Abstract
Crassostrea virginica is one of the most common estuarine bivalves in the United States’ east coast and is frequently found in archaeological sites and sub-fossil deposits. Although there have been several sclerochronological studies on stable carbon and oxygen isotopes in the shells of this species, less is known about δ15N values within their shells, which could be a useful paleoenvironmental proxy to assess estuarine nitrogen dynamics. Modern C. virginica samples were collected in Chesapeake Bay for comparison with archaeological shells from nearby sites ranging in age from ~100 to 3,200 years old. Left valves were sampled by milling the hinge area and the resulting powder was analyzed for %N and δ15N values. Comparison of δ15N values between C. virginica shells shows relatively constant values from ~1250 BC to ~1800 AD. After ~1800 AD, there are rapid increases in 15N enrichment in the shells, which continue to increase in value up to the modern shell values. The increase in δ15N values is evidence of early anthropogenic impact in Chesapeake Bay. These results corroborate the observation that coastal nitrogen pollution occurred earlier than the 19th century and support the use of oyster shell δ15N values as a useful environmental proxy.
Similar content being viewed by others
Introduction
Anthropogenic pollution, especially nitrogen, is a major concern in coastal estuaries1,2,3. Increased nitrogen loading can potentially lead to eutrophication, which can cause anoxic conditions in water bodies and die-off events of marine organisms in dead zones4,5,6. Since the 1970s, regulations governing nitrogen inputs have been introduced, but in order for these policies to be effective, it is imperative to know base levels of nitrogen in the area. In most cases, however, base levels before anthropogenic influences began are unknown. If mollusk shells serve as a paleoenvironmental proxy for environmental N levels using stable nitrogen isotope (δ15N) values, scientists could generate a base level of nitrogen by studying the record of nitrogen isotopes stored in the shells, as well as how nitrogen sources and concentrations have fluctuated over time.
Bivalve shell geochemistry is commonly used to assess environmental parameters at the time the shell grew, such as sea surface temperature and salinity7,8,9,10,11,12. The eastern oyster Crassostrea virginica has large environmental tolerances; able to survive significant variations in temperature (20–45 °C), salinity (0–42 psu), and food supply13. Since they are sessile filter feeders in physical contact with water, their soft tissues and shells can record environmental changes by incorporating organic materials from the surrounding water body14,15. C. virginica shells are primarily calcitic, but have a thin aragonite layer in the ligostracum and muscle scar16. This species has previously been used for numerous stable isotope studies on stable carbon and oxygen isotopes within the shell (e.g. refs 17, 18, 19, 20), and recently has been introduced as a possible paleoenvironmental proxy for environmental N levels by analyzing the shell δ15N values15,21,22,23. Anthropogenic nitrogen sources, specifically sewage and industrial pollutants, are typically more 15N enriched than non-anthropogenic sources24,25,26, making it possible to trace nitrogen inputs into estuaries by studying the δ15N values in the tissue and shell organic fraction.
Unlike the soft tissue samples that were traditionally used in biological and environmental studies of mollusks, shell material can potentially serve as a better proxy for past conditions due to the increased chance of preservation over time. Mollusk shell material accretes sequentially, sometimes in annual growth layers, which could potentially be used as a high resolution temporal proxy for historical nitrogen levels, as well as creating a time-series of nitrogen loading over the life of the organism11,26,27. Furthermore, the shell material is never metabolized, so it can record the environmental conditions over a longer period of time than soft tissues12,26,27. It has previously been established that the calcite shell organic fraction chemistry reflects that of the organisms’ soft tissues, with minimal variability in δ15N values (~0–2‰) between the soft tissues and shell material27,28.
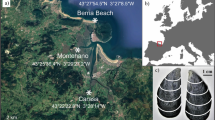
Here we determine if C. virginica shells record historical evidence of N levels in the Rhode River basin in Chesapeake Bay during the late Holocene to assess when anthropogenic N loading began. In doing so, we seek to better validate and establish this novel environmental proxy. To test for Chesapeake Bay nitrogen in historic and prehistoric mollusk shells, we sampled archaeological and modern (living) oysters from the Rhode River Estuary at the Smithsonian Environmental Research Center in Edgewater, Maryland (38°53′N 76°32′W) (Fig. 1). Our archaeological samples date to the Woodland Period (including the Early [1200–500 BC], Middle [500 BC-AD 900], and Late [AD 900–1600]), and the 18th and 19th centuries21. During the Woodland period in the Chesapeake, Native American peoples practiced a largely hunting and gathering lifestyle and their populations appeared to expand during the Middle Woodland and especially during the Late Woodland, when maize agriculture appeared in parts of the region29,30,31. European colonization intensified in the area in the 17th century, with colonial settlements clearing the forests and establishing large plantations on pristine agricultural land32,33. During the 19th century, industrialization rapidly increased in the eastern US and continues today33.
