Abstract
Methicillin-resistant Macrococcus caseolyticus strains from bovine and canine origins were found to carry a novel mecD gene conferring resistance to all classes of β-lactams including anti-MRSA cephalosporins. Association of β-lactam resistance with mecD was demonstrated by gene expression in S. aureus and deletion of the mecD-containing island in M. caseolyticus. The mecD gene was located either on an 18,134-bp M. caseolyticus resistance island (McRImecD-1) or a 16,188-bp McRImecD-2. Both islands were integrated at the 3′ end of the rpsI gene, carried the mecD operon (mecD-mecR1m-mecIm), and genes for an integrase of the tyrosine recombinase family and a putative virulence-associated protein (virE). Apart from the mecD operon, that shared 66% overall nucleotide identity with the mecB operon, McRImecD islands were unrelated to any mecB-carrying elements or staphylococcal cassette chromosome mec. Only McRImecD-1 that is delimitated at both ends by direct repeats was capable of circular excision. The recombined excision pattern suggests site-specific activity of the integrase and allowed identification of a putative core attachment site. Detection of rpsI-associated integrases in Bacillus and S. aureus reveals a potential for broad-host range dissemination of the novel methicillin resistance gene mecD.
Similar content being viewed by others
Introduction
Macrococcus is evolutionarily closely related to the genus Staphylococcus, but possesses a distinctly smaller genome with a size of 2.1 Mb in the species M. caseolyticus1. There are currently eight species included in the genus Macrococcus, namely M. caseolyticus, M. equipercicus, M. bovicus, M. carouselicus, M. brunensis, M. hajekii, M. lamae and M. canis2,3,4. Macrococcus is mainly found commensally on the surface of the body of animals and has also been isolated from raw milk and dairy products5,6,7. Methicillin-resistant strains were reported for M. caseolyticus from chicken, bovine milk and humans1,8,9,10 and for other Macrococcus species from dogs11,12.
Methicillin resistance in Staphylococcus and Macrococcus is due to the production of an alternative penicillin-binding protein (PBP2a, also called PBP2′ or MecA, MecB, and MecC) that has low affinity for almost all β-lactams13,14. In the presence of drug concentrations that inactivate native PBPs through irreversible acylation, PBP2a conserves its transpeptidase activity allowing cell-wall biosynthesis to continue15. PBP2a is encoded by a structural mec gene so far identified as mecA, mecB and mecC, and its presence can be predicted phenotypically using a screen for cefoxitin or oxacillin resistance16. The mec gene is organized in an operon with its regulators mecR1 and mecI coding for an integral-membrane sensor/transducer and a transcriptional repressor, respectively17. The mec-mecR1-mecI gene complex is homologous to blaZ-blaR1-blaI controlling β-lactamase expression through an inducible proteolytic pathway18 and crosstalk between the systems has been observed19,20,21,22.
The classical mecA gene is carried by a unique class of mobile genetic elements, the staphylococcal cassette chromosome mec (SCCmec)23, originally found in methicillin-resistant S. aureus (MRSA)24. Since then, a wide structural diversity of SCCmec elements has been described in both coagulase-positive and coagulase-negative staphylococci that carry either the mecA or the mecC gene25. The SCCmec elements are site-specifically integrated into the chromosomal orfX gene, have characteristic flanking repeat sequences, a mec gene complex and cassette chromosome recombinase(s) (ccr) responsible for integration/excision of the element.
In Macrococcus, methicillin resistance has been associated with the mecB gene (formerly mecAm), a distantly related mecA homologue1. It is present in a mec gene complex (blaZm-mecB-mecR1m-mecIm) which includes the regulators and additionally the blaZ homologue blaZm. The mecB gene complex was originally found associated with transposon Tn6045 located on either large plasmids or integrated into the chromosome as part of a SCCmec-like element in M. caseolyticus1,8. Recently, a SCCmec carrying mecB independent of Tn6045 was reported in a hemolytic Macrococcus strain (M. canis sp. nov., formerly identified as M. caseolyticus)4,12.
Since 2014, several cases of bovine mastitis and an infection in a dog caused by methicillin-resistant mecB-negative M. caseolyticus strains were observed by our diagnostic unit at the Institute of Veterinary Bacteriology in Bern, Switzerland. This prompted us to investigate the nature of this β-lactam resistance by whole genome sequencing and gene expression which revealed the new methicillin resistance gene mecD on novel resistance islands.
Results and Discussion
Identification of a novel acquired PBP2a in M. caseolyticus
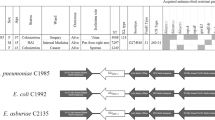
M. caseolyticus strains IMD0819 and IMD0473 were isolated from bovine mastitis milk and strain KM0211 from an infection site of a dog (Table 1). They all exhibited resistance to penicillin, cefoxitin and oxacillin but did not carry any of the described methicillin-resistance genes (Table 1). Whole genome sequencing and tblastn analysis using PBP2a encoded by mecA, mecB and mecC as queries revealed a novel mec gene homologue in the three β-lactam-resistant strains but not in the β-lactam-sensitive M. caseolyticus strain KM1352 (Table 1). The mec gene was designated mecD according to the SCCmec guidelines for reporting new mecA gene homologues which defines a new mec gene type if the nucleotide (nt) sequence shares less than 70% identity to any known mec genes13. The mecD gene was identical in strains IMD0819, IMD0473 and KM0211 and shared 69% nt identity and 63% amino acid (aa) identity with mecB of M. caseolyticus strain JCSC54021 and less than 62% nt and 53% aa identity with all other PBP2a of the Mec family (Fig. 1).
Evolutionary analysis was performed for nucleotide sequences using the UPGMA method in MEGA756. The percentage of nucleotide (nt) and amino acid (aa) identity between mecD and other mec genes was determined by sequence alignment with Clustral OMEGA [ http://www.ebi.ac.uk/Tools/msa/clustalo/]. *) mecC1 of S. xylosus S04009 does not encode a functional PBP2a due to a frameshift mutation close to the 5′ end of the gene.
Regulation and expression of mecD
The mecD gene complex
The mecD gene was preceded by two regulatory genes mecR1m and mecIm transcribed in the opposite direction. The gene organization was similar to regulated systems containing other mec structural genes or blaZ. Comparison of mecD regulatory genes with these systems demonstrated that they possessed the highest sequence similarity with those of mecB, namely 61% nt identity with the homologous sensor/transducer gene mecR1m and 70% with the transcriptional repressor gene mecIm of M. caseolyticus JCSC5402 (Supplementary Figs S1 and S2). The genomes of the mecD-containing M. caseolyticus strains contained no further mec/bla regulators and no homologous genes to the β-lactamase blaZ or the antirepressor mecR2. The absence of a β-lactamase was confirmed by negative results in the nitrocefin test.
