during mock infection of layers with SM. However, since SM continued to reside in the host much longer than the study period, sporadic internal egg contamination beyond the experimental time period was not ruled out.
Discussion
The involvement of live poultry as well as poultry meat and products in transmitting SM infections to humans is well documented4,9,19. Insight into chicken host-pathogen interactions is important not only to elucidate SM bacterial pathogenesis, but also from an epidemiological perspective. To date no significant studies related to chicken SM infection have been reported. In this study, dissemination of SM infection was monitored by using GFP expressing JOL1575-GFP. The main drawback of whole body imaging and tracking is background fluorescence emitted by auto-fluorescing tissues20. Background fluorescence may preclude in-depth analysis of GFP signal and could mask the investigation. However, in this study we validated the results by using uninfected control chickens. Thin biopsies encompassing several portions of the internal organs were excised, and micro-tissue sections were prepared for fluorescence microscopy. Experimental SM infection was comparable to Salmonella infection in avian species. SM had a similar pattern of intestinal invasion at the early phase of infection. However, the infection was not associated with severe enteritis, hemorrhage, or secretory diarrhea.
Host-species differences in complement activity have long been known and attributed to differences in complement proteins21,22. Complement killing can be impeded by expression of an outer membrane protein encoded by the resistance to complement killing (rck) gene, which is present in some NTS strains and interferes with the formation of the membrane attack complex. However, whole-genome sequences of the SM strains revealed the absence of the rck gene. Amino acid homology and sequence alignments of Rck proteins from other Salmonella strains showed low similarity with any outer membrane proteins of SM strain (Supplementary Fig. 3). O-antigen (O-Ag) also determines the susceptibility of bacterial cells to complement-mediated killing23. The SM and ST display smooth phenotype with complete O-chain length. However, our data revealed differences in susceptibility, SM is more susceptible to chicken serum than to rabbit serum at least with the current isolates tested (Fig. 1). This may explain in part why SM infection of chickens is mild and may be more virulent in rabbits and other mammals, such as humans. However, this hypothesis requires further investigation, evaluating extensively on several SM isolates.
Invasion of the gut epithelium is essential to Salmonella pathogenesis, and differences in virulence among serotypes may be related to differences in invasiveness. Both in vivo and in vitro invasion models have been used24, but in vitro models may not incorporate all possible invasion pathways25. The lack of suitable chicken epithelial cell lines further limits the use of in vitro models26. In this study, a similar pattern of invasiveness was observed between SM strains and the reference strain, ST, both in chicken and rabbit intestinal loops. In chickens, infection of the GI tract by S. Typhimurium results in an influx of heterophils and inflammation whereas non-flagellated S. Pullorum and S. Gallinarum infections cause only limited inflammation. Based on post mortem examination, the swelling or inflammation observed in intestinal loops of the present study was highly similar between SM and ST infected loops (Supplementary Fig. 4). It is tempting to presume that the flagella of SM may evoke an inflammatory response similar to that of flagellated ST. Moreover, the avr gene is absent from SM, which has been suggested to inhibit the inflammatory process, particularly cytokine and chemokine induction, by acting on the NF-kB signaling pathway27. However, this hypothesis requires further investigation.
The replication and survival of Salmonella inside macrophages is key to the progression of systemic infection in both mammals and birds28. Salmonella mutants that cannot survive within macrophages are less virulent29. The present study indicated that SM and ST have similar patterns of macrophage uptake and survival (Fig. 3). However, the cytotoxic and apoptotic effects of SM strains were not assessed. SM replication and survival may be comparable to those of ST in vivo.
Salmonella starts invading chickens from the gastrointestinal tract. In this study, we observed that the intestinal epithelium, duodenum, ileum, and jejunum were infected by SM-GFP with no site preference. In the initial stage of infection, spleen, liver, and cecum were affected (Fig. 3). Chicken host specific S. Gallinarum induces anemia and septicemia, it further causes gastrointestinal tract hemorrhage, massive inflammatory infiltration, and ulceration of the intestinal wall30. Typically 6–10 days after experimental infection, the birds succumbed to the infection30,31. However, in this study SM-infected chickens had no conspicuous pathological lesions on the internal organs. None of the chickens died due to SM experimental infection. In a separate experiment, we observed that SM persisted in the ceca and spleen of chicken for 31 days after experimental infection.
