Abstract
The linkage between N2O emissions and the abundance of nitrifier and denitrifier genes is unclear in the intensively managed calcareous fluvo-aquic soils of the North China Plain. We investigated the abundance of bacterial amoA for nitrification and narG, nirS, nirK, and nosZ for denitrification by in situ soil sampling to determine how the abundance of these genes changes instantly during N fertilization events and is related to high N2O emission peaks. We also investigated how long-term incorporated straw and/or manure affect(s) the abundance of these genes based on a seven-year field experiment. The overall results demonstrate that the long-term application of urea-based fertilizer and/or manure significantly enhanced the number of bacterial amoA gene copies leading to high N2O emission peaks after N fertilizer applications. These peaks contributed greatly to the annual N2O emissions in the crop rotation. A significant correlation between annual N2O emissions and narG, nirS, and nirK gene numbers indicates that the abundance of these genes is related to N2O emission under conditions for denitrification, thus partly contributing to the annual N2O emissions. These findings will help to draw up appropriate measures for mitigation of N2O emissions in this ‘hotspot’ region.
Similar content being viewed by others
Introduction
Nitrous oxide (N2O) is a powerful long-lived greenhouse gas and has a 300-times stronger warming effect than that of carbon dioxide in the troposphere on a 100-yr time horizon1. It also reacts with ozone in the stratosphere and became the dominant substance in ozone depletion in the 21st century2. Nitrous oxide is emitted from both natural and anthropogenic sources, and agricultural soils amended with chemical nitrogen (N) fertilizers and/or manure are mainly anthropogenic sources, which contribute up to 66% of the current global anthropogenic N2O emissions3 and approximately 75% of anthropogenic emission in China4. This makes agricultural soils an important target in the efforts to mitigate anthropogenic N2O emissions both regionally and globally5.
A better understanding of the processes, mechanisms and factors controlling N2O production and emission is a prerequisite for managing agricultural N2O emissions. Much progress has been made in the last five decades6. Although abiotic reactions are important N2O sources under certain circumstances7, biological processes play more important roles through at least four microbially mediated processes, i.e., nitrifier nitrification, nitrifier denitrification, denitrifier denitrification and coupled nitrification-denitrification8,9,10,11,12 which are enhanced by the application of N fertilizers and/or manure to most agricultural soils. The occurrence of each N2O production process and its contribution to the total N2O emissions depend on the prevailing soil conditions such as pH, temperature, moisture, oxygen, and microbial available C and N13,14. The above factors could be classified into three categories, i.e., edaphic conditions, climatic conditions and agricultural management practices15. Due to the temporal and spatial variation in these factors, specifically measuring N2O fluxes and studying the processes among typical soil-climatic regions are important for the mitigation of agricultural N2O emissions locally and can greatly contribute to the global total N2O budget3.
The North China Plain (NCP), an important agricultural region of China with an area of approximate 35 million hectares, is located in northeast China (32–41°N, 113–120°E) on the alluvial plain of the Yellow River and has a warm-temperate sub-humid climate with cold winters and hot summers16. The current agricultural practice is a very intensive double-cropping cereal system with irrigated winter wheat and rain-fed summer maize rotations characterized by the application of large amounts of fertilizer N and irrigation with large amounts of groundwater to obtain relatively high yields16. These practices lead to substantial total N2O emissions in this region which has become a ‘hotspot’ of national N2O emissions with global significance17. Our laboratory and field experiments on these intensively managed low-carbon calcareous soils over numerous years have found that high peaks of N2O emissions (mainly occurred within the first week after fertilization) were always induced by the application of large amounts of NH4+- or urea-based fertilizers to the soils, with no strong emissions during other periods; even the high nitrate concentration in the soils contribute only a small proportion of the annual N2O emissions18. Moreover, a total of 67–88% of the N2O is emitted during the summer maize season in the annual N2O emissions18,19,20,21,22,23,24,25,26,27. The N2O emission factors are generally lower than the IPCC (2006)28 default value (1%), with 0.10–0.59% on an annual basis, 0.08–0.21% in the winter wheat season, and 0.44–0.59% in the summer maize season16,18,20,22,26,27. We speculated that high ammonia oxidation activity linked to denitrification by heterotrophic denitrifiers or by nitrifiers are the major processes causing the observed instant high N2O peaks18,24,27,29. In the present study we quantify the relevant functional genes of the soil microbial community to further elucidate the biological mechanism of N2O emissions in this important region.
The community composition of both nitrifiers and denitrifiers is controlled by distal and proximal factors30. Distal factors are those factors that control the composition and diversity of nitrifying and denitrifying communities over the long term, and the proximal factors are those factors that affect instantaneous nitrification and denitrification rates. The ammonia oxidizing bacteria (AOB) community could be shaped by distal factors such as soil pH, ammonia and oxygen availability through which proximal factors such as application of NH4+- or urea-based fertilizer and/or manure and temporal soil temperature and moisture affect the nitrification rate31,32,33,34. Denitrifier community composition is structured over the long term by distal controls including the temperature and moisture conditions, substrate availability, competition and disturbances. The rate and kinetics of denitrification at any particular moment are controlled by proximal factors such as oxygen, carbon and nitrate availability35.
It is still not clear how the abundance of the bacterial amoA gene for nitrification and functional genes for denitrification respond instantly to N fertilization, how they relate to high N2O emission peaks, or how long-term incorporated straw and/or manure affect these linkages in the intensively managed low-carbon calcareous soils of the NCP. Our hypotheses are as follows. Firstly, under temporally high NH4+ concentrations the ammonia oxidizing bacteria will actively grow and accumulate high concentrations of NO2−, leading to anoxic conditions which, in turn, induce denitrification by heterotrophic denitrifiers, and the above chain processes cause the high N2O emission peaks after N fertilization. Secondly, long-term incorporated straw and/or manure will increase soil organic carbon (SOC) and total nitrogen (TN) and lead to increased abundance of denitrification functional genes as a distal control and the denitrifiers act under depleted oxygen conditions when rainfall or irrigation occur, thus partly contributing to annual N2O emissions.