Sample locations are represented by their corresponding archaeological time periods. The site labeled “Dock” denotes the location of modern oyster sampling. Map was created in ArcMap version 10.3 and modified in Adobe Illustrator version CC (http://desktop.arcgis.com/en/arcmap/10.3/main/get-started/whats-new-in-arcgis.htm and http://www.adobe.com/products/illustrator.html, respectively).
Although Native Americans practiced agriculture and cleared forests in the region, these impacts were more localized and less widespread than the major landscape clearance efforts of Euro-American colonists and later peoples from the 17th century onward. Historic period land clearance resulted in significant sedimentation34, and likely nutrient loading, into the Bay. These data lead Kirby and Miller34 to hypothesize that an increase in observed growth rates of oysters from archaeological sites on the Potomac and Patuxent rivers during the 18th and early 19th century may have been due to an increase in planktonic primary productivity from nutrient loading during early eutrophication. The initial period of faster growth was later followed by a size decrease that they speculate was from intensive eutrophication and environmental deterioration, such as increased occurrences of harmful algae and parasitic disease after AD 1860. They speculate that other environmental changes, such as changes in water chemistry and an increase in the degree of fishing disturbances, in Chesapeake Bay during this time could affect the growth rate of C. virginica, but these environmental conditions are unlikely to cause as significant of an effect in shell growth rates as eutrophication. Our δ15N data provide a means to test this hypothesis and to help develop a novel proxy for assessing N in marine shellfish.
Results
One-way ANOVA tests for δ15N values between time periods produced an F-value of 297.3 and a significance of p < 0.001 at α = 0.05 (data summarized in Table 1). This indicates that the sample variance between different time periods was greater than the variance within time periods. A Tukey Post Hoc test yielded results that show that archaeological samples from the Early Woodland (1250–800 BC) are significantly different from Middle Woodland (500 BC-AD 900), 19th century, and modern samples. Archaeological samples from the Middle Woodland were significantly different from Early Woodland, latter 19th century, and modern samples. Archaeological samples from the Late Woodland (AD 900–1600), 18th and early 19th century were significantly different from latter 19th century and modern samples. Later 19th century and modern shells were significantly different from all other time periods (Fig. 2).
The F-value calculated by a one-way ANOVA for percent nitrogen (%N) was 34.8 with a significance of p < 0.001 at α = 0.05. A Tukey Post Hoc test yielded results that show archaeological samples from the Early, Middle, and Late Woodland periods were significantly different only from the modern samples (data summarized in Table 2). The 18th and 19th century samples were only significantly different from the modern samples and the modern samples were significantly different from all other time periods (Fig. 3).
Discussion
Our results indicate increased N loading to Chesapeake Bay started as early as the 19th century and substantially increased to the present. Time-series comparisons of the sampled shells display distinct trends, with both %N and δ15N values following a roughly exponential curve with higher concentrations of N and more 15N enriched shells occurring in the 19th century and modern samples. The %N remains more constant than the δ15N values, but increases ~0.07% from the 18th century to modern shells (Fig. 3). The δ15N values remain relatively constant from the Early Woodland until the first part of the late 18th to 19th century (i.e., 1750–1800) but then increase ~3‰ from the late 18th century to mid late 19th century (i.e., 1850–1900) (Fig. 2). There was another substantial increase in δ15N values (~1‰) between the mid to late 19th century shells and the modern collected shells, which is correlated to a 10-fold increase in reactive N species globally from 1860 to 199035.
δ15N values in mollusk shells have been shown to faithfully track the values in soft tissues and ambient POM26,27,36. In contrast, variation in mollusk shell %N has not been validated as a meaningful proxy for ambient N concentrations and additional analysis of modern samples is required before interpretation of such data is possible. Although the relatively constant %N suggests little to no N loss from the shells over time, our discussion will focus solely on δ15N values.