The pathology of the alimentary tract differs among Salmonellae infections32. Immuno-histochemical analysis of tissue samples from SM mock-infected chickens revealed little evidence of enteritis (Fig. 4). Few samples had mild neutrophil infiltration due to invading SM pathogens. SM bacteria were detected on the apical portion of the intestinal villi. However, no disruption of villi morphology was evident. Splenic and cecal epithelial cells also harbored bacteria without severe cellular infiltration or signs of chronicity. Liver and uterine sections did not harbor SM or show evidence of inflammation or infiltration. Hence, these findings further indicate that chickens maybe asymptomatic carriers of SM infections. This carrier state may result in excretion of large numbers of bacteria in their feces. They may transmit the pathogen by contaminating water or food sources, thus could be responsible for foodborne epidemics. However, in order to evaluate the gravity of the epidemiological risk factor, it would be necessary to investigate the persistency of SM in chickens and viability of the organism in the environment.
Salmonella serotypes frequently transmit infection from hens to eggs and progeny18. Systemic infection recurs at the onset of egg laying and may spread to the reproductive tract. This phenomenon is related directly to the physiological and hormonal changes associated with sexual maturity in hens33. In this study, eggs were only contaminated during the first week after SM infection. Physiological stress and changes in homeostasis among the layers may be responsible for SM spreading to the eggs, especially during the initial stages of infection. Our in vivo tracking and bacterial isolation experiments with SM-GFP did not detect significant SM in the chicken ovaries. This result suggests that SM egg contamination may arise from a hematogenous route, unlike S. Enteritidis, which colonizes the reproductive tract and internally contaminates eggs before being laid34. Egg contamination may occur sporadically beyond the 4-week study period since the chickens continued to harbor SM in their system. Contaminated eggs produced by infected laying hens are thought to be a main source of human Salmonella infection35. However, external soiling of eggshells can also lead to subsequent entry into the eggs, so the route of internal egg contamination by a Salmonella serovar must be validated. We observed low level of internal egg contamination possibly by hematogenous route. Despite the level of contamination, it may still pose a foodborne illness threat.
In conclusion, we studied the effects of SM infection in vivo and in vitro. We confirmed the organs targeted by SM infection in chickens by GFP tagged SM. SM sensitivity to chicken serum complement may contribute to attenuated SM infection in chickens, apart from inherent host resistance to the organism. SM infection in chickens is sub-clinical as only minimal morbidity was observed in experimentally infected chickens. Immunohistochemical studies revealed SM in epithelial cells of the intestine, spleen, and cecum; however, the anatomical features had minimal disruptions. Eggs were internally contaminated following mock-infection by SM suggesting a hematogenous route, rather than invasion of reproductive cells, was responsible for transient egg contamination. These findings also indicate that SM infection is asymptomatic in chicken hosts. Chickens carrying SM infection pose a risk of poultry-associated SM outbreaks in humans.
Materials and Methods
Bacterial strains
All the plasmids and bacterial strains used in this study are listed in Table 2. The wild-type SM strains used in this study were isolated from chickens in South Korea. The strains were characterized by biochemical testing with the API 20E panel36 and genetic profiling36 with a special focus on the Salmonella pathogenicity island (Table 2). For in vivo tracking, a green fluorescent protein (GFP)-tagged SM strain JOL1575GFP, was generated by electro-transforming wild-type SM JOL1575 with the pEcoli-6xHN-GFPuv plasmid (Clontech, US). JOL1575GFP was grown in medium supplemented with 40 μg/mL ampicillin. SM and Salmonella Typhimurium (ST) strains were routinely grown on LB broth and LB agar plates aerobically at 37 °C. All bacterial strains were stored at −80 °C in growth medium containing 20% glycerol.
Ethics Statement
All animal experimental procedures were approved (CBNU2015-00085) by the Chonbuk National University Animal Ethics Committee in accordance with the guidelines of the Korean Council on Animal Care. All chickens and rabbits used in the study were housed and maintained humanely; they were provided water and antibiotic-free food ad libitum.
In vitro serum complement sensitivity assay
The serum complement sensitivity of SM strains was ascertained with chicken and rabbit sera using a protocol described earlier37. Fresh sera, negative of anti-Salmonella antibodies were collected from chickens and rabbits. All the bacterial strains were grown to the late log phase, 1 × 103 CFU/100 μl each of JOL1575, JOL1577, JOL401, and DH5α control cultures were incubated separately with 100 μL of PBS, 100 μL of 50% complemented serum and 100 μL of de-complemented serum for 1 hr at 37 °C. After incubation, each test lot was plated on an LB agar plate and CFU were enumerated after overnight incubation at 37 °C. The data were represented as mean percentage reduction. The non-pathogenic lab strain DH5α was used as an internal control to measure complement activity and was most susceptible to complement killing. Three replicates were made for each assay and the experiment was performed twice.