We have therefore quantified the functional genes in soils that were sampled in situ three times and have performed an Illumina-based 16S rRNA gene sequencing analysis in the 2012–2013 winter wheat-summer maize rotation based on a long-term field experiment that began in 2006 at the Shangzhuang Research Station in suburban Beijing. N2O emissions and the concentrations of ammonium, nitrite and nitrate were determined at the same time. The field experiment had been running for seven years at the time of soil sampling. We were therefore able to effectively quantify the relatively long-term effects of different fertilization regimes including chemical fertilizer N combined with straw and/or manures on the total bacterial copy numbers (16S rDNA) and the populations of ammonia-oxidizing and heterotrophic denitrifying bacteria.
Results
Changes in the soil chemical and biological properties after 7 years
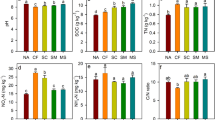
The SOC in Nopt, CNopt and CM increased by 15.8, 18.5, and 67.1%, and the TN correspondingly increased by 10.1, 15.2 and 52.5%, respectively, compared with the N0 treatment. This indicates that all N fertilization treatments tended to increase SOC and TN compared to the N0 control but the effect was only statistically significant for the manure treatment (Table 1). The soil C/N ratio and NH4+ concentration did not change significantly, and the ammonium concentration was very low, approximately 2 mg N kg−1, among the four treatments.
The nitrate concentration in the Nopt, CNopt and CM treatments was 38.5, 36.5 and 26.2 mg N kg−1, respectively, approximately 1.5 to 2.6 times higher than in the N0 control. The soil potential nitrification rate in Nopt, CNopt and CM was 3.39, 3.90 and 3.80 times significantly higher than in the N0 control, respectively. The soil potential denitrification rate in Nopt, CNopt and CM was 1.09, 1.80 and 3.12 times higher compared with the N0 control; CNopt and CM were significantly higher than in the Nopt and N0 treatments, respectively; and only the CM treatment was significantly higher than the other three treatments.
The soil pH measured on the sampling date of 16th April was approximately 7.57–7.75 and was not significantly different among the four treatments (Table S3). However, it was significantly lower in the three fertilization treatments compared to the N0 control when measured on the sampling dates of 9th August and 14th August, indicating that urea hydrolysis and nitrification would reduce the soil pH shortly in the calcareous soil studied36.
The above changes in the C and N status among treatments are likely the most important distal drivers of bacterial community composition, the abundance of nitrifier and denitrifier genes and related to instant generation of N2O by proximal drivers.
Nitrous oxide emission
N2O fluxes were very low on 16th April and there were no significant differences among the four treatments (Fig. 1a) during this zero-N fertilization period; there were even negative fluxes in the N0 and Nopt treatments which may be explained by the low soil temperature (9.6 °C) and moisture content (approximately 11%) in April (Table S3)20,23. The fluxes appeared to be higher on 9th August than on 16th April in all treatments but were not significantly different (although CM treatment was the highest) due to the rising soil temperature (24–26 °C) and moisture content (approximately 16%) in August, and the higher concentrations of nitrite and nitrate in the soil matrix (Table S3; Fig. 2a,b,c). Surprisingly, there was still 3.2–10.4 mg N kg−1 of nitrite in the fertilized soils on 9th August, which is unusual in the soil studied (further explanation in the Discussion section). Although the SOC and TN status changed among the different treatments after 7 years, the N2O fluxes did not respond significantly during the periods of zero fertilization in the studied soil.
N2O fluxes on the sampling dates in 2013 (a); N2O data in the studied year of the 2012–2013 winter wheat-summer maize rotation (b); and N2O emission factor (c). Different letters in (a) indicate significant differences (P < 0.05) among treatments on the same sampling date, and different letters in (b) and (c) indicate significant differences (P < 0.05) among treatments in annual base.
However, the N2O fluxes increased greatly within 7 days during the 10th leaf fertilization of summer maize on the third soil sampling date of 14th August (Fig. 1a). The fluxes in the N fertilization treatments (Nopt, CNopt, CM) were all significantly higher than in the N0 control, increasing to 7.0, 4.3 and 6.1-times, respectively, but were not statistically significant among the fertilization treatments, possibly due to the high spatial variation of measured N2O fluxes within the field plots. High concentrations of NH4+, NO2− and NO3− were detected in all of the fertilization treatments and were significantly higher in the CM treatment. The high NH4+ concentration in the CM treatment might result mainly from the high N mineralization due to the significantly higher SOC and TN contents in this treatment37,38 (Table 1). A significantly high NH4+ concentration in the CM treatment also induced high NO2− and NO3− concentrations in the soil matrix as a consequence of the typical nitrification chain.
The above results indicate that the high N2O emission was mainly derived from the processes of urea hydrolysis to NH4+ and then oxidation to NO2− and finally to NO3−. These processes were little affected by the distal drivers of the background C and N status but rather by N fertilization events. We will further explain this critical, instant and strong emission period of high N2O peaks by bacterial amoA abundance in the next section.