Promoter-operator structure
Analysis of the 171-bp intergenic region between mecD and mecR1m-mecIm revealed a divergent promoter pair for bidirectional transcription in an overlapping 32-bp fragment (Fig. 2). Promoters were predicted using the program BPROM that recognizes −35 and −10 consensus sequences for the bacterial sigma factor 7026. In addition, an operator site was identified using the consensus sequence for mec/bla divergons defined by Garcia-Castellanos27 (Fig. 2). The operator site is located within the mecR1m and mecD promoter region suggesting that binding of one MecIm dimer can repress transcription of mecD and mecR1m-mecIm simultaneously. The perfect consensus sequence for the mec/bla divergon found in M. caseolyticus suggests possible control of mecD expression by other MecI/BlaI proteins as is the case in S. aureus where BlaI and MecI can exchange with each other20,28. In addition to the palindromic sequences present in the mec operator, perfect and imperfect inverted repeats were found in the region between the mecD promoter and start site that might function as regulatory sequence in genomic DNA or RNA transcript (Fig. 2).
The −10 and −35 promoter sequences are underlined. Start codons and ribosomal binding sites (RBS) are in bold type. The operator sequence is highlighted in green and inverted repeats marked by arrows. The consensus sequence [A/G]NATTACA[A/T]NTGTA [A/G][T/G]NT (with bases acceptable for one given position between square brackets and “N” for any base) was used to identity operator sequence recognized by MecI/BlaI repressors27. The sequence is shown for M. caseolyticus IMD0819 (Genbank acc. no KY013611, position 15264–15440).
mecD regulators: MecR1m and MecIm
Resistance to β-lactams mediated by blaZ and mecA is controlled through an inducible proteolytic signal transduction pathway in S. aureus18,29. The mecD regulators were therefore analyzed for the presence of aa-patterns and domains known to play a functional role in this pathway. MecR1m contained a putative C-terminal penicillin-binding domain (PBD) involved in β-lactam sensing (Ser315-Lys561). The three signature motifs conserved within PBPs/β-lactamases were also present30: motif 1 including the catalytic serine (SxxK) was found at aa position 369–372 (Ser-Thr-Tyr-Lys), motif 2 (SxN) at position 417–419 (Ser-Val-Asn) and motif 3 (KTG[T/S]) at position 504–507 (Lys-Thr-Gly-Thr). The protein also showed a classical zinc-binding motif (HExxH) at position 183–187 (His-Glu-Ile-Thr-His) and a potential autolytic cleavage site (Lys272-Arg273) for activation of the metalloproteinase according to BlaR18.
The MecIm repressor was predicted to contain an N-terminal DNA-binding domain (Met1-Val73) with a winged helix-turn-helix structure matching entries cl21459 and pfam12802 in the Conserved Domain Database (CDD) (https://www.ncbi.nlm.nih.gov/cdd/). The putative recognition helix (Ser41-Asn56) was the most conserved segment that shared 80% sequence identity with the recognition helix α3 of the MecI protein of S. aureus N31527, while the overall aa identity was much lower with only 47% identity (Supplementary Fig. S2). The potential cleavage site for MecIm repressor inactivation was found between Asn101-Phe102 corresponding to the sites demonstrated in staphylococcal MecI and BlaI18,29.
MecD
The mecD gene encodes a 678-aa protein with a C-terminal transpeptidase domain (Ser330-Glu678) (CDD: pfam00905 and cl21491) that contains the three signature motifs conserved within PBPs (see paragraph mecD regulators). Motif 1 was present at position 406–409 (Ser-Thr-Gln-Lys), motif 2 at position 465–467 (Ser-Asp-Asn) and motif 3 at position 605–608 (Lys-Thr-Gly-Thr) followed by an alanine typically found in class B PBPs. The N-terminal section consisted of a transmembrane-helix (Lys7-Leu25, N-terminus inside) as predicted using TMpred software (http://www.ch.embnet.org/) and a non-penicillin binding domain (nPBD) (Glu27-Leu329) with an N-terminal extension subdomain (Glu27-Ala140) (CDD: pfam05223) typically found in PBP2a. Compared to MecA, MecD showed a distantly related nPBD and a more conserved transpeptidase domain with 37% and 64% aa identity, respectively.
mecD -mediated resistance phenotype
Minimal inhibitory concentrations (MICs) of different classes of β-lactam antibiotics were determined for the three mecD-positive M. caseolyticus strains and the mecD-negative strains KM1352 and CCUG 15606 T (Table 2). As control, two mecD-deletion mutants of strain IMD0819 were included. These mutants had lost either an 18,134-bp (strain IMD0819_20) or a 20,907-bp (strain IMD0819_33) genomic element containing the mecD gene (see paragraph construction of McRImecD-1 deletion variants). Compared to the mecD-negative strains, the mecD-containing strains IMD0819, IMD0473 and KM0211 showed several fold higher MICs of penicillins including the penicillinase-resistant drugs oxacillin and temocillin. The MICs were also higher for 2nd (cefoxitin), 3rd (cefotaxime and ceftazidime) and 4th (cefepime) generation cephalosporins as well as for carbapenems (ertapenem, imipenem and meropenem). The MIC for the anti-MRSA cephalosporins, ceftobiprole and ceftaroline were also at least 16-fold (ceftobiprole) and 8-fold (ceftaroline) higher in the mecD-containing strains. The transpeptidase domain of MecD provides similar residues to those identified to be important for ceftobiprole binding within the active site region of MecA (Tyr446, Thr600, Met641)31, namely Phe449, Thr608, Met650. The Tyr to Phe substitution in MecD should not decrease ceftobiprole binding since only aromatic stacking interaction was observed between Tyr446 and ceftobiprole31. The reason for reduced susceptibility may be associated with other structural differences between MecA and MecD, e. g. the diverse nPBDs. The nPBD (also called allosteric domain) of MecA is involved in allosteric control of the transpeptidase’s active site32. Ceftaroline binds to allosteric sites in the nPBD, causing a rearrangement of salt bridges and predisposes MecA to acylation by a second β-lactam molecule32. The nPBD of MecD is only distantly related to that of MecA and neither equivalent residues for ceftaroline binding nor a similar scatter of charged aa are obvious which questions an analogue allosteric control mechanism for MecD.