In vivo intestinal loop assay
The intestinal loop assay was performed on rabbit and chicken models (n = 3 each). The assay was conducted as described previously with minor modifications26. Experimental animals were fasted and maintained with water only for 24 hr. They were anesthetized by administering 30 mg of tiletamine-zolazepam (Zoletil 50, Virbac, US) and 11.66 mg of xylazine (Ropum, Bayer, Germany). Anesthesia was maintained with 15 mg of tiletamine-zolazepam and 11.66 mg of xylazine. The animals were covered with a sterile surgical blanket, and chicken abdomens were de-feathered. For chickens, a ventral approach was used while a lateral approach was used for rabbits. After the incision area was disinfected, the abdomen was opened, and the jejunum was carefully exposed. Nine to ten ligated loops were constructed with polypropylene blue monofilament surgery suture (Prolene, Ethicon, US). The loops were 4 cm long and separated by spacer loops to allow excision of the loops and minimize the risk of contamination from adjacent loops. Ligated loops were inoculated with 1 × 108 CFU of JOL1575, JOL1577, or JOL401. After inoculation, the loops were reintroduced into the abdomen, and the abdominal wall was sutured. Two hours later, 600 μL of PBS containing gentamicin (300 mg/mL) was added to each loop. After another hour the subject was euthanized with an anesthetic overdose. Three 1 × 1-cm biopsies were taken from each loop. The biopsies were placed in a 15 mL tube and homogenized with 2 mL of ice-cold PBS with 1% triton X-100 by using a tissue homogenizer (T-10 Basic Ultra Turrax, IKA, Germany) for 4 min. After 10 min, 20-fold serial dilutions of the homogenate were made in PBS, and 100 mL of each dilution was spread on BGA agar. The plates were incubated aerobically overnight at 37 °C. Salmonella colonies were confirmed using PCR protocol described earlier36, and CFU were enumerated. The data were expressed as mean log10 CFU/cm.sq ± SEM.
Isolation of intraperitoneal macrophage and invasion assay
Sephadex-elicited abdominal exudate cells were harvested from brown nick layer chickens at 7 weeks of age as described previously38. Invasion analysis was performed in triplicate with minor modifications to previously reported method39. Five milliliters of 1% sephadex G100 solution was injected intraperitoneally in 5- to 6-week-old chickens. Three days after injection, the animals were fasted for 12 hr and euthanized. The feathers were wetted with 70% ethanol, and the skin was peeled off the ventral abdominal area. The exposed muscle layer was again wiped with ethanol. Fifty to 80 mL of chilled cell culture medium RPMI-1640 was injected into the peritoneal cavity through a 24-gauge needle. The abdomen was massaged gently but thoroughly with the liquid medium still inside. Exudates containing intraperitoneal macrophages were harvested via ventral surgical incision. The harvested cells were pelleted and washed with medium by resuspension and re-centrifugation at 2500 RPM (700 RCF) for 8 min. Intraperitoneal macrophages was resuspended in 1 mL of RPMI-1640, and the total viable cell count determined. Macrophage cells (5 × 105) were seeded on 96-well tissue culture plates (SPL, Korea) and kept at 37 °C overnight in a CO2 incubator. The sensitivity of each strain against gentamicin was preliminarily tested. Bacterial invasion was estimated by lysing adherent cells with 100 μL of 1% Triton X in PBS and plating the lysate on LB agar at adequate dilutions.
Macrophage uptake of SM and reference strains was assessed as described earlier40 with minor modifications. The experiment was performed by infecting chicken primary intraperitoneal macrophages with a multiplicity of infection (MOI) of 10:1. To initiate infection, 5 × 106 CFU of SM or ST was added in triplicates to macrophage cell cultures. The cell culture plates were centrifuged at 200 g for 5 min. The macrophages were incubated at 37 °C in a 5% CO2 atmosphere for 20 min. The culture supernatant was removed with a sterile pipette and replaced with 100 μL of RPMI supplemented with 100 μg/mL gentamicin to kill extracellular bacteria. The macrophages were incubated at 37 °C in a 5% CO2 atmosphere for 48 hr. The culture media and monolayers were washed twice with an equal volume of RPMI. The cells were lysed with 0.5% Triton-X after 10 min of incubation. The released bacteria were harvested and plated directly on BGA plates and incubated at 37 °C overnight. Salmonella colonies were confirmed using PCR protocol described earlier36. The experiment was conducted twice and the data were expressed as mean log10 CFU/mL ± SEM.