The annual N2O emissions were in the sequence CM > CNopt > Nopt > N0 and were significantly higher in the CNopt and CM treatments than in the Nopt and N0 treatments (Fig. 1b). These emissions were increasing to 2.6, 4.3 and 6.2 times in the Nopt, CNopt, and CM treatments compared to the N0 control. This increase corresponded well to the order of the total N applied rate (Table 2) and followed the order of the SOC and TN status among treatments (Table 1). We further calculated the emission factor to investigate the N2O emissions per unit of N input in order to determine whether the manure and/or straw N interacted with chemical N to stimulate N2O emissions per unit of N input39.
The emission factors also increased in the sequence CM > CNopt > Nopt, but there were no significant differences among these treatments, possibly also due to the high spatial variation of measured N2O fluxes in the field plots (Fig. 1c). In our study the annual N2O emission factor was 0.20–0.40%, much lower than the IPCC default value of 1%28 but was in line with our previous studies18,20,23,24,25.
Abundance of the 16S rRNA gene and nitrification and denitrification genes
The order of the 16S rRNA gene copy number was CM > CNopt > Nopt > N0 on all three sampling dates (Fig. 3), a similar order to SOC, but only significantly higher in the CM treatment compared with the other three treatments. The 16S rRNA gene copy numbers in the CM treatment were 1.6–1.9-, 1.7–2.1- and 1.3–2.2-times larger than in the CNopt, Nopt, and N0 treatments on the three sampling dates, indicating that soil with long-term incorporated manure harbored the largest 16S rRNA gene number. Surprisingly, the 16S rRNA gene copies in all four treatments were higher on the 16th April soil sampling date than on 9th August or 14th August, possibly due to the different crops because the first sampling date was winter wheat and the last two sampling dates were summer maize (further explanation in the Discussion section). The soil 16S rRNA gene copy numbers in the CNopt treatment appeared to be 27.2, 16.7 and 27.7% higher than in the Nopt treatment on the three sampling dates, indicating that straw return increased the soil 16S rRNA gene copy number but this was not statistically significant.
The gene copy numbers of bacterial amoA in all three fertilization treatments were significantly higher than in the N0 control but were not significantly different among fertilization treatments on all three sampling dates (Fig. 4), reflecting the legacy of historical long-term application of urea-based fertilizer and/or manure as distal drivers to regulate nitrification. These numbers were 1.4e + 7 to 1.9e + 7 and 1.4e + 7 to 1.6e + 7 on 16th April and 9th August, respectively. On the third day after the 10th leaf fertilization, the amoA gene number of Nopt, CNopt and CM increased 14.7, 124.5 and 107.6% compared to that before fertilization (on 9th August), respectively, indicating that the 10th leaf fertilization of maize enhanced the amoA gene copy number as proximal factor and led to production of high N2O emission peaks in this short period (Fig. 1a). This result further confirms our previous conclusion that ammonia oxidation is an engine to generate nitrous oxide in this intensively managed calcareous Fluvo-aquic soil24.
The abundance of the denitrification genes encoding nitrate reductase (narG), nitrite reductases (nirS and nirK) and N2O reductase (nosZ) followed a similar sequence, namely CM > CNopt > Nopt > N0, on 16th April and 9th August, but in most cases these genes were significantly higher only in the CM treatment compared with the other three treatments (Fig. 5a,b,c,d), which seems to follow the trend of SOC and TN. The third sampling date on 14th August, after fertilization, likely disturbed this trend, and none of the genes were significantly different among fertilization treatments. These results indicate that the abundance of denitrification genes was regulated by both distal drivers, such as SOC and TN, and proximal drivers, such as N fertilization. The gene copy numbers of narG, nirS and nirK in different treatments on 16th April and 9th August corresponded well with the daily N2O emissions during these two zero fertilization periods and with the annual N2O emissions (Fig. 1a,b). The gene copy numbers of narG, nirS and nirK in different treatments on 14th August seem also to partly correspond with high N2O emission after fertilization. Although the nosZ gene number was significantly higher in the CM treatment in most cases, the N2O emission factor in this treatment was still higher than that of the other treatments, likely due to the effect of the nosZ gene being partly offset by increased effects of narG, nirS and nirK genes.
Sequencing analysis
The PCoA score plots based on the Bray-Curtis distance reveal that fertilization with straw and/or manure changed the structure of the soil microbiota (Fig. 6a) and there were statistically significant differences between the N0 control and the fertilization treatments (Fig. 6b), i.e., Nopt, CNopt and CM were separate from N0. Among the fertilization treatments, Nopt was separated from the CNopt treatment in the PCoA score plots, but this separation was not statistically significant; CM was significantly separated from the Nopt and CNopt treatments (Fig. 6a,b). The bacterial community on 14th August after fertilization was also separated from those on the first two sampling dates. These results further indicate that the soil microbial community was influenced by both distal drivers such as changes in the SOC and TN, and proximal drivers, such as N fertilization application.
(a) Principal coordinate analysis (PCoA) score plots based on the Bray-Curtis distance. (b) Clustering of soil microbiota based on Mahalanobis distances calculated with a multivariate analysis of variance (MANOVA). Each point represents the mean principal coordinate (PC) score of three replicate soil samples from one treatment at one time point. ***P < 0.001, **P < 0.01.
Correlations between N2O emission and some soil chemical and biological parameters
The annual N2O emissions were significantly correlated with SOC and TN, potential denitrification rate (PDNR), amoA gene number, denitrification functional gene numbers (narG, nirS, nirK, nosZ) and 16S rRNA gene number by Spearman’s rank correlation analysis (Table 3) but were not significantly correlated with soil potential nitrification rate (PNR), likely due to all of the fertilization treatments having a high soil PNR (Table 1). Thus, soil PNR is not a limiting factor for controlling N2O production and emission. All of the other soil properties and abundance of functional genes were significantly correlated with each other except for amoA with SOC and TN and PNR with TN, which corresponds well with the above results showing that the amoA gene number did not respond to distal control (SOC and TN) but responded strongly to instant fertilization as a proximal control (Fig. 4).