The complete structure of mecR1m-mecIm suggests inducible mecD expression in the presence of β-lactams. To study mecD expression, two plasmids were constructed using S. aureus-E. coli shuttle vector pTSSCm33. Plasmid pTSSCm-D1 contained mecD with its upstream 171-bp intergenic region and plasmid pTSSCm-D2 contained the entire mecD operon including the regulator genes mecR1m and mecIm. Both plasmids were electroporated into S. aureus RN4220 and selected for tetracycline resistance encoded on the vector. Transformants were only readily obtained with pTSSCm-D2 which contained the regulated mecD operon but not with pTSSCm-D1 which carried unregulated mecD. RN4220 colonies harboring pTSSCm-D1 were only obtained using Chromagar MRSA II for selection. These colonies grew slowly when re-streaked on agar containing tetracycline and only recovered their normal growth after several passages. They presented high level β-lactam resistance after the first subculturing but the level of resistance decreased after subsequent passages (e.g. MIC of oxacillin dropped from 32 mg/L to 2 mg/L). These results indicated that constitutive mecD expression has a deleterious effect and expression level needs to be downregulated for growth in the absence of β-lactams in S. aureus. Similarly, instability of unregulated plasmid-carried mecA was also observed in S. aureus, where the presence of mec or bla regulators was demonstrated to stabilize mecA integrity and expression34.
RN4220 cells carrying unregulated mecD on the plasmid pTSSCm-D1 presented MICs of β-lactams all above the values measured for RN4220 alone or those containing the empty vector (Table 2). Cells harboring pTSSCm-D2 with the regulated mecD showed 8-fold increase in the MIC of cefoxitin and at least 4-fold increased values for oxacillin, ceftaroline and cefotaxime, but no increase of MIC for ceftobiprole, ertapenem and imipenem indicating that these β-lactams are poor inducer for mecD expression in S. aureus. Importantly, in the presence of mecD, cefoxitin MIC values were always measured above the EUCAST breakpoint defined for MRSA screening (MIC: R > 4)35, also confirming the production of a low affinity PBP2a in RN4220. Compared to S. aureus, phenotypic resistance mediated by mecD was remarkably higher in M. caseolyticus with all the tested β-lactams (Table 2). Production of MecD seems to be well adapted for M. caseolyticus and to be suboptimal for S. aureus. Expression of an additional PBP may interfere with cell wall metabolism and needs to be regulated. In S. aureus, MecI functions as a strong transcriptional repressor and prompt induction and expression of mecA is usually not achieved if it is solely controlled through the mecR1-mecI regulators19,20. Enhanced expression of β-lactam resistance is observed for mecA controlled through the blaZ regulators and also the anti-repressor mecR219,20,21,36. It might be possible that the mecD phenotype could be stronger in another S. aureus background than RN4220, that lacks both mecR2 and blaR1-blaI.
Characterization of chromosomal resistance islands carrying mecD
McRImecD-1
The mecD gene was located on a nearly identical 18,134-bp element (99.97% nt identity) in strains IMD0819 and KM0211. The element was designated M. caseolyticus resistance island mecD one (McRImecD-1). In both strains, McRImecD-1 was integrated into the 3′ end of the 30S ribosomal protein S9 gene (rpsI), flanked by imperfect extended direct repeats (DR1 and DR2) of either 160, 161 or 163 bp (Fig. 3). At the 5′ end, McRImecD-1 carried an open reading frame (orf) encoding an integrase (int) of the tyrosine recombinase family and two upstream divergent oriented orfs coding for putative DNA-binding proteins with helix-turn-helix (HTH) motifs, which belong either to the transcriptional Cro/C1-type (orf3) or to the excisionase/Xis family (orf4). The gene organization resembles the int-stl(-str)-xis structure found in SaPIs of S. aureus, but further characteristics of these phage-related chromosomal island like pri, rep, pif and terS genes37 were absent in McRImecD-1. Besides the mec operon, McRImecD-1 contained 17 predicted orfs, including one gene encoding a potential virulence factor (virE), genes for restriction-modification system (hsmRI and hsrRI) and for a putative DNA recombination-mediator protein (dprA) (Fig. 3). VirE contained the virulence-associated protein E domain (CDD: pfam05272) and shared 36% aa identity with the VapE protein encoded on SePIfusB-857 of S. epidermidis NTUH-85738. The DNA modification methyltransferase HsmMI and the restriction endonuclease HsrRI displayed 46 and 65% aa identity to those enzymes of the BsuBI/PstI type II system of B. subtilis, respectively39. DprA of McRImecD-1 was up to 51% identical to homologues of Bacillus species (WP_034289281) but only 30% identical to the native homologue of M. caseolyticus (GenBank: WP_012656740). DprA proteins are ubiquitously found in bacteria. In Bacillus subtilis, DprA has been shown to be involved in natural competence and mediating homologous recombination through recruitment of RecA to ssDNA40,41.
Comparison was performed with sequences of M. caseolyticus strains JCSC5402 (Genbank acc. no. NC_011999, position 215180–250630), IMD0473 (Genbank acc. no KY013610), IMD0819 (Genbank acc. no KY013611), KM0211 (Genbank acc. no KY013612) and KM1352 (Genbank acc. no KY013613) using Easyfig software57. Gray areas indicate regions with between 68% to 100% nucleotide sequence identity. Regions encompassing McRImecD-1, McRImecD-2 and McCIIMD0819 are indicated by horizontal black lines and flanking direct repeats (DRs) by vertical blue lines. The open reading frames (orfs) are represented by arrows: mecD, mecR1m and mecIm are shown in red, orfs encoding integrase (int) or transposase (tnp) are shown in yellow, reverse transciptases (rt) in beige, orfs associated with restriction-modification in green and virulence-associated orfs in mauve; the orfs occurring in all M. caseolyticus strains are shown in black and additional strain-specific orfs in blue. The primers used in this study are indicated by small black arrowheads (a), mecD-F; (b), mecD-R; (c), truA-F; (d), s66-R; (e), int-0819-F; (f), int-0473-F; (g), araC-F; (h), orf21-R; (i), orf20-F; (j), cop-R) and cleavage sites for restriction endonucleases HindIII (H3) and HincII (H2) and EcoRI (E1) by thin vertical lines.
McRImecD-1 represents a new element that shows only fragmentary sequence identity with GenBank entries. The fragment containing the int gene (position 7261–8482 in IMD0819, GenBank acc. no KY013611) shared 77% nt identity with a unique integrase in the chromosome of M. caseolyticus JCSC5402 (Fig. 3) (GenBank acc. no NC_011999) and in the draft genome of S. aureus 930918–3 (ABFA01000015.1). In addition, the genome of M. caseolyticus JCSC5402 and S. aureus 930918–3 shared 85 and 88% identity with a fragment containing the orf8 of McRImecD-1 (position 12182–12418 in IMD0819), respectively. The same fragment was also found with 81% identity in the draft genome of S. hominis LRKNS031 (LXRS01000085.1). The fragment containing the mec operon of McRImecD-1 (position 13185–17475 in IMD0819) shared 66% overall identity with the mecB-containing fragment of the plasmid pMCCL2 of M. caseolyticus JCSC5402 (AP009486.1), indicating a novel mec operon type13 on the new element McRImecD-1.