In vivo tracking of green fluorescent protein-tagged SM JOL1575-GFP dissemination in chickens
Immunologically mature SPF chickens (n = 4) were inoculated with the tagged SM, JOL1575GFP. Uninoculated chickens were kept as controls. At specified time intervals 48 hr after inoculation, chickens were euthanized and organs were isolated aseptically with great care to avoid internal blood-borne Salmonella contamination. Ten representative tissue sections were made from each organ. The tissue slices were compressed between a microslide and coverslip. The sections were observed under a fluorescent microscope (Axio Imager 2, Zeiss., Germany) and images were acquired in both the bright field and GFP spectra (AxioVision, Zeiss, Germany).
Histopathology and immunohistochemical examination of affected organs
To investigate the colonization and progress of SM infection in chickens, 22-week-old brown nick layer chickens (n = 4) were inoculated with JOL1575 or JOL1577. Twenty days after inoculation, representative tissue samples of the liver, spleen, uterus, intestines, and cecum were collected and stored in neutral buffered 10% formalin. Tissue sections were prepared, deparaffinized, stained with hematoxylin and eosin (H&E), and examined by light microscopy (Axio Imager 2, Zeiss, Germany) and digital imaging software (Axio Vision, Zeiss, Germany).
Immunohistochemical staining of the tissue sample was carried out as described previously41 with modifications. Paraffin sections were deparaffinized in xylene and graded ethanol and hydrated to distilled water. Three percent hydrogen peroxide was used to block endogenous peroxidase activity. Heat-induced antigen retrieval was carried out at 100 °C for 30 min in antigen retriever citrate buffer (Sigma-Aldrich, US). Non-specific sites on the tissue were blocked with 5% skim milk followed by overnight incubation with a primary chicken anti-Salmonella polyclonal antibody at 1:1000 dilution. After blocking with 10% goat serum, a secondary goat anti-chicken IgY HRP antibody at 1:1000 dilution was added. Each step was followed by washing with PBS for 5 min three times. All incubations were done in a humidified atmosphere. Vectastain DAB (Vector Laboratories, US) was used as a colorigenic substrate. The sections were counterstained with hematoxylin and mounted with MM 24 Mounting Media (Leica Biosystems, Germany) for microscopy.
Internal egg contamination
Twenty- to 22-week-old layer hens were grouped (n = 10) and inoculated orally with 108 CFU SM wild-type strains JOL1575 and JOL1577. Eggs were collected regularly for 4 weeks, and processed every other day. Eggs were cleaned and soaked in 70% ethanol for 3 min. After the egg surface was completely dry, the egg contents were collected in a 500 mL sterile breaker. An equal amount of buffered peptone water was added to dilute the egg contents. After thorough homogenization with sterile spatula, 200 μL of sample was directly plated on BGA plates. Ten milliliters of Rappaport Vassiliadis broth was added to an equal amount of the sample and enriched at 42 °C for 48 hr. The sample was plated on BGA and the bacterial load was measured. SM-specific primers were used to confirm the Salmonella-like bacteria. This experiment was repeated in triplicate.
Statistical analysis
Statistical analyses were used wherever applicable. Analyses were performed with SPSS 16.0 (SPSS Inc., USA). One-way analysis of variance (ANOVA) and Student’s t-tests were used to determine statistically significant differences, with P values of ≤ 0.05 or ≤ 0.01 considered significant.
Additional Information
How to cite this article: Lalsiamthara, J. and Hwa Lee, J. Pathogenic traits of Salmonella Montevideo in experimental infections in vivo and in vitro. Sci. Rep. 7, 46232; doi: 10.1038/srep46232 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
04 May 2017
A correction has been published and is appended to both the HTML and PDF versions of this paper. The error has not been fixed in the paper.
04 May 2017
Scientific Reports 7: Article number: 46232; published online: 07 April 2017; updated: 04 May 2017 The Acknowledgements section in this Article has been omitted. It should read: “Acknowledgements This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MISP) (No.
