Abstract
Restoration is increasingly considered an essential tool to halt and reverse the rapid decline of vital coastal ecosystems dominated by habitat-forming foundation species such as seagrasses. However, two recently discovered pathogens of marine plants, Phytophthora gemini and Halophytophthora sp. Zostera, can seriously hamper restoration efforts by dramatically reducing seed germination. Here, we report on a novel method that strongly reduces Phytophthora and Halophytophthora infection of eelgrass (Zostera marina) seeds. Seeds were stored in seawater with three different copper sulphate concentrations (0.0, 0.2, 2.0 ppm) crossed with three salinities (0.5, 10.0, 25.0 ppt). Next to reducing seed germination, infection significantly affected cotyledon colour: 90% of the germinated infected seeds displayed a brown cotyledon upon germination that did not continue development into the seedling stage, in contrast to only 13% of the germinated non-infected seeds. Copper successfully reduced infection up to 86% and the 0.2 ppm copper sulphate treatment was just as successful as the 2.0 ppm treatment. Infection was completely eliminated at low salinities, but green seed germination was also dramatically lowered by 10 times. We conclude that copper sulphate treatment is a suitable treatment for disinfecting Phytophthora or Halophytophthora infected eelgrass seeds, thereby potentially enhancing seed-based restoration success.
Similar content being viewed by others
Introduction
Seagrasses form vital habitats in coastal zones across the world and provide important ecosystem services such as coastal protection, carbon sequestration and biodiversity enhancement1,2,3. However, seagrass beds are currently experiencing rapid losses worldwide as a result of human-induced eutrophication, coastal pollution and habitat destruction4,5. To halt and reverse these losses, many restoration efforts are being undertaken6,7, with fifty percent of these restoration attempts concerning Zostera marina (eelgrass), the most widespread seagrass species in the temperate northern hemisphere6. Restoration projects involving eelgrass beds use a variety of techniques including transplantation of sods, rhizome fragments, seeds, seed-bearing shoots, or seedlings. Methods vary among sites, but there has been a general increase of projects using seeds for restoration8,9,10,11,12,13,14,15. Compared to more traditional approaches, using seeds is more cost-effective16, allows for maintaining high genetic diversity of the restored population17,18, and storage of seeds outside the growing season allows for reduction of seed loss compared to natural conditions19.
Although application of seeds in restoration is promising, Govers, et al.20 recently discovered that two oomycete pathogens, Phytophthora sp. and Halophytophthora sp. may pose a threat to seed-based restoration as they (1) reduce seed germination by six times, and (2) occur widespread across the Atlantic with very high infection rates (up to 99%). Moreover, the threat for restoration was further highlighted by the finding of Phytophthora infection in Z. marina seeds of currently ongoing restoration projects in the Netherlands, Denmark, Sweden and the USA20.
Phytophthoras are well known from terrestrial systems. For instance, the infamous Phytophthora infestans causes billions of euros of yearly damage to potato crops in the European union21, and related species, Phytophthora ramorum and Phytophthora cinnamomi, are threatening fragile ecosystems, causing massive die-offs of forests in California and Australia22,23. Since we now know that Phytophthora species can also infect and affect marine plants20,24, with infection rates as high as 99% in natural populations and a widespread distribution20, it is very likely that these pathogens can negatively affect seed-based eelgrass restoration projects. For example, seed loss due to Phytophthora and Halophytophthora infection can be as high as 40%20, which can have serious economic consequences as restoration is very costly with an estimated average price per seed of $0.1716.
We therefore aimed to develop a method that reduces seed loss by Phytophthora and/or Halophytophthora infection during seed storage. We conducted a full-factorial experiment treating stored seeds with three salinities (0.5, 10.0, 25.0 ppt) and three copper sulphate concentrations (0.0, 0.2, 2.0 ppm). Copper-containing compounds are known as effective fungicides counteracting Phytophthora infection of agricultural crops25,26. In addition, low salinity treatment may be a cost-effective and natural method of reducing Phytophthora infection, as shown for terrestrial crops due to enhanced susceptibility of salt-stressed plants and increased Phytophthora sporangium production in high salinity conditions27,28,29.