Downstream of McRImecD-1, strain IMD0819 contained 4 additional orfs (orf21-orf24) and another DR (DR3) not present in the other M. caseolyticus strains (Fig. 3). The 404-bp DR3 shared 91% identity overall with a 405-bp region containing DR2. The segment between DR2 and DR3 represented a 2,773-bp chromosomal island (CI) with three orfs and was called McCIIMD0819. Together, McRImecD-1 and McCIIMD0819 constituted a composite island in IMD0819. While only partial sequence similarity (coverage <20%) was found between McCIIMD0819 and Staphylococcus/Bacillus GenBank entries, McCIIMD0819 showed 97% overall identity to E. faecium strains UC7265 (JRHQ01000038.1) and UC7267 (ASAM01000028.1). The three orfs (orf21-orf23) of McCIIMD0819 encode hypothetical proteins: ORF21 contained a putative domain of bacteriocin-processing endopeptidases (CDD: cl00296) and ORF23 contained a domain for AraC family transcriptional regulators (CDD: COG3708). ORF24, situated downstream of McCIIMD0819, encoded a putative AAA family ATPase that shared 44% aa identity with a protein of a Bacillus species (WP_069304013).
McRImecD-2
McRImecD-2 consisted of a 16,188-bp insert in the chromosome of IMD0473. It was delimitated by the attachment (att) site defined at the 3′ end of rpsI (see paragraph excision and circularization of McRImecD-1-McCIIMD0819 subunits) and the s66 family peptidase gene (s66) identified as a core genome sequence in all other M. caseolyticus strains (Fig. 3). A DR at the right side of McRImecD-2 was missing as well as a chromosomal segment including the cop gene suggesting that deletion took place at this locus in IMD0473. The mecD-containing segment of McRImecD-2 was similar to that of McRImecD-1 (position 11280–17867 in IMD0819) comprising orf6 to orf14 (99.97% nucleotide identity) (Fig. 3). However, McRImecD-2 contained different integrase and virE genes with 77% identity and 75% identity to those of McRImecD-1 at the left side, and a completely different sequence on the right side, which carried a putative transposase and two possible reverse transcriptases (RTs). The transposase displayed similarity to those of the IS30 family (CDD: COG2826), and was also present in the genome of IMD0819. It shared up to 51% aa identity with transposases found in Staphylococcus and Enterococcus species. The RTs displayed the RT motifs 3, 4 and 5 and a domain typically found in bacterial retrotransposons and retrons (CDD: cd01646)42. Both proteins showed less than 40% aa identity with all other GenBank entries.
The chromosomally integrated structures of McRImecD-1 and McRImecD-2 were confirmed by HindIII and HincII restriction analysis of long-range PCR products spanning mecD with the 5′ end region and 3′ end region of the islands, respectively (Fig. 3). The amplicons were obtained using primer pairs mecD-R and truA-F and mecD-F and s66-R (Fig. 3 and Supplementary Table S1). The structure of the mecD-negative strain KM1352 was verified by EcoRI digest of the PCR product amplified with primers truA-F and s66-R.
Mobility of McRImecD-1 and McRImecD-2
Site-specific integrases associated with mecD
The integrase situated on the 5′ end of the mecD resistance islands may catalyze integration and excision of the elements. The integrases in McRImecD-1 and McRImecD-2 differed slightly from each other, sharing 81% aa identity. Both consisted of a 388-aa protein that contained a N-terminal SAM-like domain found in phage integrases (CDD: pfam14569) and the conserved residues described for tyrosine recombinase in the C-terminal section, including the active site tyrosine at position 367 and two invariant arginines at position 210 and 33443. The integrase of McRImecD-2 was 98% identical to the integrases found in the chromosome of M. caseolyticus JCSC5402 and in the draft genome of S. aureus 930918–3, both strains being negative for mecD. In JCSC5402, the integrase was associated with a unique sequence that encodes a putative type III restriction modification system and was also delimitated by DRs (Fig. 3). In S. aureus 930918–3 and the M. caseolyticus strains JCSC5402, IMD0819, KM0211 and IMD0473, the integrase was inserted downstream of the rpsI gene, suggesting a site-specific activity of the enzyme. Distantly related integrases that share up to 46% aa identity with the integrases of M. caseolyticus strains were also found next to the rpsI gene in Bacillus species (Fig. 4a). Compared with other integrases of the tyrosine recombinase family, mecD-associated integrases showed 40 to 43% identity to the integrases of SaPIbov1/2 and of uncharacterized inserts in S. intermedius, S. hominis and S. pseudintermedius (Fig. 4a). The analysis suggests that the integrases of mecD resistance islands have a common ancestor with rpsI-associated integrases of Bacillus and the potential to transfer genetic information to S. aureus and probably other staphylococci.
(a) Phylogenetic tree of integrases of the tyrosine recombinase family. Colored boxes group members that share homologous integration sites (rpsI, gene for 30S ribosomal protein S9; sodm, superoxide dismutase gene; bcat, gene for branched-chain amino acid aminotransferase; guaA, GMP synthetase gene; groEL, chaperonin gene; rpsR, gene for 30S ribosomal protein S18; nlpA, gene for component of ABC-type metal ion transport system; smrB, gene for SsrA-binding protein). Analysis was performed for amino acid (aa) sequences using the UPGMA method in MEGA756. Host strains and genetic elements if known are indicated. References for the used sequences can be found in Supplementary Table S3. (b) Putative core attachment sites recognized by rpsI-associated integrases. For M. caseolyticus strains, the imperfect direct repeats (DR) carrying the att consensus sequence are indicated. For all other species the att site found at the 3′ end of the rpsI gene is given. The rpsI stop codon is underlined. Positions that hold variant bases are unshaded. All species, except S. aureus N315 and M. caseolyticus KM1352, carry an integrase gene downstream of rpsI.