We interpret this substantial increase in δ15N values to be increased anthropogenic inputs into Chesapeake Bay during the 1800s. During the Woodland Period, it is unlikely that Native Americans37 had any significant impact on the N loading of the bay. While there was urban development during the 18th century, the impacts were likely more local in scale, as population levels were comparatively low in much of the region.
Our results are similar to several sediment nutrient loading studies in Chesapeake Bay over the last ~3000 years, with the exception of when δ15N values began to rapidly increase within the Bay38. Bratton et al.35 observed significant increases in δ15N values from 1750–1800 AD, whereas we determined a later time period of 15N enrichment. This is likely due to sampling location, however, since Bratton et al.’s study locations were closer to the Susquehanna River, which is a larger source of N for Chesapeake Bay35 than our study location. Zimmerman and Canuel38 also show increases in δ15N values beginning between the 18th and 19th centuries depending on study location within Chesapeake Bay, with their more northerly collected sediment core showing 15N enrichment ~100 years before their southern core. A study location farther north in the bay would also not have as long of a period to mix with estuarine waters before deposition, so it could potentially record higher δ15N values at an earlier time period. Another possibility for the timing differences between this study and Bratton et al.35 is due to the dating methods used. While our study solely used 14C from corrected and calibrated archaeological oysters21, Bratton et al.35 used pollen stratigraphy, 14C of shell material within the cores, and the short-lived radioisotopes 137Cs and 210Pb within recently deposited sediment4. Since pollen was primarily used to relatively date the sediment, it is possible that the absolute dates obtained by their 14C analysis could permit larger error in the dating due to reworking and bioturbation of the sediments.
During the 19th century, population sizes and industrialization in the northeast US expanded exponentially39, which likely had a profound impact on the ecological health of the Bay. Between 1830 and 1880, over 80% of the forest surrounding Chesapeake Bay had been cleared, which, in combination with plowing for agriculture, greatly increased the sediment accumulation in the bay4,5,20,35,39,40,41,42. During this time, the population size nearby Chesapeake Bay increased threefold39, and the increased amounts of sewage discharge and erosion caused by plowing likely increased the N inputs and 15N enrichment in the Bay24. The significant decrease in oyster populations at this time also contributed to the failing health of the Bay due to decreased filtration rates of the bay water39 and increased sedimentation rates which correlates to previous findings of decreased diatom diversity and increase in the centric:pennate diatiom ratio since the beginning of the 19th century4. Therefore, it is likely that the POM and sediment of the bay were more enriched in 15N. Consequently, we would expect a substantial shift in δ15N values during the 19th century due to increased amounts of sewage and eroded soil entering the bay, which was evident in the results of our shell study.
A similar pattern was present in size changes in archaeological oysters. Archaeological size data from throughout the Chesapeake Bay indicate a significant increase in oyster size during the Historic period (18th to 19th centures) when compared to earlier Woodland times34,37. Research at sites on the Potomac and Patuxent rivers documents this trend in detail, with oysters first increasing in size during the 18th and early 19th centuries and then decreasing dramatically after AD 1860, perhaps from intensive eutrophication34. It has been suggested that the degree of organic matter loading into Chesapeake Bay has significantly increased within the last two centuries38,43,44, however, Cornwell et al. did not determine substantial increases in N deposition within the sediment, which could be due to increased algal blooms in the water column38,44 and metabolic activity of estuarine organisms43. Our data support this hypothesis, providing the first direct evidence of N loading in this location of the bay during the 19th century, particularly in the latter half of the 19th century.
The potential effects of diagenetic alteration of %N and δ15N values must be considered. Loss of shell organic matter, and consequently N, likely occurs after burial and may be evident in the %N trends noted in our study. We argue, however, that such loss would not explain the observed δ15N trends. First, the observed trend in δ15N values would require preferential loss of 15N, which is contradictory to the kinetics of stable isotopes. Secondly, it is difficult to imagine why diagenetic influence of N isotopes would stop in pre-seventeenth century shells, and even reverse to some extent in the earliest samples. Third, Black28 was not able to influence shell δ15N values during a series of prehistoric cooking methods studies unless the shell was heated to the point at which it began to physically disintegrate. Finally, the close correspondence between our data and the sediment core-based proxies of anthropogenic N noted above indicates that the shell data trends accurately follow levels of environmental N. We argue that the trends in δ15N values noted in this study are primarily the result of changes in the sources contributing to the oysters’ habitat. The source for the somewhat higher δ15N values in the Early Woodland samples is not readily apparent.
