Abstract
Fluorinated amino acids serve as valuable biological probes, by reporting on local protein structure and dynamics through 19F NMR chemical shifts. 2-fluorohistidine and 4-fluorohistidine, studied here with DFT methods, have even more capabilities for biophysical studies, as their altered pKa values, relative to histidine, allow for studies of the role of proton transfer and tautomeric state in enzymatic mechanisms. Considering the two tautomeric forms of histidine, it was found that 2-fluorohistidine primarily forms the common (for histidine) τ-tautomer at neutral pH, while 4-fluorohistidine exclusively forms the less common π-tautomer. This suggests the two isomers of fluorohistidine can also serve as probes of tautomeric form within biomolecules, both by monitoring NMR chemical shifts and by potential perturbation of the tautomeric equilibrium within biomolecules. Fluorine also enables assignment of tautomeric states in crystal structures. The differences in experimental pKa values between the isomers was found to arise from solvation effects, providing insight into the polarization and molecular properties of each isomer. Results also encompass 13C and 19F NMR chemical shifts, from both tautomers of 2-fluorohistidine and 4-fluorohistidine in a number of different environments. This work can serve as a guide for interpretation of spectroscopic results in biophysical studies employing 2-fluorohistidine and 4-fluorohistidine.
Similar content being viewed by others
Introduction
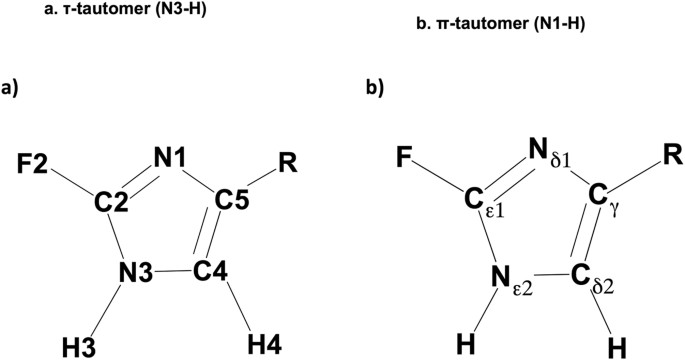
Histidine is one of the most chemically versatile amino acids, with the neutral imidazole ring having the potential to act as Lewis acid, Lewis base, hydrogen bond donor, and hydrogen bond acceptor. Further adding to its chemical diversity is the fact that the imidazole side chain of histidine can exist in two tautomeric states: the τ (N3-H or Nε2-H) and π (N1-H or Nδ1-H) tautomers, as shown in Fig. 1. Likely as a consequence of this chemical “flexibility”, histidine is present in ~50% of enzyme active sites1. The pKa (~6) of the imidazole ring in histidine is close to physiological pH, which makes it an important amino acid in protein function2,3,4,5. The fluorinated analogues of histidine, which exist as two different regioisomers, have dramatically lower pKa values than canonical histidine: ~1 (aqueous) for 2-fluorohistidine (2-FHis) and ~2 (aqueous) to >3 (proteins) for 4-fluorohistidine (4-FHis). This paper focuses on the properties of 2-FHis and 4-FHis, shown in both tautomeric forms in Fig. 1, including the origin of the differences in pKa between the two regioisomers. It also serves as a guide for use and spectroscopic interpretation of fluorohistidine in biophysical studies.
Multiple methods for the synthesis of fluorohistidines have been developed6,7,8,9,10. The ability to incorporate these analogues of histidine into proteins has facilitated mechanistic studies of proton transfer (discussed below) and characterization of protein structure using 19F-NMR11,12,13,14,15,16,17,18,19,20. Interestingly, the fluorine chemical shifts of 2- and 4-fluorohistidine exhibit anomalous pH-dependencies: 2-FHis is deshielded upon protonation, while the 19F in 4-FHis becomes more shielded at low pH21. We recently used electronic structure calculations to show that electron delocalization/localization appears to give rise to the differences in 19F shielding22.
Both 2-FHis and 4-FHis have been successfully incorporated into proteins via chemical or biosynthetic methods3,23,24,25,26,27,28. Fluorohistidine-labeling of proteins has provided valuable structural and mechanistic information in several instances23,25,29,30, and has great potential for future work. Studies by Eichler et al. showed that 2-FHis, and to a lesser extent 4-FHis, could be biosynthetically incorporated through protein expression in E. coli30. Prior to this, Jackson et al., in an amazing effort, chemically synthesized ribonuclease A, incorporating 4-fluorohistidine to show that the pH-dependence of enzyme activity could be shifted to lower pH values29. In crystallography, the fluorine electron density at the 2 or 4 position of the imidazole ring in histidine provides a marker so that carbon and nitrogen nuclei can be distinguished. Based on this information and inferred hydrogen bonding sites from other heavy atoms, tautomeric forms and nitrogen identities (N1 vs. N3) of histidine can be assigned with low ambiguity in protein crystal structures, as seen in work by Bann and co-workers for anthrax protective antigen labeled with 2-fluorohistidine25.