Discussion
We investigated the bacterial amoA for nitrification in situ during both the zero-fertilization period and a fertilization event using a long-term field experiment with different fertilization regimes in an intensively managed low-carbon calcareous soil on the NCP. These results show that the long-term application of urea-based fertilizer and/or manure induced and enhanced bacterial amoA gene copies, which was related to a strong nitrification process, and consumed O2 in the soil matrix then triggered denitrification as shown by our previous study24. The N2O emission induced by ammonia oxidation is also likely dependent on the heterotrophic respiration rate because incorporation of straw and/or manure aggravated the emissions23. This suggests that the ammonia oxidizing bacteria will actively grow under temporally high NH4+ concentrations, leading to microoxic or anoxic conditions, which in turn induce denitrification by heterotrophic denitrifiers or by nitrifiers, and the above chain processes lead to the high N2O emission peaks after N fertilization. The triggering of denitrification was further demonstrated by enhanced nirS and nirK genes in the current study. Previous studies in the same cropping rotation system showed that these peaks occurring instantly after N fertilization contributed up to 30–70% of the annual N2O emissions and were the key periods for controlling N2O emissions on the NCP18,20,22,25,27,40. This process may be slowed down using nitrification inhibitors and reducing field N2O emissions by up to 77% using DMPP18 and 55% using the liquid nitrification inhibitor Piadin22. These emissions may be reduced by as much as 80–99% using DMPP or DCD in laboratory experiments24,29 in calcareous fluvo-aquic soils from the NCP.
Our results provide molecular microbial evidence to illustrate that ammonia oxidation is an engine as the start for generating N2O in the study soil, which is why nitrification inhibitors are good for reducing N2O emissions18,22,24,29 in this soil. The results of the bacterial amoA gene agree with other related studies under similar soil and climatic conditions33,37,41,42.
It has been reported that AOAs are ubiquitous in soils but do not respond to NH4+ oxidization or N2O production in intensively managed agricultural soils32,43. Currently, the soils where AOAs have a significant impact on NH4+ oxidization are mainly acidic44. One study conducted in our study region also revealed that Nitrosospira-like AOBs were dominant over AOAs in oxidizing NH4+42. Therefore, the contribution of AOAs to N2O production is small in our calcareous soil and this is why we did not quantify the amoA gene of archaea in our study.
Few studies have investigated the abundance of denitrification genes and their linkage with N2O production in our study soil, especially considering the distal and proximal control. We quantified the critical functional genes narG, nirS, nirK, and nosZ for denitrification and associated their abundances with annual N2O emissions. The significant correlations between annual N2O emissions and narG, nirS, and nirK gene numbers show that these genes abundances were related to production of N2O under some favorable conditions for denitrification; for example, rainfall or irrigation induce a low oxygen concentration in soil microsites18,19,20,21,22,23,24,25,26,27, which may be further supported by the significant correlation between annual N2O emissions and SOC and between narG, nirS, and nirK gene abundances and SOC. These findings further explain our previous results showing that the small pulses occurred when rainfall or irrigation events occurred in the same cropping system, especially during the warm and moist summer18,19,20,21,22,23,24,25. Therefore, the increased abundance of denitrification genes by long-term incorporated manure can produce N2O in our study soil and also partly contribute to the annual N2O emissions, which were enhanced by increased SOC and TN. A similar study was conducted using soils sampled in Broadbalk Wheat and the “Broadbalk Wilderness” long-term experiments45 but using the incubation technique labeled with KNO3 solution to investigate the influence of different long-term fertilization and cultivation treatments on denitrifier communities and to produce N2O. The results show that bacteria containing nirK were most likely responsible for the increased denitrification potential associated with high SOC and TN, which generally agrees with our findings.
Why was there nitrite accumulation on 9th August (Fig. 2b) when the soil ammonium concentration was quite low (Fig. 2a)? We suspected that it might be due to the reduction of nitrate in more anaerobic microsites together with high levels of dissoluble organic carbon induced by fast root metabolism during the period of strong summer maize growth at higher soil temperatures (approximately 24 °C) and water content (approximately 16%)46,47. Thus, this nitrite accumulation could contribute to the annual N2O emissions. This hypothesis is supported by a recent study that demonstrated the significant contribution of nitrite to N2O emissions in maize-cropped soil29,48.
The explanation for the lower 16S rRNA gene copy number on the two sampling dates of maize (9th and 14th August) than on the first sampling date of wheat (16th April) (Fig. 3) was likely due to competition for resources between crop roots and soil microbes49. Summer maize roots might exhaust their resources during fast growth and suppress the growth of microbes, which may result in a lower 16S rRNA gene copy number in the summer maize season. Few studies have reported this phenomenon and it merits further study.
Our study highlights the linkage of instant high N2O emission peaks with the abundance of the bacterial amoA gene for nitrification; annual N2O emissions and a small N2O pulse after rainfall or irrigation with the abundance of denitrification genes, providing insight into the mechanism of N2O production and the factors controlled by distal and proximal drivers in this intensively managed calcareous fluvo-aquic soil (Fig. 7). It is critical to suppress the growth of bacteria containing the amoA gene instantly after urea-based fertilization to mitigate N2O emission in these strong nitrification soils. We also need to be concerned about the enhanced abundances of denitrification functional genes under favorable conditions.