This study demonstrates the potential utility of shell δ15N proxies. The abundance of this and similar species in archaeological sites worldwide suggests that detailed records of anthropogenic N impacts can be created from shell midden deposits. Such baseline data are valuable not only in addressing modern pollution concerns, but also in assessing the extent of ancient human habitation, land-use, agricultural practice, and related activities. This approach may be less expensive than sediment core analysis and therefore permit wide geographic coverage to facilitate regional N reconstructions. Additionally, archaeological contexts may allow reliable age control of samples, which is sometimes difficult in cores with disturbed sediments. Sclerochronological shell sampling also permits seasonal analysis of δ15N values in large, fast growing species27, which provides even more detailed information. Further research is needed to better assess the validity of shell %N data, the impacts of diagenesis and prehistoric cooking methods, and the possible transport of shells before burial.
For the δ15N values in C. viriginica shells from Chesapeake Bay, there are significant changes in δ15N values of the shells over time. The δ15N values remain relatively constant from ~3,200 years ago until 1750–1800, but significantly increase after 1850 to the live-collected shells. While there is a possibility that diagenetic processes may have influenced these data, the sharp increase in δ15N values between ~1750–1800 and 1850-1900 appears to be primarily due to the industrial revolution, animal wastes, and soil erosion. The subsequent increases from 1850 to 2013 are due to a significant increase in human population, synthetic fertilizer use, sewage discharge, erosion, and other anthropogenic pollution sources in Chesapeake Bay over the last two centuries. Different N pollutants have varying δ15N values (i.e., animal wastes range from ~10–20‰ and synthetic fertilizer ~0‰), which explains the large increase in δ15N values between ~1750–1800 and 1850–1900 and the smaller increase from ~1850–1900 to modern samples. Between ~1750–1800 and 1850–1900, the main N pollutants in Chesapeake Bay were isotopically enriched, leading to a significant increase in δ15N values from earlier periods. The widespread use of synthetic fertilizers in the 20th century likely caused the reduced increase in δ15N values between ~1850–1900 to modern samples since synthetic fertilizer has more depleted δ15N values than earlier sources while increasing the %N in the Bay.
Methods
Oyster shell samples were collected from six different archaeological sites within the Rhode River Estuary of Chesapeake Bay (Fig. 1). Researchers with the Smithsonian Institution collected archaeological samples while excavating shell middens in the region21,37. Modern oyster samples were collected from an oyster reef located adjacent to one of the archeological sites and in relatively close proximity to the other archaeological sites.
Ninety archaeological shell samples ranging in age from ~100 to 3,200 years old and thirty modern shell samples were analyzed for %N and δ15N values. The ancient shells were all well preserved and robust (i.e., limited taphonomic effects, no noticeable alteration of color of the shell or ligament scar, and non-chalky texture). The soft tissues were removed from the modern shells and the shells were cleaned using DI water and a soft bottlebrush followed by five minutes of ultrasonic cleaning. The shells were dried overnight at room temperature. The left valve of each shell sample was bisected along the longest axis using a slow-speed diamond-wafering saw. Each shell was subsampled using a hand held micro-drill. In order to create a lifetime average of the %N and δ15N values, a transect was drilled in cross-section through the resilifer to a depth of approximately 2 mm (Fig. 4). No carbonate powder from the exterior surface of any valve was sampled. Gillikin et al.27 has shown that the oxidative cleaning required for coral and foraminifera samples is not required for dense bivalve shells. The powdered samples were stored in 4.5 mL round-bottomed borosilicate vials until elemental analyzer –isotope ratio mass spectrometry (EA-IRMS) analysis.