Results
Seed quality tests prior to the experiment
Eighty-six percent of all seeds used in the experiment was infected by either P. gemini (74%) or both P. gemini and Halophytophthora sp. Zostera (11%). None of the seeds were infected by Halophytophthora sp. Zostera alone. Germination tests showed that seeds displayed cotyledons of either brown or green colouration upon germination (Fig. 1). Germinated seeds with brown cotyledons were observed not to develop further into seedlings, whereas germinated seeds with green cotyledons did continue to develop. Infection affected colouration of cotyledons of germinated seeds: P. gemini and P. gemini + Halophytophthora sp. Zostera infected seeds had significantly more brown-coloured cotyledons than non-infected seeds (X2-test, p < 0.001 for each infection) (Fig. 2). Of all P. gemini infected seeds, 48% of the seeds had brown-coloured cotyledons after germination, while only 6% had green coloured cotyledons (ratio 8:1). None of the seeds that were infected by both oomycetes displayed green cotyledons after germination. In contrast, only 10% of all non-infected seeds germinated with brown cotyledons and a majority of 65% with green cotyledons (ratio 1:7). Total infection significantly reduced green-coloured germination by 13× (GLMM, z = −5.525, p < 0.001).
Infected seeds germinated mostly with brown cotyledons, whereas non-infected seeds germinated green (n = 102, n = 15, n = 20 for P. gemini,P. gemini + Halophytophthora sp. Zostera and non-infected seeds respectively). P. gemini and P. gemini + Halophytophthora sp. Zostera infected seeds had significantly more brown-coloured cotyledons than non-infected seeds (X2-test, p < 0.001 for each infection).
Experimental results
In total, only 11% of all seeds was infected after the experiment, 6% by Halophytophthora sp. Zostera, 4% by P. gemini, and 1% by both P. gemini and Halophytophthora sp. Zostera. Total infection was significantly reduced by copper treatment (GLMM, z = −2.021, p = 0.043), by 9× in the 0.2 ppm copper treatment and by 6× in the 2.0 ppm copper treatment, with no significant difference between both copper treatments (Fig. 3). Thus, the 0.2 ppm copper sulphate treatment was just as effective reducing infection as the 2.0 ppm copper sulphate treatment. Of the non-copper treated seeds, 36% remained infected at the end of the experiment, implying a reduction in infection of 2.5× during storage without treatment. Low salinity (0.5 ppt) completely eliminated infection (GLMM, z = 2.208, p = 0.027): none of the 0.5 ppt treated seeds were infected by P. gemini or Halophytophthora sp. Zostera after the experiment. The 10.0 and 25.0 ppt salinity treatments had similar (14 and 16% respectively) infection rates (Fig. 3a).
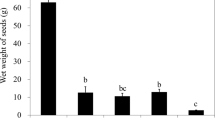
Infection significantly reduced Z. marina green seed germination by 1.5× (Fig. 4a): 31% of total infected seeds showed green germination versus 46% of the non-infected seeds (GLMM, z = −2.948, p = 0.0032). In contrast, more infected seeds germinated displayed brown cotyledons (39%) than non-infected seeds (26%) (GLMM, z = −3.975, p < 0.001) (Fig. 4b). In addition to infection, also salinity strongly affected germination. Seeds stored in freshwater (0.5 ppt) displayed significantly reduced green germination (Fig. 4a), <7.5% germinated with green cotyledons in contrast to >55% green germination of seeds stored at higher salinities (GLMM, z = 5.473, p < 0.001). Conversely, freshwater storage promoted brown seed germination: >50% of the 0.5 ppt stored seeds displayed brown germination versus only 7–30% of seeds stored at higher salinities (GLMM, z = −5.189, p < 0.001). Copper treatment affected neither green nor brown germination.
(a) Treatment effects on Green seed germination and (b) treatment effects on brown seed germination. Infection significantly reduced germination (GLMM, z = −2.948, p = 0.0032) and infected seeds displayed more brown cotyledons than non-infected seeds (GLMM, z = −3.975, p < 0.001). Seeds stored in freshwater displayed significantly reduced germination (GLMM, z = 5.473, p < 0.001) and higher brown cotyledon levels upon germination (GLMM, z = −5.189, p < 0.001). Bars represent sem (n = 4).
Discussion
Restoration of eelgrass beds using seeds often suffers from low recruitment rates30,31,32,33. Large numbers of seed may be lost due to predation15,34, bioturbation35 and currents and waves36. In addition, the recently discovered Phytophthora and Halophytophthora species20 may also enhance seed loss owing to strongly reduced germination of infected seeds. Here, we present a successful, inexpensive, and simple method to strongly reduce infection (by 86%) in seeds stored for restoration purposes, using low concentrations of copper sulphate dissolved in seawater. Moreover, we also report on the first successful application of a fungicide on a Halophytophthora species. This has never been attempted before because these oomycetes, although related to Phytophthora species, are generally saprophytes, decomposing leaves in salt marshes and mangrove forests37,38,39.