Methods
Site description and soil sampling
The soils on the NCP are derived from alluvial loess transported by the Yellow River and its tributaries and most of the soils are calcareous Fluvisols or Cambisols (FAO Soil Classification System) with a silt texture and relatively uniform profile characterized by high mineralization, high nitrification and low denitrification rates17,38. The calcareous soils have a pH of 7.5–8.5 and an organic matter content of approximately 10–15 g kg−1. Annual cumulative mean temperature for days with temperatures above 10 °C is 4000–5000 °C and the annual precipitation is 500–700 mm with 60–70% of the rainfall occurring during the summer (July–September)23. The long-term field experiment began in October 2006 and is located at Shangzhuang Research Station (39°48′ N, 116°28′ E) of China Agricultural University in suburban Beijing. The expression “long-term” in this paper refers to fertilization treatments compared to the no fertilizer control after a period of seven years.
The cropping system was a winter wheat-summer maize rotation. The top 20 cm of the soil profile, sampled at the beginning of the field experiment, had 28% clay, 32% silt, and 40% sand contents and a pH of 8.1 (soil: water ratio 1:2.5). The bulk density was 1.31 g cm−3 and the nutrient contents were as follows: SOC 7.1 g kg−1, TN 0.8 g kg−1, NO3-N 24.5 mg kg−1, NH4-N 1.20 mg kg−1, Olsen P 7.8 mg kg−1 and available K 76.2 mg kg−1. The soil is a typical calcareous fluvo-aquic soil widely distributed across the NCP.
Eight treatments were set up as described in a previous paper50. Our current study sampled from four treatments, i.e., N0, Nopt, CNopt and CM, in the 2012–2013 winter wheat-summer maize rotation based on the significant difference of some soil chemical and biological properties at 0–20 cm soil depth sampled before the sowing of winter wheat in 2012 (Table 1). N0 is no N application, wheat and maize straw removed; Nopt and CNopt are chemical fertilizer N application according to the improved Nmin (NO3−-N + NH4+-N) test, wheat and maize straw removed or returned, respectively; CM is cattle manure supplementary applied N based on the N balance calculation, wheat and maize straw returned. The design is a completely randomized block with three replicates and each plot area is 64 m2 (8 × 8 m). Winter wheat was sown at the beginning of October and harvested in the middle of the following June and summer maize was subsequently sown and harvested at the end of September. The chemical N fertilizer used was urea. Straw of both maize and wheat was chopped mechanically into 5–8 cm lengths. Chemical fertilizers and chopped maize straw were incorporated into the soil with tillage at the beginning of October before wheat was sown, and the wheat straw was mulched on the soil surface after the wheat harvest.
In the 2012–2013 winter wheat-summer maize rotation basal fertilizer was applied and winter wheat was sown on 2 October 2012 (Table 2), and winter irrigation (60 mm) was carried out on 17 November 2012. We collected the first soil samples (0–20 cm) at the jointing stage of winter wheat on 16 April 2013 to investigate the effects of long-term treatments on the soil bacterial community composition and the abundance of functional genes. After this sampling, jointing fertilizer was applied on 21 April 2013, followed by 60 mm of irrigation; irrigation (60 mm) was carried out on 14th May 2013 at the heading stage of winter wheat. Winter wheat was harvested on 21st June 2013, summer maize was sown immediately, and the 4th leaf fertilizer of maize was applied on 20th July 2013. The second and third soil samples (0–20 cm) were collected before one day (9th August) and after three days (14th August), respectively, when the 10th leaf fertilization of maize was performed on 11th August 2013 to investigate the effects of N fertilization on N2O emissions, the corresponding bacterial community composition and the abundance of functional genes.
Five soil cores were taken from each plot and were mixed to form one composite sample; the samples were stored in an ice box before returning to the laboratory within half a day. The samples were divided into two parts. One part of the fresh soil was used to measure the chemical properties, including ammonium, nitrite, nitrate, water contents and pH, immediately after arriving at the laboratory. The remainder of each sample was stored at −80 °C for subsequent DNA extraction and downstream analysis, including the quantitative real-time PCR (Q-PCR) of the 16 S rRNA gene, functional genes of amoA gene of bacterial, nitrate reductase gene (narG), nitrite reductase genes (nirS and nirK), and N2O reductase gene (nosZ), by high-throughput sequencing of 16S rRNA based on an Illumina platform analysis (Illumina Inc., San Diego, CA).
N2O emission measurements
N2O emissions were measured using the closed static chamber method as detailed in Huang et al.23. They were measured on days of first two soil sampling dates, i.e., on 16th April and 9th August. For third soil sampling day on 14th August, daily measurements were carried out for 7 days after the 10th leaf fertilization on 11th August in order to cover the entire N2O peaking period during this N fertilization event. During the whole crop rotation, daily measurements were also carried out for 7 days after each fertilization event and 5 days after each rainfall or irrigation event; for the remaining periods the emissions were measured twice per week and once per week when the soil was frozen23.
In the current study N2O emissions were calculated daily as μg N2O-N m−2 d−1 on 16th April and 9th August (Fig. 1a), but the N2O emissions on 14th August were calculated as the average emissions over 7 days (peak lasting period) after N fertilization in order to reflect the total N2O emissions induced by N fertilization. The annual N2O emissions (Fig. 1b) were calculated from the sum of measurement days and zero-measurement days (estimated by linear interpolation)23. The annual N2O emission factor was calculated by subtracting the annual N2O emissions of the control treatment from those of the fertilization treatment and then divided by the total N application rate, including straw N and manure N (Fig. 1c), according to IPCC methodology.
Determination of soil properties
Mineral N was extracted using 1 M KCl solution at a soil:water ratio of 1:5 (w/v) and determined using a continuous flow analyzer (AA3, Seal Analytical, Norderstedt, Germany). Soil nitrite was measured as described by Stevens and Laughlin51, the soil nitrification and denitrification potential were determined according to Hart et al.52 and Tiedje et al.53 respectively. Details can be found in the Supplementary Information (SI).