References
Coburn, B., Grassl, G. A. & Finlay, B. B. Salmonella, the host and disease: a brief review. Immunol. Cell Biol. 85, 112–8 (2007).
Centers for Disease Control and Prevention (CDC). Salmonella Montevideo infections associated with salami products made with contaminated imported black and red pepper – United States, July 2009-April 2010. Morb. Mortal. Wkly. Rep. 59, 1647–1650 (2010).
Kim, J.-Y. et al. Case Report: Bacteremia Due to Salmonella enterica Serotype Montevideo Producing Plasmid-Mediated AmpC β -Lactamase (DHA-1). Ann. Clin. Lab. Sci. 34, 214–217 (2004).
Harada, T. et al. Molecular epidemiological investigation of a diffuse outbreak caused by Salmonella enterica serotype Montevideo isolates in Osaka Prefecture, Japan. Foodborne Pathog. Dis. 8, 1083–8 (2011).
Unicomb, L. E. et al. Sesame seed products contaminated with Salmonella: three outbreaks associated with tahini. Epidemiol. Infect. 133, 1065–1072 (2005).
Stöcker, P. et al. Outbreak of Salmonella Montevideo associated with a dietary food supplement flagged in the Rapid Alert System for Food and Feed (RASFF) in Germany, 2010. Euro Surveill. Bull. Eur. sur les Mal. Transm. = Eur. Commun. Dis. Bull. 16, 20040 (2011).
Gieraltowski, L. et al. Nationwide outbreak of Salmonella Montevideo infections associated with contaminated imported black and red pepper: warehouse membership cards provide critical clues to identify the source. Epidemiol. Infect. 141, 1244–52 (2013).
Hedican, E. et al. Salmonellosis outbreak due to chicken contact leading to a foodborne outbreak associated with infected delicatessen workers. Foodborne Pathog. Dis. 7, 995–7 (2010).
CDC. Multistate outbreak of human Salmonella Montevideo infections linked to live poultry in backyard flocks. (2012). Available at: http://www.cdc.gov/Salmonella/montevideo-06-12/index.html.
Gordon, H. S. & Hoffman, S. J. Serous arthritis of the knee joint; report of a case caused by Salmonella Typhosa and Salmonella Montevideo in a child. J Am Med Assoc 141, 460 (1949).
Katsoulis, E., Pallett, A. & Bowyer, G. W. Septic arthritis of the knee by Salmonella montevideo. Ann. R. Coll. Surg. Engl. 86, 272–274 (2004).
O’Connor, K. Acute myocarditis precipitated by Salmonella Montevideo infection: a case report. Ir Med J 93, 21–22 (2000).
Linklater, K. A. Abortion in sheep associated with Salmonella Montevideo infection. Vet Rec 112, 372–374 (1983).
Schleifer, J. H., Juven, B. J., Beard, C. W. & Cox, N. A. The susceptibility of chicks to Salmonella Montevideo in artificially contaminated poultry feed. Avian Dis. 28, 497–503 (1984).
Santos, R. L. Pathobiology of Salmonella, intestinal microbiota, and the host innate immune response. Front. Immunol. 5, 1–7 (2014).
Altier, C. Genetic and environmental control of Salmonella invasion. J Microbiol 43 Spec No, 85–92 (2005).
Hansen-Wester, I. & Hensel, M. Salmonella pathogenicity islands encoding type III secretion systems. Microbes Infect. 3, 549–559 (2001).
Chappell, L. et al. The immunobiology of avian systemic salmonellosis. Vet. Immunol. Immunopathol. 128, 53–59 (2009).
Dominguez, M. et al. Outbreak of Salmonella enterica serotype Montevideo infections in France linked to consumption of cheese made from raw milk. Foodborne Pathog Dis 6, 121–128 (2009).
Kocher, B. & Piwnica-Worms, D. Illuminating Cancer Systems with Genetically Engineered Mouse Models and Coupled Luciferase Reporters In Vivo . Cancer Discov. 3 (2013).
Hänsch, G. M., Hammer, C. H., Vanguri, P. & Shin, M. L. Homologous species restriction in lysis of erythrocytes by terminal complement proteins. Proc. Natl. Acad. Sci. USA 78, 5118–21 (1981).
Schwab, G. E. & Reeves, P. R. Comparison of the bactericidal activity of different vertebrate sera. J. Bacteriol. 91, 106–112 (1966).