The use of copper as a fungicide already started in 19th century France40. Although not known at the time, copper-based compounds are effective fungicides because they are lethal to sporangia, zoospores and chlamydospores of fungi, algae and oomycetes25,26. Nowadays, many copper-based chemicals are registered and commercially used to reduce crop loss from Phytophthora infection41,42,43. The most widely used copper-based agrochemicals include copper oxychloride, copper hydroxide and cuprous oxide. These chemicals, however, have low water solubility and are therefore applied in relative large amounts40. This may cause severe environmental damage because copper is toxic to most higher plants and animals above generally 0.20–0.30 μg g−1 DW44. Copper toxicity in plants may lead to lipid peroxidation, protein oxidation and DNA damage40. In addition, copper toxicity may induce iron deficiency leading to leaf chlorosis44. Thus, a fine balance exists between the use of copper-based chemicals for plant protection and phytotoxicity. Another copper-based compound, copper sulphate (CuSO4), often used in an aqueous medium as algicide in for instance ponds, fish tanks, and swimming pools25,45,46, has also been effectively used to eliminate Phytophthora propagules from natural streams25, and both copper and sulphate ions are naturally present in ecosystems. We therefore selected this fungicide/algicide for the potential treatment of infected Z. marina seeds stored for restoration purposes. Seed treatment with copper was successful and concentrations of 0.2 ppm were just as effective in reducing infection as 2.0 ppm treatments. Similar disease reduction was also found in Phytophthora cryptogea infected gerberas treated with a 0.28 ppm copper solution47. Both copper concentrations used in our experiment (0.2 ppm and 2.0 ppm) are most likely below copper toxicity levels for Z. marina because seed germination was not affected by these treatments. This is surprising, because Lyngby and Brix48 showed that copper concentrations of 5 μM (0.32 ppm) or more can be toxic to adult Z. marina plants by reducing growth and inducing leaf necrosis. Possibly, high copper concentrations are more detrimental to adult plants than to seeds. Nevertheless, we recommend a 0.2 ppm instead of a 2.0 ppm copper treatment to reduce Phytophthora and Halophytophthora infection of Z. marina seeds. In addition, we have used these copper treatments in containers where concentrations could be carefully controlled. We do not recommend application in the field as one would need to account for currents and tides and effective concentrations would likely only to be reached when applying large amounts of copper sulphate, potentially leading to environmental damage. Thus, seed treatment by copper sulphate is recommended only during storage for restoration purposes and not in a natural population in the field.
Results of salinity treatments on infection and seed germination of Z. marina seeds were mixed; although infection by Phytophthora and Halophytophthora was completely eliminated in fresh water (0.5 ppt), the quality of the germinated seeds (brown instead of green cotyledons) was also severely impaired. Reduction of infection by low salinities may be due to limited tolerance of halophile P. gemini and Halophytophthora sp. Zostera. However, preliminary tests showed growth of these oomycetes on a selective growth medium (ParpH) with sterilized freshwater (Meffert & Van Rijswick, pers. comm. 2016). In addition, P. inundata, a close relative of P. gemini, is known from both freshwater and marine environments24, indicating a broad salt tolerance. Therefore, we conclude it is more likely that P. gemini and Halophytophthora sp. Zostera were outcompeted by other microorganisms (fungi or algae) in freshwater, also because we observed a black fungus (Phialophora malorum) on the seed coats in the 0.5 ppt treatments (including 0.2 and 2.0 ppm copper treatments). P. malorum was only observed in the freshwater treatments and may not only have affected Phytophthora or Halophytophthora infection by competition, but potentially also reduced seed germination in those treatments. This, however, needs further investigation.
Our results are in line with the effects of salinity on Phytophthora infection in terrestrial crops. In those systems, increased salinity generally enhances the effects of Phytophthora infection27,28,29. This is due to enhanced susceptibility to pathogens as a result of salt-stress29, but also due to increased sporangium production of Phytophthora49 under saline conditions. A similar trend – lower P. gemini and Halophytophthora sp. Zostera infection at low salinities – indicates adaptation of these species to the marine environment50. Correspondingly, another oomycete pathogen adapted to saline conditions, Labyrinthula zosterae (wasting disease), is also inhibited by low salinities51,52. In addition, Z. marina appears more susceptible to Labyrinthula infection at high salinities53,54.