Soil molecular analysis
DNA extraction
DNA was extracted from frozen soil using a method based on the CTAB (hexadecyl trimethyl ammonium bromide) method54 with some modification, details are described in the SI.
Real-time PCR
Quantification of 16 S rRNA genes, and the functional genes amoA of ammonia oxidizing bacteria (AOB), nirS, nirK, narG and nosZ was performed on a Lightcycler 96 system (Roche, Basel, Switzerland) using triplicate samples of diluted DNA (10 ng μl−1). Primer pairs of Uni331F/Uni79755, amoA-1F/2R56, narG-f/r57, nirS cd3A/R3cd58, nirK1040/FlaCu59,60, and nosZ-2f/2r61 were used for their quantification (Table S1). Each plate included purified plasmid standards and negative controls in triplicate. The amplification efficiency of 16S rRNA, amoA, narG, nirS, nirK, and nosZ was 87, 85, 86, 78, 85 and 93%, respectively. Much more details can be found in the SI.
Illumina-based 16S rRNA gene sequencing
The gene-specific sequences that were used targeted the 16S rRNA gene V3 and V4 regions. The primers that were used for the Amplicon PCR are listed in Table S2. The target fragment was approximately 550 bp. More details can be found in the SI. The 16S rRNA gene sequence information in this study has been submitted to the NCBI Sequence Read Archive (SRA) database under accession number SRP083579.
Statistical analysis
The data were computed using Microsoft 2010, and the results are reported as the means (±standard error), and the figures were created using SigmaPlot v. 12.5 and Visio 2013. Differences in the soil properties and functional genes were analyzed by one-way analysis of variance and compared by Duncan’s multiple range test at the 5% level using IBM SPSS Statistics 20. We calculated the functional gene numbers considering the effects of treatment and neglected sampling time to assess the significance of correlations between N2O emissions and related soil parameters. A non-parametric analysis was used to generate a Spearman’s rank correlation matrix using IBM SPSS Statistics 20. The method of dealing with sequencing data is described in the SI.
Additional Information
How to cite this article: Yang, L. et al. Linkage between N2O emissions and functional gene abundance in an intensively managed calcareous fluvo-aquic soil. Sci. Rep. 7, 43283; doi: 10.1038/srep43283 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Solomon, S. et al. In Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (eds Solomon, S. et al.) 33–34 (Cambridge University Press, Cambridge, 2007).
Ravishankara, A. R., Daniel, J. S. & Portmann, R. W. Nitrous Oxide (N2O): The dominant ozone-depleting substance emitted in the 21st century. Science. 326, 123–125 (2009).
Drawing Down N2O to Protect Climate and the Ozone Layer. A UNEP (United Nations Environment Programme) Synthesis Report. Nairobi, Kenya (2013).
NDRC. The People’s Republic of China National Greenhouse Gas Inventory. China Environmental Science Press, Beijing (2014).
Tian, H. et al. The terrestrial biosphere as a net source of greenhouse gases to the atmosphere. Nature 531, 225–228 (2016).
Butterbach-Bahl, K., Baggs, E. M., Dannenmann, M., Kiese, R. & Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos. Trans. R. Soc. London, Ser. B 368, 91–97 (2013).
Bremner, Sources of nitrous oxide in soils. Nutr. Cycl. Agroecosys. 49, 7–16 (1997).
Wrage, N., Velthof, G. L., van Beusichem, M. L. & Oenema, O. Role of nitrifier denitrification in the production of nitrous oxide. Soil Biol. Biochem. 33, 1723–1732 (2001).
Wrage, N., van Groenigen, J. W., Oenema, O. & Baggs, E. M. A novel dual-isotope labelling method for distinguishing between soil sources of N2O. Rapid Commun. Mass. Spectrom. 19, 3298–3306 (2005).
Kool, D. M. et al. Nitrifier denitrification can be a source of N2O from soil: a revised approach to the dual-isotope labelling method. Eur. J. Soil Sci. 61, 759–772 (2010).
Baggs, E. M. Soil microbial sources of nitrous oxide: recent advances in knowledge, emerging challenges and future direction. Curr. Opin. Environ. Sustain. 3, 321–327 (2011).
Zhu, X., Burger, M., Doane, T. A. & Horwath, W. R. Ammonia oxidation pathways and nitrifier denitrification are significant sources of N2O and NO under low oxygen availability. Pro. Natl. Acad. of Sci. USA 110, 6328–6333 (2013).
Butterbach-Bahl, K. & Dannenmann, M. Denitrification and associated soil N2O emissions due to agricultural activities in a changing climate. Curr. Opin. Environ. Sustain. 3, 389–395 (2011).
Venterea, R. T. et al. Challenges and opportunities for mitigating nitrous oxide emissions from fertilized cropping systems. Front. Ecol. Environ. 10, 562–570 (2012).
Snyder, C. S., Bruulsema, T. W., Jensen, T. L. & Fixen, P. E. Review of greenhouse gas emissions from crop production systems and fertilizer management effects. Agric. Ecosyst. Environ. 133, 247–266 (2009).
Ju, X. T. et al. Reducing environmental risk by improving N management in intensive Chinese agricultural systems. Pro. Natl. Acad. of Sci. USA 106, 3041–3046 (2009).
Zhou, F. et al. A new high-resolution N2O emission inventory for China in 2008. Environ. Sci. Technol. 48, 8538–8547 (2014).
Ju, X. & Christie, P. Calculation of theoretical nitrogen rate for simple nitrogen recommendations in intensive cropping systems: A case study on the North China Plain. Field Crops Res. 124, 450–458 (2011).