Excision and circularization of McRImecD-1-McCIIMD0819 subunits
Spontaneous formation of circular DNA molecules containing mecD was tested by PCR and sequencing using divergent primers specific for mecD and int (primers a and e/f in Fig. 3). Two PCR products were obtained with IMD0819, one product with KM0211 and none with the McRImecD-2 containing strain IMD0473 (Supplementary Table S4). PCR was also performed to detect the chromosomal segment remaining after excision of the McRImecD-1 subunits using convergent primers specific for truA and cop (primers c and j in Fig. 3) and a short elongation time to avoid amplification of the entire insert. The resulting fragments indicated one deletion in KM0211 and two deletion variants in IMD0819 (Supplementary Table S4). Furthermore, PCR products for possible excision of McCIIMD0819 in strain IMD0819 were obtained using divergent (primers g and h) and convergent primer (primers i and j) pairs placed inside and outside of McCIIMD0819, respectively. Sequencing results confirmed circularization of McRImecD-1, composite McRImecD-1-McCIIMD0819 and McCIIMD0819 caused by recombination between DRs. Consistently, the left DR was incorporated in the circular molecule and the right DR remained as a joining region on the chromosome (Supplementary Table S4). This pattern suggests site-specific as well as orientation-specific enzymatic activity most probably encoded by the integrase of the mecD resistance islands. Homologous recombination mediated by DprA protein also encoded on McRImecD-1 would lead to a random recombination of DRs. The absence of circular McRImecD-2 molecule in IMD0473 can be explained by the lack of flanking DRs. A 61-bp core att site representing a putative target for the integrase was found in the extended imperfect DR sequences. This consensus sequence includes the 3′ end of rpsI gene and was also found in S. aureus and Bacillus species (Fig. 4b). In M. caseolyticus, att-DR1 of IMD0819, KM0211 and IMD0473 were identical and specified to be part of the mecD resistance islands. On the other hand, att-DR2 of KM0211 included the displaced 3′ end of rpsI after McRImecD-1 insertion and was identical to the DR of the methicillin-sensitive strain KM1352. Notably, positions of mismatches clarified that cleavage for strand exchange must have taken place among the first 8 bases of the att sites (Fig. 4b).
Construction of McRImecD-1 deletion variants
To induce excision of McRImecD-1, cells were grown in the presence of subinhibitory concentrations of ciprofloxacin. This treatment has been reported to induce SOS response, excision of bacteriophages and the movement of SaPIs44,45. Using replica plating, two cefoxitin-susceptible clones were obtained from IMD0819 (2 of 251 clones tested) but none from KM0211 (0 of 445). Sequencing revealed excision of McRImecD-1 in the deletion mutant IMD0819_20 and the composite element McRImecD-1-McCIIMD0819 in the deletion mutant IMD0819_33. The chromosomal segments were joined by DR2 in IMD0819_20 and DR3 in IMD0819_33. Both strains had lost β-lactam resistance as confirmed by susceptibility measurement (Table 2).
Conclusions
The novel mecD gene has been demonstrated to confer resistance to all classes of β-lactams including anti-MRSA cephalosporins, ceftobiprole and ceftaroline in M. caseolyticus. A transfer to S. aureus may jeopardise the efficacy of the last generation cephalosporins in MRSA. The mecD gene was located on genomic islands McRImecD-1 and McRI-mecD-2 associated with a putative virulence gene and a site-specific integrase suggesting a potential for dissemination. Although a mechanism of horizontal gene transfer is not obvious due to the absence of genes for conjugative transfer or for interaction with phage packaging machinery, circular excisions containing mecD were observed. This characteristic resembles SCCmec elements that also do not encode genes for transfer but serine recombinases capable of element excision and circularization23,46. The integrase of the tyrosine recombinase family located on mecD-resistance islands is suggested to recognize a conserved core att site present at the 3′ end of rpsI gene. Conservation of this att site in Bacillus and Staphylococcus species and the detection of a similar rpsI-associated integrase in the S. aureus strain 930918–347 suggest a potential for mecD elements to be acquired also by Staphylococcus species.
The presence of novel genetic elements containing a new methicillin- resistance gene in clinical M. caseolyticus strains from animal origin emphasizes once again the potential of bacteria to adapt to novel environments and to resist antimicrobial selective pressure of β-lactam antibiotics, which are widely used in veterinary medicine.
Materials and Methods
Bacterial strains and growth conditions
The origin and characteristics of the M. caseolyticus strains used in this study are listed in Table 1. They were obtained from the diagnostic unit of the Institute of Veterinary Bacteriology at the University of Bern. The samples were taken by veterinarians for diagnostic purposes therefore not requiring ethical approval or a permit for animal experimentation according to the current Swiss legislation (Federal Animal Protection Law, 455 (https://www.admin.ch/opc/de/classified-compilation/20022103/index.html). Strains were routinely cultivated on trypticase soy agar plates containing 5% sheep blood (TSA-SB) (Becton, Dickinson and company, Franklin Lakes, NJ, USA) at 37 °C. Species identification was performed using matrix-assisted laser desorption/ionization time-of flight mass spectrometry (MALDI-TOF MS) (microflex LT, Bruker Daltonics, Bremen, Germany). The laboratory strains E. coli DH5α and S. aureus RN422048 were used for cloning and transformation experiments. They were cultivated in Luria-Bertani (LB) broth with shaking or on LB agar plates at 37 °C under aerobic conditions. The recombinant DH5α and RN4220 strains containing the S. aureus-E. coli shuttle vector pTSSCm33 or derived constructs were selected and routinely grown using 10 mg/l tetracycline in the growth medium.
DNA preparation and PCRs
Plasmid DNA and genomic DNA were isolated using the peqGOLD Plasmid Miniprep Kit I and the peqGOLD Bacterial DNA kit (Peqlab Biotechnologie GmbH, Erlangen, Germany), respectively. To improve lysis of M. caseolyticus, cells were first incubated in Solution I of the kit supplemented with 50 mg/l of lysostaphin (Sigma-Aldrich, St Louis, MO, USA) and 2 g/l of lysozyme (Roche Diagnostics, Rotkreuz, Switzerland) for 20 min at 37 °C. For analytical PCR reactions, FIREPol® DNA polymerase (Solis BioDyne, Tartu, Estonia) and GoTaq® Long PCR Master Mix (Promega, Madison, WI, USA) were used for short (<2.5 kb) and long amplicons (up to 20 kb), respectively. Insert amplifications for plasmid construction were performed using High-Fidelity DNA polymerases (Pfu DNA polymerase [Promega] or the Phusion Hot Start II High-Fidelity DNA polymerase [Thermo Fisher Scientific, Waltham, MA, USA]) according to the manufacturer’s instructions. All relevant primers used in this study are listed in Supplementary Table S1. The presence of mecD was confirmed by PCR using primers mecD-F (5′-TCCTTTAGCGATAGATGGTGAA) and mecD-R (5′-CTCCCATCTTTTCTCCATCCT).