Reduced quality of the pre-seedling stage of freshwater-treated seeds indicates that storing seeds at very low salinities for a longer period (110 days in our case) is detrimental to seed quality. These results are in concert with the findings of Xu, et al.11 who reported severely impaired morphology and growth of Z. marina seedlings grown at salinities <20 ppt. Although germination tests were conducted at 20.0 ppt, negative effects of storing seeds in freshwater appeared to be lasting. In addition, storing Zostera seeds at low salinities may also increase seed loss by enhancing premature germination15,55,56. Taking this into account and because both infection and germination were similar in the 10.0 and 25.0 ppt salinity treatments, we recommend storing seeds for restoration purposes at higher (i.e. >20 ppt) salinities.
To conclude, we present a novel method to reduce Z. marina seed loss by Phytophthora and Halophytophthora infection in seeds to be used for restoration purposes.
Treatment with 0.2 ppm copper sulphate strongly reduced infection but did not affect seed quality and is therefore recommended during storage of infected seeds. Although storing seeds at low salinity (0.5 ppt) effectively reduced oomycete infection, storage under these conditions is not recommended since this treatment strongly impaired seed quality. Infection during storage has been documented from multiple countries (the Netherlands, Denmark, Sweden, USA) and seed loss due to infection may be as high as 40%20. These results may thus be applicable to all seed-based Z. marina restoration projects around the world8,9,11,15,55,57
Materials and Methods
Seed collection
Seed-bearing shoots where collected at low tide, in an intertidal mixed Zostera marina/Zostera noltii meadow at the German barrier island Sylt (54.799 °N, 8.296°E). Collection took place in early September 2015 and shoots were transported to the lab at 6 °C. Here, these shoots where placed in aerated water tanks (15 kg shoots per 10,000 L, 30 ppt salinity, ambient temperature and light conditions, closed circulation), allowing the seeds to further ripen. Seeds were collected from the bottom of the aerated water tanks after 3 weeks. Of these, we selected 360 seeds of medium to advanced maturity11 for the experiment. Another 30 seeds from 4 different storage containers (120 in total) were individually tested for P. gemini and/or Halophytophthora sp. Zostera infection before the start of the experiment using both visual and molecular techniques20.
Experimental set-up
To study the effects of both copper sulphate and salinity on Phytophthora and Halophytophthora infection of Z. marina seeds, we crossed three copper sulphate (CuSO4) treatments (0.0, 0.2, 2.0 ppm) with three salinity treatments (0.5, 10.0, 25.0 ppt), resulting in 9 different treatments. Copper sulphate concentrations were based upon minimal algicidal levels (0.2 ppm)58 and maximal copper concentrations (~2.0 ppm) advised for usage to control fungi in ponds and fish tanks with high alkalinity45. We used 4 replicates per treatment, using 0.47 L cylindrical pvc units that were closed at the bottom59. Each unit was equipped with a porous membrane in the lower half of the column, through which individual aeration was added to ensure total oxygenation of the water column. Through the same membrane, each experimental unit was connected to a peristaltic pump that continuously injected water from a basin with the assigned treatment, into each unit. Experimental units where placed in a large, empty tank, allowing excess water to overflow, thus creating a continuous flow-through system60,61,62 (Masterflex ® 7568–10 Peristaltic Tubing Pumps, Cole-Parmer, USA) with a refreshment rate of once every two days (10 ml h−1). Overflow water was discharged to a waste water container to ensure that water of the experimental units was never brought in contact with that of other units. Salinity treatments were prepared from deionized water and Tropic Marin synthetic sea salt63 and checked twice a week using a multi-probe meter (556 MPS, YSI). Copper sulphate (CuSO4) was added to the treatment water basin and measured every week using a Copper checker (Hanna, HC – HI702) and adjusted if necessary. At the start of the experiment, Copper concentrations decreased rapidly as a result of adsorption of the copper ions to the treatment basins, thus requiring regular corrections by addition of CuSO4. However, after an initial charging phase of two weeks, copper concentrations remained constant. We placed 10 seeds in a nylon mesh bag (0.1 mm) attached to a glass bead anchor in the middle of every experimental unit at the start of the experiment. The entire experiment was conducted in a dark, climate controlled room at 5 °C to ensure optimal seed storage conditions8,9,55,57 and to prevent premature germination15.