Gao, B., Ju, X. T., Zhang, Q., Christie, P. & Zhang, F. S. New estimates of direct N2O emissions from Chinese croplands from 1980 to 2007 using localized emission factors. Biogeosciences 8, 3011–3024 (2011).
Gao, B. et al. Nitrous oxide and methane emissions from optimized and alternative cereal cropping systems on the North China Plain: a two-year field study. Sci. total Environ. 472, 112–124 (2014).
Gao, B. et al. The impact of alternative cropping systems on global warming potential, grain yield and groundwater use. Agric. Ecosyst. Environ. 203, 46–54 (2015).
Hu, X. K. et al. Greenhouse gas emissions from a wheat-maize double cropping system with different nitrogen fertilization regimes. Environ. Pollut. 176, 198–207 (2013).
Huang, T., Gao, B., Christie, P. & Ju, X. Net global warming potential and greenhouse gas intensity in a double-cropping cereal rotation as affected by nitrogen and straw management. Biogeosciences 10, 7897–7911 (2013).
Huang, T. et al. Ammonia-oxidation as an engine to generate nitrous oxide in an intensively managed calcareous fluvo-aquic soil. Sci. Rep. 4, 3950 (2014).
Bao, Q. L., Ju, X. T., Gao, B., Qu, Z., Christie, P. & Lu, Y. H. Response of nitrous oxide and corresponding bacteria to managements in an agricultural soil. Soil Sci. Soc. Am. J. 76, 130–141 (2012).
Yan, G. X. et al. Two-year simultaneous records of N2O and NO fluxes from a farmed cropland in the northern China plain with a reduced nitrogen addition rate by one-third. Agric. Ecosyst. Environ. 178, 39–50 (2013).
Cui, F., Yan, G., Zhou, Z., Zheng, X. & Deng, J. Annual emissions of nitrous oxide and nitric oxide from a wheat–maize cropping system on a silt loam calcareous soil in the North China Plain. Soil Biol. Biochem. 48, 10–19 (2012).
IPCC. N2O emissions from managed soils and CO2 emissions from lime and urea application. In IPCC guidelines for national greenhouse gas invertories 4 (eds Eggleston, H. S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K. ) 11.1–11.54. Hayama, Japan: IGES (2006).
Ma, L., Shan, J. & Yan, X. Y. Nitrite behavior accounts for the nitrous oxide peaks following fertilization in a fluvo-aquic soil. Biol. Fertil. Soils 51, 563–572 (2015).
Braker, G. & Conrad, R. Diversity, structure, and size of N2O-producing microbial communities in soils--what matters for their functioning? Adv. Appl. Microbiol. 75, 33–70 (2011).
Chu, H. et al. Community structure of ammonia-oxidizing bacteria under long-term application of mineral fertilizer and organic manure in a sandy loam soil. Appl. Environ. Microbiol. 73, 485–491 (2007).
Di, H. J. et al. Nitrification driven by bacteria and not archaea in nitrogen-rich grassland soils. Nat. Geosci. 2, 621–624 (2009).
Shen, J. P., Zhang, L. M., Zhu, Y. G., Zhang, J. B. & He, J. Z. Abundance and composition of ammonia-oxidizing bacteria and ammonia-oxidizing archaea communities of an alkaline sandy loam. Environ. Microbiol 10, 1601–1611 (2008).
Ai, C. et al. Different roles of rhizosphere effect and long-term fertilization in the activity and community structure of ammonia oxidizers in a calcareous fluvo-aquic soil. Soil Biol. Biochem. 57, 30–42 (2013).
Wallenstein, M. D., Myrold, D. D., Firestone, M. & Voytek, M. Environmental controls on denitrifying communities and denitrification rates: Insights from molecular methods. Ecol. Appl. 16, 2143–2152 (2006).
Venterea, R. T. & Rolston, D. E. Mechanistic modeling of nitrite accumulation and nitrogen oxide gas emissions during nitrification. J. Environ. Qual. 29, 1741–1751 (2000).
Tejada, M., Hernandez, M. T. & Garcia, C. Soil restoration using composted plant residues: Effects on soil properties. Soil Till. Res. 102, 109–117 (2009).
Hartmann, T. E., Yue, S. C., Schulz, R., Chen, X. P., Zhang, F. S. & Müller, T. Nitrogen dynamics, apparent mineralization and balance calculations in a maize-wheat double cropping system of the North China Plain. Field Crop Res. 160, 22–30 (2014).
Jäger, N., Stange, C. F., Ludwig, B. & Flessa, H. Emission rates of N2O and CO2 from soils with different organic matter content from three long-term fertilization experiments - a laboratory study. Biol. Fertil. Soils 47, 483–494 (2011).
Ding, W. X., Cai, Y., Cai, Z. C., Yagi, K. & Zheng, X. H. Nitrous oxide emissions from an intensively cultivated maize-wheat rotation soil in the North China Plain. Sci. Total Environ. 373, 501–511 (2007).
Ge, Y., Zhang, J. B., Zhang, L. M., Yang, M. & He, J. Z. Long-term fertilization regimes affect bacterial community structure and diversity of an agricultural soil in Northern China. J. Soils Sediments 8, 43–50 (2008).
Xia, W. et al. Autotrophic growth of nitrifying community in an agricultural soil. ISME J. 5, 1226–1236 (2011).
Jia, Z. & Conrad, R. Bacteria rather than Archaea dominate microbial ammonia oxidation in an agricultural soil. Environ. Microbiol. 11, 1658–1671 (2009).
Prosser, J. I. & Nicol, G. W. Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends Microbiol. 20, 523–531 (2012).