Genome sequencing and analysis
M. caseolyticus strains KM1352, IMD0819, IMD0473 and KM0211 were sequenced using Illumina MiSeq technology. Genomic DNA was extracted using the UltraClean® Microbial DNA Isolation Kit (MO BIO Laboratories, Carlsbad, CA, USA). Library preparation and sequencing were performed according to the manufacturer’s standard protocols using MiSeq Reagent kit v2 (Illumina, Little Chesterfield, UK) at the Labormedizinisches Zentrum Dr. Risch, Bern-Liebefeld, Switzerland. Draft genomes were assembled de novo using Geneious version R9.1.5 (Biomatters, Auckland, New Zealand). Contigs were analyzed for the presence of antimicrobial resistance genes using BLAST and ResFinder49. To obtain larger scaffolds of region of interest (required for genome of IMD0819 and IMD0473), target contigs were aligned to the chromosome of M. caseolyticus JCSC5402 (GenBank: NC_011999) to identify adjacent contigs and gaps were filled by PCRs and Sanger sequencing (ABI PRISM 3100 genetic analyzer, Applied Biosystems, Foster City, CA, USA) (IMD0819: connection of contig1-contig21; IMD0473: connection of contig26-contig56-contig11). The genomic structure was subsequently confirmed by long-range PCR amplification and restriction analysis in all sequenced strains (Supplementary Table S2). Prodigal software for gene finding in prokaryotes was used to define orfs50. Annotation of the orfs was performed manually by BLAST homology and putative function of the translated orfs analyzed against Prosite entries51 and Conserved Domain Database (CDD)52. Spontaneous formation of circular DNA molecules and the chromosomal region remaining after excision was analyzed by PCR using specific divergent and convergent primer pairs and GoTaq® Long PCR Master Mix (Supplementary Table S4). If more than one PCR product was obtained, fragments were gel purified prior to Sanger sequencing.
Recombinant strains
Curing of mecD from M. caseolyticus IMD0819 was carried out using cells growing with shaking in LB broth and in the presence of subinhibitory concentrations of ciprofloxacin (0.1 or 0.025 mg/l) for 6 h at 37 °C. Dilutions were plated on TSA-SB and single colonies analyzed by replica plating using LB agar containing 5 mg/l cefoxitin for negative selection. Deletion of mecD-containing fragments in the susceptible clones was determined by PCR and Sanger sequencing.
Two recombinant plasmids for mecD expression were generated in E. coli DH5α and electroporated into S. aureus RN422053,54. Insert sequences were obtained from M. caseolyticus strain IMD0819 through PCR amplification and introduced into the vector pTSSCm. The mecD gene including its native promoter was amplified with primers mecD-XhoI-F and mecD-SpeI-R (Pfu polymerase) and the mecD-mecR1m-mecIm fragment was obtained using primers mec-XhoI-F and mecD-SpeI-R (Phusion Hot Start II High-Fidelity DNA polymerase) (Supplementary Table S1). The cloning primers carried a SpeI or a XhoI site in the 5′-overhang to facilitate ligation of the PCR products into the pTSSCm vector after restriction with XhoI and SpeI endonucleases. The new constructs were named pTSSCm-D1 (mecD) and pTSSCm-D2 (mecD-mecR1m-mecIm). Their structures were verified based on restriction digestion patterns and Sanger sequencing.
Antimicrobial susceptibility testing
MICs were determined in Müller-Hinton broth through the microdilution technique16 using Sensititre EUST and EUVSEC2 plates (Thermo Fisher Scientific). Additionally, MICs of oxacillin (OXA), cefoxitin (FOX), penicillin (PEN), ceftobiprole (CBP) and ceftaroline (CRL) were determined using serial two-fold dilutions ranging from 0.25 mg/l to 128 mg/l (for OXA, FOX and PEN) and 0.125 mg/l to 64 mg/l (for CBP and CRL). Stock solution of ceftobirole (BAL0009141, Batch: 08004R25F) (Basilea Pharmaceutica AG, Basel, Switzerland) was prepared as described55. The dephosphorylated active form of ceftaroline (U3, Batch: CI 148/09) (AstraZeneca, Cambridge, UK) was freshly dissolved in 0.1 M sodium phosphate pH 7.5 at 1 mg/ml prior use. The production of β-lactamase was tested on BBL™ DrySlide™ Nitrocefin (Becton, Dickinson and Company).
Data Availability
The nucleotide sequences of mecD resistance islands and flanking regions have been deposited in the GenBank under the accession number KY013611 for M. caseolyticus IMD0819, KY013612 for M. caseolyticus KM0211, KY013610 for M. caseolyticus IMD0473 and KY013613 for M. caseolyticus KM1352.
Additional Information
How to cite this article: Schwendener, S. et al. Novel methicillin resistance gene mecD in clinical Macrococcus caseolyticus strains from bovine and canine sources. Sci. Rep. 7, 43797; doi: 10.1038/srep43797 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Baba, T. et al. Complete genome sequence of Macrococcus caseolyticus strain JCSCS5402, [corrected] reflecting the ancestral genome of the human-pathogenic staphylococci. J. Bacteriol. 191, 1180–1190, doi: 10.1128/JB.01058-08 (2009).
Kloos, W. E. et al. Delimiting the genus Staphylococcus through description of Macrococcus caseolyticus gen. nov., comb. nov. and Macrococcus equipericus sp. nov., and Macrococcus bovicus sp. no. and Macrococcus carouselicus sp. nov. Int. J. Syst. Bacteriol. 48 Pt 3, 859–877, doi: 10.1099/00207713-48-3-859 (1998).
Mannerová, S. et al. Macrococcus brunensis sp. nov., Macrococcus hajekii sp. nov. and Macrococcus lamae sp. nov., from the skin of llamas. Int. J. of Syst. Evol. Microbiol. 53, 1647–1654, doi: 10.1099/ijs.0.02683-0 (2003).
Gobeli Brawand, S. et al. Macrococcus canis sp. nov., a skin bacterium associated with infections in dogs. Int. J. of Syst. Evol. Microbiol. Epub ahead of print, doi: 10.1099/ijsem.0.001673 (2016).
Giannino, M. L., Marzotto, M., Dellaglio, F. & Feligini, M. Study of microbial diversity in raw milk and fresh curd used for Fontina cheese production by culture-independent methods. Int. J. Food Microbiol. 130, 188–195, doi: 10.1016/j.ijfoodmicro.2009.01.022 (2009).
Fontana, C., Cappa, F., Rebecchi, A. & Cocconcelli, P. S. Surface microbiota analysis of Taleggio, Gorgonzola, Casera, Scimudin and Formaggio di Fossa Italian cheeses. Int. J. Food Microbiol. 138, 205–211, doi: 10.1016/j.ijfoodmicro.2010.01.017 (2010).
Zhong, Z. et al. Bacterial microbiota compositions of naturally fermented milk are shaped by both geographic origin and sample type. J. Dairy Sci. 99, 7832–7841, doi: 10.3168/jds.2015-10825 (2016).