Phytophthora and Halophytophthora analysis
After 105 days of incubation, the experiment was terminated and seeds were retrieved from the mesh bags and stored in 20 ml vials with 5 ml water from the assigned treatment. These samples, 360 seeds in total, were immediately transferred to the lab for Phytophthora and Halophytophthora analysis. Next, seeds were individually placed (one seed per well) on sterile 12 wells tissue culture plates with a growth area of 3.8 cm2 per well with ParpH selective growth medium64, see Govers, et al.20 for a visual representation of this method. ParpH is an oomycete-selective growth agar growth medium to which selected antibiotics are added to promote growth of PhytophthoraHalophytophthora, and Pythium species and to suppress growth of non-pythiaceous fungi64. Seeds were incubated on ParpH for 4 weeks, with a natural daylight cycle at room temperature (18–20 °C) with 2–3 ml of artificial seawater added to every well. After 3 and 7 days, presence of P. gemini and/or Halophytophthora sp. Zostera was individually scored for each seed by identification based on colony morphology20. In addition, seed germination and cotyledon colour (brown/green) of seeds incubated on ParpH with seawater was score daily up to four weeks of seed incubation. Oomycete species identification was also performed by sequence analysis of the internal transcribed spacer regions (ITS1 and ITS2) of the ribosomal DNA gene repeat on 4 samples of colonies additionally grown on cherry decoction agar (CHA)24. See Govers, et al.20 for more details on the visual and molecular identification methods.
Statistical analysis
Infection (based on visual assessment on parpH) and germination were analyzed by Generalized linear mixed models with binomial distribution (GLMM, lme4 package in R 3.2.3). We included treatments (salinity, copper addition) as fixed factors and experimental unit as random factors in our models. First, we tested the complete model with all treatments and interactions and then stepwise reduced the model by excluding non-significant interactions, starting with the most complex interactions. We hereby reduced the statistical model until only significant factors were left65. We used Tukey HSD posthoc tests to separate treatment effects. We report GLMM results as z values and P values. Effects of infection on cotyledon colouration were additionally tested by Chi-squared tests. P values < 0.05 were considered statistically significant.
Additional Information
How to cite this article: Govers, L. L. et al. Copper treatment during storage reduces Phytophthora and Halophytophthora infection of Zostera marina seeds used for restoration. Sci. Rep. 7, 43172; doi: 10.1038/srep43172 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
van der Zee, E. et al. How habitat-modifying organisms structure the food web of two coastal ecosystems. Proc. R. Soc. B, doi: 10.1098/rspb.2015.2326 (2016).
Fourqurean, J. W. et al. Seagrass ecosystems as a globally significant carbon stock. Nature Geoscience 5, 505–509, doi: 10.1038/ngeo1477 (2012).
Christianen, M. J. A. et al. Low-canopy seagrass beds still provide important coastal protection services. Plos One 8, e62413, doi: 10.1371/journal.pone.0062413 (2013).
Orth, R. J. et al. A Global Crisis for Seagrass Ecosystems. Bioscience 56, 987–996 (2006).
Waycott, M. et al. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proceedings of the National Academy of Sciences 106, 12377–12381, doi: 10.1073/pnas.0905620106 (2009).
van Katwijk, M. M. et al. Global analysis of seagrass restoration: the importance of large-scale planting. Journal of Applied Ecology 53, 567–578, doi: 10.1111/1365-2664.12562 (2016).
van Katwijk, M. M. et al. Guidelines for seagrass restoration: Importance of habitat selection and donor population, spreading of risks, and ecosystem engineering effects. Marine Pollution Bulletin 58, 179–188, doi: 10.1016/j.marpolbul.2008.09.028 (2009).
Tanner, C. E. & Parham, T. Growing Zostera marina (eelgrass) from seeds in land-based culture systems for use in restoration projects. Restoration Ecology 18, 527–537, doi: 10.1111/j.1526-100X.2010.00693.x (2010).
Marion, S. R. & Orth, R. J. Innovative Techniques for Large-scale Seagrass Restoration Using Zostera marina (eelgrass) Seeds. Restoration Ecology 18, 514–526, doi: 10.1111/j.1526-100X.2010.00692.x (2010).
Orth, R. J., Luckenbach, M. L., Marion, S. R., Moore, K. A. & Wilcox, D. J. Seagrass recovery in the Delmarva Coastal Bays, USA. Aquatic Botany 84, 26–36, doi: 10.1016/j.aquabot.2005.07.007 (2006).