Clark, I. M., Buchkina, N., Jhurreea, D., Goulding, K. W. & Hirsch, P. R. Impacts of nitrogen application rates on the activity and diversity of denitrifying bacteria in the Broadbalk Wheat Experiment. Philos. Trans. R. Soc. London, Ser. B. 367, 1235–1244 (2012).
Baggs, E. M., Richter, M., Hartwig, U. A. & Cadish, G. Nitrous oxide emissions from grass swards during the eighth year of elevated atmospheric pCO2 (Swiss FACE). Global Change Bilo. 9, 1214–1222 (2003).
Højberg, O., Binneruo, S. J. & Sørensen, J. Potential rates of ammonium oxidation, nitrite oxidation, nitrate reduction and denitrification in the young barley rhizophere. Soil Biol. Biochem. 28, 47–54 (1996).
Maharjan, B. & Venterea, R. T. Nitrite intensity explains N management effects on N2O emissions in maize. Soil Biol. Biochem. 66, 229–238 (2013).
Inselsbacher, E. et al. Short-term competition between crop plants and soil microbes for inorganic N fertilizer. Soil Biol. Biochem. 42, 360–372 (2010).
Qiu, S. et al. Improved Nitrogen Management for an Intensive Winter Wheat/Summer Maize Double-cropping System. Soil Sci. Soc. Am. J. 76, 286–297 (2012).
Stevens, R. J. & Laughlin, R. J. Nitrite transformation during soil extraction with potassium chloride. Soil Sci. Soc. Am. J. 59, 933–938 (1995).
Hart, S. C., Stark, J. M., Davidson, E. A. & Firestone, M. K.. In Microbiological and Biochemical properties: Nitrogen mineralization, immobilization, and nitrification. Methods of Soil Analysis: Part 2 (eds Bigham, J. M. ) 985–1018 (Madison, 1994).
Tiedje, J. M., Sexstone, A. J., Parkin, T. B., Revsbech, N. P. & Shelton, D. R. Anaerobic processes in soil. Plant Soil 76, 197–212 (1984).
Griffiths, R. I., Whiteley, A. S., O’Donnell, A. G. & Bailey, M. J. Rapid Method for Coextraction of DNA and RNA from Natural Environments for Analysis of Ribosomal DNA- and rRNA-Based Microbial Community Composition. Appl. Environ. Microbiol. 66, 5488–5491 (2000).
Nadkarni, M. A., Martin, F. E., Jacques, N. A. & Hunter, N. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiol. Soc. 148, 257–266 (2002).
Rotthauwe, J. H., Witzel, K. P. & Liesack, W. The ammonia monooxygenase structural gene amoA as a functional marker: Molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl. Environ. Microbiol. 63, 4704–4712 (1997).
Bru, D., Sarr, A. & Philippot, L. Relative abundances of proteobacterial membrane-bound and periplasmic nitrate reductases in selected environments. Appl. Environ. Microbiol. 73, 5971–5974 (2007).
Kandeler, E., Deiglmayr, K., Tscherko, D., Bru, D. & Philippot, L. Abundance of narG, nirS, nirK, and nosZ genes of denitrifying bacteria during primary successions of a glacier foreland. Appl. Environ. Microbiol. 72, 5957–5962 (2006).
Henry, S. et al. Quantification of denitrifying bacteria in soils by nirK gene targeted real-time PCR. J. Microbiol. Methods 59, 327–335 (2004).
Hallin, S. & Lindgren, P. E. PCR detection of genes encoding nitrite reductase in denitrifying bacteria. Appl. Environ. Microbiol. 65, 1652–1657 (1999).
Henry, S., Bru, D., Stres, B., Hallet, S. & Philippot, L. Quantitative Detection of the nosZ gene, encoding nitrous oxide reductase, and comparison of the abundances of 16S rRNA, narG, nirK, and nosZ genes in soils. Appl. Environ. Microbiol. 72, 5181–5189 (2006).
Acknowledgements
We thank Professors Lars R Bakken and Åsa Frostegård for their constructive discussions during the writing, and valuable comments and suggestions on the draft manuscript. We thank Dr Peter Christie for revising the formatting and English. This work was funded by the National Natural Science Foundation of China (41230856, 41471190, 41301237) and the Newton Fund (Grant Ref. BB/N013484/1).
Author information
Authors and Affiliations
Contributions
X.T.J. and X.J.Z. designed the experiment. L.Q.Y. carried out the experiments and performed the data analyses. X.T.J., L.Q.Y. and X.J.Z. substantially contributed to interpreting the results and writing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yang, L., Zhang, X. & Ju, X. Linkage between N2O emission and functional gene abundance in an intensively managed calcareous fluvo-aquic soil. Sci Rep 7, 43283 (2017). https://doi.org/10.1038/srep43283
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep43283
This article is cited by
-
Distinct Denitrifying Phenotypes of Predominant Bacteria Modulate Nitrous Oxide Metabolism in Two Typical Cropland Soils
Microbial Ecology (2023)
-
Response of N2O emission and denitrification genes to different inorganic and organic amendments
Scientific Reports (2022)
-
Effect of Fertilization Regimes and Seasonal Change on nosZ-Denitrifying Bacterial Community in a Double-Rice Paddy Field
Journal of Soil Science and Plant Nutrition (2022)
-
Oxygen concentrations regulate NO, N2O, and N2 kinetics and nitrogen transformation in a fluvo-aquic soil using a robotized incubation system
Journal of Soils and Sediments (2021)
-
Fate of 15N-labelled urea when applied to long-term fertilized soils of varying fertility
Nutrient Cycling in Agroecosystems (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.