Tsubakishita, S., Kuwahara-Arai, K., Baba, T. & Hiramatsu, K. Staphylococcal cassette chromosome mec-like element in Macrococcus caseolyticus . Antimicrob. Agents Chemother. 54, 1469–1475, doi: 10.1128/AAC.00575-09 (2010).
Cicconi-Hogan, K. M. et al. Short communication: Prevalence of methicillin resistance in coagulase-negative staphylococci and Staphylococcus aureus isolated from bulk milk on organic and conventional dairy farms in the United States. J. Dairy Sci. 97, 2959–2964, doi: 10.3168/jds.2013-7523 (2014).
Micheel, V. et al. Screening agars for MRSA: evaluation of a stepwise diagnostic approach with two different selective agars for the screening for methicillin-resistant Staphylococcus aureus (MRSA). Mil. Med. Res. 2, 18, doi: 10.1186/s40779-015-0046-1 (2015).
Rubin, J. E. & Chirino-Trejo, M. Inducibly cefoxitin-resistant Macrococcus-like organism falsely identified as methicillin-resistant Staphylococcus aureus on CHROMagar with oxacillin. J. Clin. Microbiol. 48, 3037–3038, doi: 10.1128/JCM.00519-10 (2010).
Gómez-Sanz, E., Schwendener, S., Thomann, A., Gobeli Brawand, S. & Perreten, V. First staphylococcal cassette chromosome mec containing a mecB-carrying gene complex independent of transposon Tn6045 in a Macrococcus caseolyticus isolate from a canine infection. Antimicrob. Agents Chemother. 59, 4577–4583, doi: 10.1128/AAC.05064-14 (2015).
Ito, T. et al. Guidelines for reporting novel mecA gene homologues. Antimicrob. Agents Chemother. 56, 4997–4999 (2012).
Hiramatsu, K. et al. Genomic basis for methicillin resistance in Staphylococcus aureus . Infect. Chemother. 45, 117–136, doi: 10.3947/ic.2013.45.2.117 (2013).
Pinho, M. G., de Lencastre, H. & Tomasz, A. An acquired and a native penicillin-binding protein cooperate in building the cell wall of drug-resistant staphylococci. Proc. Natl. Acad. Sci. USA 98, 10886–10891, doi: 10.1073/pnas.191260798 (2001).
CLSI. The Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 9th ed., vol. 32, no. 2. Approved standard M07-A9. (Clinical and Laboratory Standards Institute, 2012).
Peacock, S. J. & Paterson, G. K. Mechanisms of methicillin resistance in Staphylococcus aureus . Annu. Rev. Biochem. 84, 577–601, doi: 10.1146/annurev-biochem-060614-034516 (2015).
Zhang, H. Z., Hackbarth, C. J., Chansky, K. M. & Chambers, H. F. A proteolytic transmembrane signaling pathway and resistance to β-lactams in staphylococci. Science 291, 1962–1965, doi: 10.1126/science.1055144 (2001).
Ryffel, C., Kayser, F. H. & Berger-Bächi, B. Correlation between regulation of mecA transcription and expression of methicillin resistance in staphylococci. Antimicrob. Agents Chemother. 36, 25–31 (1992).
McKinney, T. K., Sharma, V. K., Craig, W. A. & Archer, G. L. Transcription of the gene mediating methicillin resistance in Staphylococcus aureus (mecA) is corepressed but not coinduced by cognate mecA and β-lactamase regulators. J. Bacteriol. 183, 6862–6868, doi: 10.1128/JB.183.23.6862-6868.2001 (2001).
Arêde, P., Ministro, J. & Oliveira, D. C. Redefining the role of the β-lactamase locus in methicillin-resistant Staphylococcus aureus: β-lactamase regulators disrupt the MecI-mediated strong repression on mecA and optimize the phenotypic expression of resistance in strains with constitutive mecA expression. Antimicrob. Agents Chemother. 57, 3037–3045, doi: 10.1128/AAC.02621-12 (2013).
Hackbarth, C. J. & Chambers, H. F. blaI and blaR1 regulate β-lactamase and PBP 2a production in methicillin-resistant Staphylococcus aureus . Antimicrob. Agents Chemother. 37, 1144–1149 (1993).
Katayama, Y., Ito, T. & Hiramatsu, K. A new class of genetic element, staphylococcus cassette chromosome mec, encodes methicillin resistance in Staphylococcus aureus . Antimicrob. Agents Chemother. 44, 1549–1555 (2000).
Chambers, H. F. & DeLeo, F. R. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat. Rev. Microbiol. 7, 629–641 (2009).
Shore, A. C. & Coleman, D. C. Staphylococcal cassette chromosome mec: recent advances and new insights. Int. J. Med. Microbiol. 303, 350–359, doi: 10.1016/j.ijmm.2013.02.002 (2013).
Solovyev, V. & Salamov, A. Automatic annotation of microbial genomes and metagenomic sequences. In Metagenomics and its applications in agriculture, biomedicine and environmental studies (Ed. Li, R. W. ). p., 61–78 (Nova Science Publishers, 2011).
García-Castellanos, R. et al. On the transcriptional regulation of methicillin resistance: MecI repressor in complex with its operator. J. Biol. Chem. 279, 17888–17896, doi: 10.1074/jbc.M313123200 (2004).
Lewis, R. A. & Dyke, K. G. MecI represses synthesis from the β-lactamase operon of Staphylococcus aureus . J. Antimicrob. Chemother. 45, 139–144 (2000).
Arêde, P. & Oliveira, D. C. Proteolysis of mecA repressor is essential for expression of methicillin resistance by Staphylococcus aureus . Antimicrob. Agents Chemother. 57, 2001–2002, doi: 10.1128/AAC.02510-12 (2013).
Sauvage, E., Kerff, F., Terrak, M., Ayala, J. A. & Charlier, P. The penicillin-binding proteins: structure and role in peptidoglycan biosynthesis. FEMS Microbiol. Rev. 32, 234–258, doi: 10.1111/j.1574-6976.2008.00105.x (2008).
Lovering, A. L. et al. Structural insights into the anti-methicillin-resistant Staphylococcus aureus (MRSA) activity of ceftobiprole. J. Biol. Chem. 287, 32096–32102, doi: 10.1074/jbc.M112.355644 (2012).
Otero, L. H. et al. How allosteric control of Staphylococcus aureus penicillin binding protein 2a enables methicillin resistance and physiological function. Proc. Natl. Acad. Sci. USA 110, 16808–16813, doi: 10.1073/pnas.1300118110 (2013).