Xu, S. et al. Salinity and temperature significantly influence seed germination, seedling establishment, and seedling growth of the eelgras Zostera marina L. PeerJ 4, doi: 10.7717/peerj.2697 (2016).
Yang, C. J. et al. Assesment of the establishment success of Zostera marina (eelgrass) from seeds in natural waters: implications for large-scale restoration. Ecological Engineering 92, 1–9, doi: 10.1016/j.ecoleng.2016.03.039 (2016).
Traber, M., Granger, S. & Nixon, S. W. Mechanical seeder provides alternative method for restoring eelgrass habitat (Rhode Island). Ecological Restoration 21, 213–214 (2003).
Pickerell, C., Scott, S. & Wyllie-Echeverria, S. Buoy-deployed seeding: demonstration of a new eelgrass (Zostera marina L.) planting method. Ecological Engineering 25, 127–136 (2005).
Infantes, E., Eriander, L. & Moksnes, P. O. Eelgrass (Zostera marina) restoration on the west coast of Sweden using seeds. Marine Ecology Progress Series 546, 31–45, doi: 10.3354/meps11615 (2016).
Busch, K. E. et al. Large-scale Zostera marina (eelgrass) restoration in Chesapeake Bay, Maryland, USA. Part I: a comparison of techniques and associated costs. Restoration Ecology 18, 490–500, doi: 10.1111/j.1526-100X.2010.00690.x (2010).
Ort, B. S. et al. Conservation of eelgrass (Zostera marina) genetic diversity in a mecosm-based restoration experiment. Plos One 9, doi: 10.1371/journal.pone.0089316.g001 (2015).
Reynolds, L. K., Waycott, M. & McGlathery, K. J. Restoration recovers population structure and landscape genetic connectivity in a dispersal-limited ecosystem. Journal of Ecology 101, 1288–1297, doi: 10.1111/1365-2745.12116 (2013).
Marion, S. R. & Orth, R. J. Factors influencing seedling establishment rates in Zostera marina and their implications for seagrass restoration. Restoration Ecology 18, 549–559 (2010).
Govers, L. L. et al. Marine Phytophthora species can hamper conservation and restoration of vegetated coastal ecosystems. Proc. R. Soc. B 283, 20160812, doi: 10.1098/rsbp/2016.0812 (2016).
Haverkort, A. J. et al. Societal costs of late blight in potato and prospects of durable resistance through cisgenic modification. Potato Research 51, 47–57 (2008).
Rizzo, D. M. & Garbelotto, M. Sudden Oak Death: Endangering California and Oregon Forest Ecosystems. Frontiers in Ecology and the Environment 1, 197–204 (2003).
Weste, G. & Marks, G. C. The Biology of Phytophthora cinnamomi in australasian forests. Annual Reviews in Phytopathology 25, 207–229 (1987).
Man in ‘t Veld, W. A., Rosendahl, K. C., Brouwer, H. & de Cock, A. W. Phytophthora gemini sp. nov., a new species isolated from the halophilic plant Zostera marina in the Netherlands. Fungal biology 115, 724–732, doi: 10.1016/j.funbio.2011.05.006 (2011).
Meadows, I. M., Colburn, G. C. & Jeffers, S. N. Evaluation of a copper hydroxide-based algicide to eliminate propagules of Phytophthora spp. in naturally infested streams in South Carolina, USA: a preliminary report. New Zealand Journal of Forestry Science 41S, S3–S5 (2011).
Howard, K., Colquhoun, I. J. & Hardy, G. The potential of copper sulfate to control Phytophthora cinnamomi during bauxite mining in Western Australia. Australasian Plant Pathology 27, 51–58 (1998).
Sanogo, S. Response of Chile pepper to Phytophthora capsici in relation to soil salinity. Plant Disease 88, 205–209 (2004).
Blaker, N. S. & MacDonald, J. D. The role of salinity in the development of Phytophthora root rot of citrus. Phytopathology 76, 970–975 (1986).
Snapp, S. S., Shennan, C. & Van Bruggen, A. H. C. Effects of salinity on severity of infection by Phytophthora parasitica Dast., ion concentrations and growth of tomato Lycopersicon esculentum Mill. New Phytologist 119, 275–284 (1991).