To obtain comparable amounts of nitrogen (i.e., mass spectrometer peak area in millivolts (mV)) between the modern and archaeological shell samples, different masses were used. Between 34–35 mg of powdered samples of modern shells were transferred to 5 × 9 mm tin capsules and 100–110 mg of archeological shells were transferred to 9 × 10 mm tin capsules. The samples were analyzed in the Alabama Stable Isotope Laboratory, in the Department of Geological Sciences at the University of Alabama. Determinations for %N and δ15N values were made using a Costech ECS 4010 elemental analyzer (EA) coupled to a Thermo Delta V isotope ratio mass spectrometer without acid pre-treatment of the samples following Versteegh et al.45. The EA was fitted with a carbon trap [glass trap (Costech part no. 071121) filled with CO2 absorbant (Costech part no. 021020)] to remove CO2 and operated with a combustion temperature of 1020 °C (reduction column = 650 °C).
The δ15N data are reported in parts per mil (‰) vs. AIR. Delta notation is calculated using the equation δ15N = [(Rsample − Rstandard)/Rstantard] * 1000. Precision (1σ) was better than 0.09‰ based on analysis of two standards over a range of isotopic values (ammonium sulfate standard IAEA-N-2; δ15N = 20.3‰) and Elemental Microanalysis high organic sediment standard B2151; δ15N = 4.4‰). Two elemental standards, acetanilide (%N = 10.36%) and B2151 (%N = 0.5%), were used to build the calibration curve for each run and check for accuracy, respectively. Thirty-four samples were analyzed in duplicate with precision better than 0.07‰. One-way ANOVA and Tukey Post Hoc tests were completed on the shell samples based on time period to determine if there were any statistically significant differences between periods over time.
Additional Information
How to cite this article: Black, H. D. et al. δ15N Values in Crassostrea virginica Shells Provides Early Direct Evidence for Nitrogen Loading to Chesapeake Bay. Sci. Rep. 7, 44241; doi: 10.1038/srep44241 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Fisher, D. & Oppenheimer, M. Atmospheric nitrogen deposition and the Chesapeake Bay estuary. Ambio. 20, 102–108 (1991).
McClelland, J. W. & Valiela, I. Linking nitrogen in estuarine producers to land-derived sources. Limnology and Oceanography. 43, 577–585 (1998).
McKinney, R. A., Lake, J. L., Charpentier, M. A. & Ryba, S. A. Using mussel isotope ratios to assess anthropogenic nitrogen inputs to freshwater ecosystems. Environmental Monitoring and Assessment. 74, 167–192 (2002).
Cooper, S. R. & Brush, G. S. A 2,500-year history of anoxia and eutrophication in Chesapeake Bay. Estuaries. 16, 617–626 (1993).
Howarth, R. W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae. 8, 14–20 (2008).
Watanabe, S., Kodama, M. & Fukuda, M. Nitrogen stable isotope ratio in the Manila clam, Ruditapes phillippinarum, reflects eutrophication levels in tidal flats. Marine Pollution Bulletin. 58, 1447–1453 (2009).
Epstein, S., Bucksbaum, R., Lowenstam, H. A. & Urey, H. C. Revised carbonate-water isotopic temperature scale. Geological Society of America Bulletin. 64, 1315–1326 (1953).
Emiliani, C., Cardini, L., Mayeda, T., McBurney, C. B. M. & Tongiorgi, E. Paleotemperature analysis of marine molluscs (food refuse) from the site of Arene Candide Cave, Italy and the Haua Fteah Cave, Cyrenaica. In: Isotopic and cosmic chemistry(ed. Craig, H., Miller, S. L. & Wasserburg, G. J. ). (North Holland, Amsterdam, 1964).
Grossman, E. L. & Ku, T. I. Oxygen and carbon isotope fractionation in biogenic aragonite: Temperature effects. Chemical Geology. 59, 59–74 (1986).
Bailey, G. N., Deith, M. R. & Shackleton, N. J. Oxygen isotope analysis and seasonality determinations: Limits and potential of a new technique. American Antiquity. 48, 390–398 (1983).
Jones, D., Quitmyer, I., Arnold, W. & Marelli, D. Annual shell banding, age, and growth rate of hard clams (Mercenaria spp.) from Florida. Journal of Shellfish Research. 9, 215–225 (1990).