Schwendener, S. & Perreten, V. New shuttle vector-based expression system to generate polyhistidine-tagged fusion proteins in Staphylococcus aureus and Escherichia coli . Appl. Environ Microbiol. 81, 3243–3254, doi: 10.1128/AEM.03803-14 (2015).
Katayama, Y., Zhang, H. Z., Hong, D. & Chambers, H. F. Jumping the barrier to β-lactam resistance in Staphylococcus aureus . J. Bacteriol. 185, 5465–5472 (2003).
EUCAST. The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. http://www.eucast.org Version 6.0 (2016).
Arêde, P., Milheiriço, C., de Lencastre, H. & Oliveira, D. C. The anti-repressor MecR2 promotes the proteolysis of the mecA repressor and enables optimal expression of β-lactam resistance in MRSA. PLoS Pathog. 8, e1002816, doi: 10.1371/journal.ppat.1002816 (2012).
Novick, R. P., Christie, G. E. & Penadés, J. R. The phage-related chromosomal islands of Gram-positive bacteria. Nat. Rev. Microbiol. 8, 541–551, doi: 10.1038/nrmicro2393 (2010).
Chen, H. J. et al. New structure of phage-related islands carrying fusB and a virulence gene in fusidic acid-resistant Staphylococcus epidermidis . Antimicrob. Agents Chemother. 57, 5737–5739, doi: 10.1128/AAC.01433-13 (2013).
Xu, G. L., Kapfer, W., Walter, J. & Trautner, T. A. BsuBI–an isospecific restriction and modification system of PstI: characterization of the BsuBI genes and enzymes. Nucleic Acids Res. 20, 6517–6523 (1992).
Kramer, N., Hahn, J. & Dubnau, D. Multiple interactions among the competence proteins of Bacillus subtilis . Mol. Microbiol. 65, 454–464, doi: 10.1111/j.1365-2958.2007.05799.x (2007).
Yadav, T. et al. Bacillus subtilis DprA recruits RecA onto single-stranded DNA and mediates annealing of complementary strands coated by SsbB and SsbA. J. Biol. Chem. 288, 22437–22450, doi: 10.1074/jbc.M113.478347 (2013).
Zimmerly, S. & Wu, L. An unexplored diversity of reverse transcriptases in bacteria. Microbiol. Spectr. 3, MDNA3-0058-2014, doi: 10.1128/microbiolspec.MDNA3-0058-2014 (2015).
Esposito, D. & Scocca, J. J. The integrase family of tyrosine recombinases: evolution of a conserved active site domain. Nucleic Acids Res. 25, 3605–3614 (1997).
Goerke, C., Köller, J. & Wolz, C. Ciprofloxacin and trimethoprim cause phage induction and virulence modulation in Staphylococcus aureus . Antimicrob. Agents Chemother. 50, 171–177, doi: 10.1128/AAC.50.1.171-177.2006 (2006).
Fornelos, N., Browning, D. F. & Butala, M. The use and abuse of LexA by mobile genetic elements. Trends Microbiol. 24, 391–401, doi: 10.1016/j.tim.2016.02.009 (2016).
Ito, T. et al. Novel type V staphylococcal cassette chromosome mec driven by a novel cassette chromosome recombinase, ccrC . Antimicrob. Agents Chemother. 48, 2637–2651 (2004).
Sivaraman, K., Venkataraman, N., Tsai, J., Dewell, S. & Cole, A. M. Genome sequencing and analysis reveals possible determinants of Staphylococcus aureus nasal carriage. BMC Genomics 9, 433, doi: 10.1186/1471-2164-9-433 (2008).
Kreiswirth, B. N. et al. The toxic shock syndrome exotoxin structural gene is not detectably transmitted by a prophage. Nature 305, 709–712 (1983).
Zankari, E. et al. Identification of acquired antimicrobial resistance genes. J. Antimicrob. Chemother. 67, 2640–2644, doi: 10.1093/jac/dks261 (2012).
Hyatt, D. et al. Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics 11, 119 (2010).
Sigrist, C. J. et al. New and continuing developments at PROSITE. Nucleic Acids Res. 41, D344–347, doi: 10.1093/nar/gks1067 (2013).
Marchler-Bauer, A. et al. CDD: NCBI’s conserved domain database. Nucleic Acids Res. 43, D222–226, doi: 10.1093/nar/gku1221 (2015).
Schenk, S. & Laddaga, R. A. Improved method for electroporation of Staphylococcus aureus . FEMS Microbiol. Lett. 73, 133–138 (1992).
Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular cloning. A laboratory manual. 2nd ed, (Cold Spring Harbor Laboratory Press, 1989).
Vaudaux, P. et al. Intensive therapy with ceftobiprole medocaril of experimental foreign-body infection by methicillin-resistant Staphylococcus aureus . Antimicrob. Agents Chemother. 49, 3789–3793, doi: 10.1128/AAC.49.9.3789-3793.2005 (2005).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for bigger datasets. Mol. Biol. Evol. 33, 1870–1874, doi: 10.1093/molbev/msw054 (2016).
Sullivan, M. J., Petty, N. K. & Beatson, S. A. Easyfig: a genome comparison visualizer. Bioinformatics 27, 1009–1010 (2011).
Acknowledgements
We thank Basilea Pharmaceutica and AstraZeneca for kindly providing ceftabiprole and ceftaroline, respectively. We also thank the diagnostic unit ZOBA at our Institute for providing clinical M. caseolyticus strains. This study was supported by the research Grant 35–539 from the Institute of Veterinary Bacteriology, University of Bern, Bern, Switzerland.
Author information
Authors and Affiliations
Contributions
V.P. and S.SC. designed the study and prepared the manuscript. Genome assembly was done by V.P. and S.SC. annotated the mecD-containing elements and performed all the molecular and phenotypic characterizations. K.C. and S.SC. constructed mecD-deleted mutants.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Schwendener, S., Cotting, K. & Perreten, V. Novel methicillin resistance gene mecD in clinical Macrococcus caseolyticus strains from bovine and canine sources. Sci Rep 7, 43797 (2017). https://doi.org/10.1038/srep43797
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep43797
This article is cited by
-
Nasotracheal Microbiota of Nestlings of Parent White storks with Different Foraging Habits in Spain
EcoHealth (2023)
-
Rapid MRSA detection via tandem mass spectrometry of the intact 80 kDa PBP2a resistance protein
Scientific Reports (2021)
-
Multi-species host range of staphylococcal phages isolated from wastewater
Nature Communications (2021)
-
Technological Applications of Macrococcus caseolyticus and its Impact on Food Safety
Current Microbiology (2021)
-
Prevalence of Panton-Valentine leukocidin in methicillin-resistant Staphylococcus aureus clinical isolates at a university hospital in Northern Cyprus: a pilot study
BMC Research Notes (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.