Eriander, L., Infantes, E., Olofsson, M., Olsen, J. L. & Moksnes, P. O. Assessing methods for restoration of eelgrass (Zostera marina L.) in a cold temperate region. Journal of Experimental Marine Biology and Ecology 479, 76–88 (2016).
Golden, R. R. et al. Large-Scale Zostera marina (eelgrass) Restoration in Chesapeake Bay, Maryland, USA. Part II: A Comparison of Restoration Methods in the Patuxent and Potomac Rivers. Restoration Ecology 18, 501–513, doi: 10.1111/j.1526-100X.2010.00691.x (2010).
Harwell, M. C. & Orth, R. J. Eelgrass (Zostera marina L.) seed protection for field experiments and implications for large-scale restoration. Aquatic Botany 64, 51–61 (1999).
Orth, R. J., Fishman, J. R., Harwell, M. C. & Marion, S. R. Seed-density effects on germination and initial seedling establishment in eelgras Zostera marina in the Chesapeake Bay region. Marine Ecology Progress Series 250, 71–79 (2003).
Infantes, E., Crouzy, C. & Moksnes, P. O. Seed Predation by the Shore Crab Carcinus maenas: A Positive Feedback Preventing Eelgrass Recovery? PLoS One 11, e0168128, doi: 10.1371/journal.pone.0168128 (2016).
Delefosse, M. & Kristensen, E. Burial of Zostera marina seeds in sediment inhabited by three polychaetes: laboratory and field studies. J. Sea Res. 71, 41–49, doi: 10.1016/j.seares.2012.04.006 (2012).
Van Katwijk, M. M. & Wijgergangs, L. Effects of locally varying exposure, sediment type and low-tide water cover on recruitment from seed. Aquatic Botany 80, 1–12, doi: 10.1016/j.aquabot.2004.04.003 (2004).
Nakagiri, A. In Aquatic Mycology across the MilleniumVol. 5 Fungal Diversity (eds Hyde, K. D., Ho, W. H. & Pointing, S. B. ) (2000).
Ho, H. H. & Jong, S. C. Halophytophthora gen. nov., a new member of the family Pythiaceae. Mycotaxon 36, 377–382 (1990).
Newell, S. Y. Established and potential impacts of eukaryotic mycelial decomposers in marine/terrestrial ecotones. Journal of Experimental Marine Biology and Ecology 200, 187–206 (1996).
Giannousi, K., Avramidis, I. & Dendrinou-Samara, C. Synthesis, characterization and evaluation of copper based nanoparticles as agrochemicals against Phytophthora infestans . RSC Advances 3, 21743–21752 (2013).
Daniell, I. R. & Falk, C. L. Economic comparison of Phytophthora root rot control methods. Crop Protection 13, 331–336 (1994).
Biles, C. L., Lindsey, D. L. & Liddell, C. M. Control of Phytophthora root rot of chile peppers by irrigation practices and fungicides. Crop Protection 11, 225–228 (1992).
Kousik, C. S. et al. Effect of cultural practices and fungicides of Phytophthora fruit rot of watermelon in the Carolinas. Crop Protection 30, 888–894, doi: 10.1016/j.cropro.2011.03.012 (2011).
Marschner, H. Mineral nutrition of higher plants. 2nd edn, (Academic Press, 1995).
Masser, M. P. & Jensen, J. W. Calculating treatments for ponds an tanks. (Southern Regional Aquaculture Center (SRAC), 1991).
Fitzgerald, G. P. & Faust, S. L. Factors affecting algicidal and algistatic properties of copper. Applied Microbiology 11, 345–351 (1963).
Toppe, B. & Thinggaard, K. Prevention of Phytophthora root rot in Gerbera by increasing copper ion concentration in the nutrient solution. European Journal of Plant Pathology 104, 359–366 (1998).
Lyngby, J. E. & Brix, H. The uptake of heavy metals in eelgrass Zostera marina and their effect on growth. Ecological Bulletins 36, 81–89 (1984).
Bouchibi, N., Van Bruggen, A. H. C. & MacDonald, J. C. Effects of ion concentration and sodium:calcium ratio of a nutrient solution on Phytophthora root rot of zoospore motility and viability of Phytophthora parasitica . Phytopathology 80, 1323–1336 (1990).
Leaño, E. M., Jones, E. B. G. & Vrijmoed, L. L. P. Why are Halophytophthora species well adapted to mangrove habitats? Fungal Diversity 5, 131–151 (2000).