O’Donnell, T. H., Macko, S. A., Chou, J., Davis-Hartten, K. L. & Wehmiller, J. F. Analysis of δ13C, δ15N, and δ34S in organic matter from the biominerals of modern and fossil Mercenaria spp . Organic Geochemistry. 34, 165–183 (2003).
Galtsoff, P. S. The American oyster: Crassostrea virginica Gremlin. Fishery Bulletin of the Fish Wildlife Service. 64, 1–480 (1964).
Dame, R. F., Spurrier, J. D. & Wolaver, T. G. Carbon, nitrogen, and phosphorus processing by an oyster reef. Marine Ecology Progress Series. 54, 249–256 (1989).
Carmichael, R. H., Annett, B. & Valiela, I. Nitrogen loading to Pleasant Bay, Cape Cod: Application of models and stable isotopes to detect incipient nutrient enrichment of estuaries. Marine Pollution Bulletin. 48, 137–143 (2004).
Andrus, C. F. T. & Crowe, D. E. Geochemical analysis of Crassostrea virginica as a method to determine season of capture. Journal of Archaeological Science. 27, 33–42 (2000).
Andrus, C. F. T. Isotope sclerochronology in southeastern US archaeology to estimate season of capture. In: Seasonality and human mobility along the Georgia Bight (ed. Reitz, E. J., Quitmyer, I. R. & Thomas, D. H. ) American Museum of Natural History Anthropological Papers(2012).
Harding, J. M., Spero, H. J., Mann, R., Herbert, G. S. & Silko, J. L. Reconstructing early 17th century estuarine drought conditions from Jamestown oysters. Proceedings of the National Academy of Sciences. 107, 10549–10554 (2010).
Thompson, V. & Andrus, C. F. T. Evaluating mobility, monumentality, and feasting at the Sapelo Island shell ring complex. American Antiquity. 76, 315–344 (2011).
Bronk Ramsey, C. Bayesian analysis of radiocarbon dates. Radiocarbon. 51, 337–360 (2009).
Rick, T. C. et al. Shell middens, cultural chronologies, and coastal settlement on the Rhode River sub-estuary of Chesapeake Bay, Maryland, USA. Geoarchaeology. 29, 371–388 (2014).
Surge, D., Lohmann, K. C. & Dettman, D. L. Controls on the isotopic chemistry of the American oyster, Crassostrea virginica: Implications for growth patterns. Palaeogeography, Palaeoclimatology, Palaeoecology. 172, 283–296 (2001).
Darrow, E. S., Carmichael, R. H., Andrus, C. F. T. & Jackson, H. E. From middens to modern estuaries, oyster shells sequester source-specific nitrogen. Geochimica Et Cosmochimica Acta. 202, 39–56 (2016).
Heaton, T. H. E. Isotopic studies of nitrogen pollution in the hydrosphere and atmosphere: A review. Chemical Geology. 59, 87–102 (1986).
Fritts, A. K., Fritts, M. W., Haag, W. R., DeBoer, J. A. & Casper, A. F. Freshwater mussel shells (Unionidae) chronicle changes in a North American river over the past 1000 years. Science of the Total Environment. 575, 199–206 (2017).
Carmichael, R. H., Hattenrath, T., Valiela, I. & Michener, R. H. Nitrogen stable isotopes in the shell of Mercenaria mercenaria trace wastewater inputs from watersheds to estuarine ecosystems. Aquatic Biology. 4, 99–111 (2008).
Gillikin, D. P. et al. High-resolution nitrogen stable isotope schlerochronology of bivalve shell carbonate-bound organics. Geochimica et Cosmochimica Acta. 200, 55–66 (2017).
Black, H. B. δ15N in mollusk shells as a potential paleoenvironmental proxy for nitrogen loading in Chesapeake Bay. (University of Alabama, 2014).
Dent, R. J. Chesapeake prehistory: Old traditions, new directions. (Plenum, New York, 1995).
Potter, S. R. Commoners, tribute, and chiefs: The development of the Algonquian culture in the Potomac Valley. (University Press of Virginia, 1993).
Gallivan, M. The archaeology of native societies in the Chesapeake: New investigations and interpretations. Journal of Archaeological Research. 19, 281–325 (2011).
Miller, H. M. Living along the “Great Shellfish Bay”: The relationship between prehistoric peoples and the Chesapeake. In Discovering the Chesapeake: The history of an ecosystem(ed. Curtin, D., Brush, G. S. & Fisher, G. ). (Johns Hopkins University Press, 2001).