Rowley, D. Sensitivity of rough gram-negative bacteria to the bactericidal action of serum. J. Bacteriol. 95, 1647–1650 (1968).
Popiel, I. & Turnbull, P. C. B. Passage of Salmonella enteritidis and Salmonella thompson through chick ileocecal mucosa. Infect. Immun. 47, 786–792 (1985).
Stone, B. J. et al. Identification of novel loci affecting entry of Salmonella Enteritidis into eukaryotic cells. J Bacteriol 174, 3945–3952 (1992).
Aabo, S. et al. Development of an In Vivo Model for Study of Intestinal Invasion by Salmonella enterica in Chickens. 68, 7122–7125 (2000).
Collier-Hyams, L. S. et al. Cutting Edge: Salmonella AvrA Effector Inhibits the Key Proinflammatory, Anti-Apoptotic NF- B Pathway. J. Immunol. 169, 2846–2850 (2002).
Barrow, P. A. Salmonella infections: immune and non-immune protection with vaccines. Avian Pathol. 36, 1–13 (2007).
Fields, P. I., Swanson, R. V., Haidaris, C. G. & Heffron, F. Mutants of Salmonella Typhimurium that cannot survive within the macrophage are avirulent. Proc. Natl. Acad. Sci. USA 83, 5189–93 (1986).
Shivaprasad, H. L. Fowl typhoid and pullorum disease. Rev. Sci. Tech. 19, 405–24 (2000).
Wigley, P., Jones, M. A. & Barrow, P. A. Salmonella enterica serovar Pullorum requires the Salmonella pathogenicity island 2 type III secretion system for virulence and carriage in the chicken. Avian Pathol. 31, 501–6 (2002).
Boyd, J. F. Pathology of the alimentary tract in Salmonella Typhimurium food poisoning. Gut 26, 935–44 (1985).
Wigley, P. et al. Infection of the reproductive tract and eggs with Salmonella enterica serovar Pullorum in the chicken is associated with suppression of cellular immunity at sexual maturity. Infect. Immun. 73, 2986–90 (2005).
Humphrey, T. J., Baskerville, A., Mawer, S., Rowe, B. & Hopper, S. Salmonella Enteritidis phage type 4 from the contents of intact eggs: a study involving naturally infected hens. Epidemiol Infect 103, 415–423 (1989).
Gillespie, I. & Elson, R. Successful reduction of human Salmonella Enteritidis infection in England and Wales. Euro Surveill 10, E051117.2 (2005).
Lalsiamthara, J. & Lee, J. H. Virulence Associated Genes-Deleted Salmonella Montevideo Is Attenuated, Highly Immunogenic and Confers Protection against Virulent Challenge in Chickens. Front. Microbiol. 7, 1634 (2016).
Lalsiamthara, J., Gogia, N., Goswami, T. K., Singh, R. K. & Chaudhuri, P. Intermediate rough Brucella abortus S19Deltaper mutant is DIVA enable, safe to pregnant guinea pigs and confers protection to mice. Vaccine 33, 2577–2583 (2015).
Qureshi, M. A., Dietert, R. R. & Bacon, L. D. Genetic variation in the recruitment and activation of chicken peritoneal macrophages. Proc Soc Exp Biol Med 181, 560–568 (1986).
Higgins, S. E. et al. Effect of probiotic treatment in broiler chicks on intestinal macrophage numbers and phagocytosis of Salmonella Enteritidis by abdominal exudate cells. Poult. Sci. 86, 2315–2321 (2007).
Matsuda, K., Chaudhari, A. A., Kim, S. W., Lee, K. M. & Lee, J. H. Physiology, pathogenicity and immunogenicity of lon and/or cpxR deleted mutants of Salmonella Gallinarum as vaccine candidates for fowl typhoid. Vet. Res. 41, 59 (2010).
Desmidt, M., Ducatelle, R. & Haesebrouck, F. Research notes: Immunohistochemical observations in the ceca of chickens infected with Salmonella Enteritidis phage type four. Poult. Sci. 77, 73–4 (1998).
Author information
Authors and Affiliations
Contributions
J.L. and J.H.L. conceived and designed the experiments. J.L. performed the experiments. J.L. and J.H.L. analyzed the data and wrote the manuscript. Both authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lalsiamthara, J., Lee, J. Pathogenic traits of Salmonella Montevideo in experimental infections in vivo and in vitro. Sci Rep 7, 46232 (2017). https://doi.org/10.1038/srep46232
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep46232
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.