Muehlstein, L. K., Porter, D. & Short, F. T. Labyrinthula sp., a marine slime mold producing the symptoms of wasting disease in eelgrass, Zostera marina . Marine Biology 99, 465–472 (1988).
Vergeer, L. H. T., Aarts, T. L. & DeGroot, J. D. The wasting disease and the effect of abiotic factors (light-intensity, temperature, salinity) and infection with Labyrinthula zosterae on the phenolic content of Zostera marina shoots. Aquatic Botany 51, 35–44 (1995).
McKone, K. L. & Tanner, C. E. Role of salinity in the susceptibility of eelgras Zostera marina to the wasting disease pathogen Labyrinthula zosterae . Marine Ecology Progress Series 377, 123–130 (2009).
van Katwijk, M. M., Vergeer, L. H. T., Schmitz, G. H. W. & Roelofs, J. G. M. Effects of salinity and nutrient loads and their interaction on Zostera marina . Marine Ecology Progress Series 190, 155–165 (1999).
Pan, J. et al. An effective method for collecting and storing seeds from Zostera marina (Eelgrass) in the Yellow Sea, China. Restoration Ecology 22, 716–722 (2014).
Conacher, C. A., Poiner, I. R., Butler, J., Pun, S. & Tree, D. J. Germination, storage and viability testing of seeds of Zostera capricorni Aschers from a tropical bay in Australia. Aquatic Botany 49, 47–58 (1994).
Kishima, J., Harada, S. & Sakurai, R. Suitable water temperature for seed storage of Zostera japonica for subtropical seagrass bed restoration. Ecological Engineering 37, 1416–1419 (2011).
Gibson, C. E. The algicidal effect of copper on a green and a blue-green alga and some ecological applications. Journal of Applied Ecology 9, 513–518 (1972).
Van der Heide, T. et al. A three-stage symbiosis forms the foundation of seagrass ecosystems. Science 336, 1432–1434 (2012).
Banach, A. M. et al. Effects of summer flooding on floodplain biogeochemistry in Poland; implications for increased flooding frequency. Biogeochemistry 92, 247–262, doi: 10.1007/s10533-009-9291-2 (2009).
Fritz, C., Lamers, L. P. M., Riaz, M., van den Berg, L. J. L. & Elzenga, T. J. T. M. Sphagnum mosses - masters of efficient N-uptake while avoiding intoxication. Plos One 9, e79991, doi: 10.1371/journal.pone.0079991 (2014).
Van der Heide, T. et al. Toxicity of reduced nitrogen in eelgrass (Zostera marina) is highly dependent on shoot density and pH. Oecologia 158, 411–419, doi: 10.1007/s00442-008-1155-2 (2008).
Govers, L. L. et al. Seagrasses are negatively affected by organic matter loading and Arenicola marina activity in a laboratory experiment. Oecologia 175, 677–685, doi: 10.1007/s00442-014-2916-8 (2014).
Jeffers, S. N. & Martin, S. B. Comparison of two media selective for Phytophthora and Pythium species. Plant Disease 70, 1038–1043 (1986).
Crawley, M. J. The R Book. (Wiley, 2012).
Acknowledgements
We thank Remco de Nooij for technical support and are grateful to volunteers of Natuurmonumenten for collecting seed material used for restoration purposes of which we used a small sample in our experiment. This study was funded by Natuurmonumenten.
Author information
Authors and Affiliations
Contributions
L.L.G., E.M.v.d.Z., T.v.d.H. conceived the study; L.L.G., E.M.v.d.Z., J.H.T.H., J.P.M., P.C.M.v.R. and W.A.M.i.t.V. conducted the practical work; L.L.G., E.M.v.d.Z. performed analyses; L.L.G. and T.v.d.H. drafted the manuscript. All authors contributed to improve the manuscript and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Govers, L., van der Zee, E., Meffert, J. et al. Copper treatment during storage reduces Phytophthora and Halophytophthora infection of Zostera marina seeds used for restoration. Sci Rep 7, 43172 (2017). https://doi.org/10.1038/srep43172
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep43172
This article is cited by
-
Seed selection and storage with nano-silver and copper as potential antibacterial agents for the seagrass Zostera marina: implications for habitat restoration
Scientific Reports (2019)
-
Multiple Halophytophthora spp. and Phytophthora spp. including P. gemini, P. inundata and P. chesapeakensis sp. nov. isolated from the seagrass Zostera marina in the Northern hemisphere
European Journal of Plant Pathology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.