Miller, H. M. Transforming a “splendid and delightsome land”: Colonists and ecological change in the Chesapeake 1607-1820. Journal of the Washington Academy of Sciences. 76, 173–187 (1986).
Kirby, M. X. & Miller, H. M. Response of a benthic suspension feeder (Crassostrea virginica gmelin) to three estuaries of anthropogenic eutrophication in Chesapeake Bay. Estuarine, Coastal, and Shelf Science. 62, 679–689 (2005).
Bratton, J. F., Colman, S. M. & Seal, R. R. Eutrophication and carbon sources in Chesapeake Bay over the last 2700 yr: Human impacts in context. Geochimica Et Cosmochimica Acta. 67, 3385–4402 (2003).
Fertig, B., Carruthers, T. J. B., Dennison, W. C., Fertig, E. J. & Altabet, M. A. Eastern oyster (Crassostrea virginica) δ15N as a bioindicator of nitrogen sources: Observations and modeling. Marine Pollution Bulletin. 60, 1288–1298 (2010).
Rick, T. C. et al. Millennial-scale sustainability of the Chesapeake Bay Native American oyster fishery. Proceedings of the National Academy of Sciences. 113, 6568–6573 (2016).
Zimmerman, A. R. & Canuel, E. A. Sediment geochemical records of eutrophication in the mesohaline Chesapeake Bay. Limnology and Oceanography. 47, 1084–1093 (2002).
Kemp, W. M. et al. Eutrophication of Chesapeake Bay: Historical trends and ecological interactions. Marine Ecology Progress Series. 303, 1–29 (2005).
Reimer, P. J. et al. IntCal09 and Marine09 radiocarbon age calibration curves, 0–50,000 years cal BP. Radiocarbon. 51, 1111–1150 (2009).
Galloway, J. N. et al. Nitrogen cycles: Past, present, and future. Biogeochemistry. 70, 153–226 (2004).
Cronin, T. et al. Climatic variability in the eastern United States over the past millennium from Chesapeake Bay sediments. Geology. 28, 3–6 (2000).
Cornwell, J. C., Conley, D. J., Owens, M. & Stevenson, J. C. A sediment chronology of the eutrophication of Cheasapeake Bay. Estuaries. 19, 488–499 (1996).
Zimmerman, A. R. & Canuel, E. A. A geochemical record of eutrophication and anoxia in Chesapeake Bay sediments: Anthropogenic influence on organic matter composition. Marine Chemistry. 69, 117–137 (2000).
Versteech, E. A. A., Gillikin, D. P. & Dehairs, F. Analysis of δ15N values in mollusk shell organic matrix by elemental analysis/isotope ratio mass spectrometry without acidification: An evaluation and effects of long-tern preservation. Communications in Mass Spectrometry. 25, 675–680 (2011).
Acknowledgements
We thank Alberto Perez-Huerta and Natasha Dimova for help with data analysis as well as the Smithsonian Institution and Smithsonian Environmental Research Center for providing modern and archaeological shell samples. Funding was provided by the University of Alabama and the Department of Geological Sciences Research and Travel Funds. Funding for instrumentation used in the project was from National Science Foundation Instruments and Facilities grant number EAR-0949303. The Alabama Stable Isotope Laboratory analyzed the samples at cost.
Author information
Authors and Affiliations
Contributions
The authors contributed equally in designing the study. T.R. provided archaeological samples and access to sites. W.J.L. analyzed samples and D.P.G. assisted with methodology improvements. H.D.B. and C.F.T.A. analyzed data and H.D.B. led the writing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Black, H., Andrus, C., Lambert, W. et al. δ15N Values in Crassostrea virginica Shells Provides Early Direct Evidence for Nitrogen Loading to Chesapeake Bay. Sci Rep 7, 44241 (2017). https://doi.org/10.1038/srep44241
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep44241
This article is cited by
-
Interpreting Past Human Diets Using Stable Isotope Mixing Models—Best Practices for Data Acquisition
Journal of Archaeological Method and Theory (2022)
-
Age and growth of Astarte borealis (Bivalvia) from the southwestern Baltic Sea using secondary ion mass spectrometry
Marine Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.