The potential for different biophysical studies based on the molecular and spectroscopic properties of 2-fluorohistidine and 4-fluorohistidine is discussed in this paper. In particular, the fluorohistidine isomers are poised to be powerful probes of histidine protonation state (tautomeric form) within proteins. Our calculations indicate that the τ-tautomer of 2-FHis is favored in the neutral state, while the π- tautomer is strongly favored for 4-FHis. Meanwhile, in canonical histidine, the τ-tautomer is more stable than the π-tautomer in the ratio of ~4:1 at pH = 831. In metalloproteins, different tautomeric forms of histidine interact with metals, such as zinc32,33, and local environments can shift the tautomeric equilibrium of histidine side chains through hydrogen bonding. Tautomerism appears to play a role in some enzymatic mechanisms34,35,36,37,38,39,40. For instance, the catalytic activity of human carbonic anhydrase II (hCAII) was investigated by Shimahara et al., revealing that the rate of proton transfer in the catalytic mechanism is highly efficient through a histidine tautomerization pathway at neutral pH34.
Traditionally, studies of histidine in protein function have used NMR spectroscopy to assign protonation state21,31,41,42,43,44,45,46,47,48,49,50. The tautomeric states of histidine can be distinguished by 15N NMR isotropic and anisotropic studies51 and C4 and C5 (Cδ2 and Cϒ) chemical shifts43. NMR experiments indicate rapid equilibration between tautomers52, and recent NMR studies of proteins have revealed hidden tautomeric states of histidine in transient protein conformations43. Work by Scheraga41, Oldfield and co-workers46 showed the utility of calculated and experimental 13C NMR shifts to evaluate tautomeric forms, and indicated that tautomeric form and chemical shifts of histidine are highly dependent on intramolecular hydrogen bonding. Results from DFT calculations presented below enable spectroscopic identification (via 19F, 13C chemical shifts) of the τ- and π-tautomers of 2- and 4-FHis in different environments (i.e., with or without intramolecular hydrogen bonding, in different dielectric environments). The nomenclature used to describe atoms in the imidazole ring throughout this paper is illustrated in Fig. 2a, alongside Fig. 2b, which shows labels used to identify the atoms of the histidine side chain in other literature reports.
Results
The tautomeric stabilities of 2-fluorohistidine and 4-fluorohistidine incorporated into the Gly-FHis-Gly tripeptide were calculated using multiple methods. These energy differences were found to be similar across a survey of electronic structure methods, as shown in Table S1 of Supplementary Information. Results discussed below are from BHandHLYP density functional, 6–311++G(d, p) basis set, and CPCM implicit solvation. The BHandHLYP functional has been recommended by Yu et al. for amino acids after a systematic study of multiple DFT methods53, and it can be seen here that it provides theoretical results for the fluorohistidine isomers that are in very good agreement with experimental data.
For 2-fluorohistidine, the τ-tautomer is more stable than the π-tautomer by 1.70 kcal/mol, resulting in a Boltzmann ratio of 94:6 τ:π tautomer at 310 K. The energy differences between the tautomers of 4-fluorohistidine are larger, with the π-tautomer being more stable than the τ-tautomer by an energy difference of 4.85 kcal/mol. The crystal structure of 2-fluorohistidine indicates protonation at the τ-tautomer position54. Although XRD data cannot conclusively give proton locations, the N3-C2 bond length compared to the N1-C2 bond length indicates protonation at N3 (τ-tautomer). In the case of 4-fluorohistidine, the stability of the π-tautomer can be rationalized by the position of electron-withdrawing fluorine in the imidazole ring: the nitrogen nearest to F4 is less basic (N3, unprotonated in the π tautomer), while the N1 position has higher electron density and is more basic.
Figure 3 illustrates how the experimental and calculated 19F NMR spectra support the calculated tautomeric stabilities, by plotting experimental titration data for 2-FHis and 4-FHis21 alongside the calculated 19F chemical shifts of both tautomers and protonated forms of the fluorohistidine isomers. In experiments, 2-fluorohistidine showed a downfield shift of 4.49 ppm upon protonation (from 59.80 ppm at pH ~7 to 64.29 ppm at pH ~1). In the same way, calculations for zwitterionic 2-fluorohistidine show deshielding of 6.0 ppm upon protonation for the τ-tautomer, while the signal shifts downfield by 9.7 ppm when the π-tautomer is protonated. For 4-fluorohistidine, the calculated chemical shift upon protonation of the π-tautomer is 2 ppm upfield (cf. 3.74 ppm upfield experimentally). In contrast, the calculated chemical shift change of the τ-tautomer (calculated to be highly unstable) is 8.6 ppm downfield, contrary to the direction of the 19F chemical shift observed in the experimental titration.
In summary, the simulated 19F NMR chemical shifts upon acid titration are in good agreement with experimental results for the tautomers that are calculated to be more stable. However, the less stable tautomers show divergence from experimental results in the simulated acid titration, with 4-FHis giving shifts in the opposite direction and 2-FHis showing a bigger change in chemical shifts than what is observed. Experimental support for the strong preference of 4-fluorohistidine for the π-tautomer is provided in a temperature-dependent 19F NMR spectrum, acquired in water (Fig. S2 of Supplemental Information). The thermal broadening and shifting of the 19F chemical shift as 4-fluorohistidine is heated to 75 °C is similar to the changes in chemical shift observed in the reference compound (trifluoroacetic acid or TFA). When the chemical shift of 4-fluorohistidine is calculated relative to TFA at every temperature point, the peak shifts by less than 0.1 ppm. If the relative energies of the 4-fluorohistidine tautomers were closer together (on the order of 1 kcal/mol or less), chemical exchange would lead to an upfield shift of the 19F peak as temperature increases, but there is no indication of this in the spectrum.
What are the effects of environment on tautomeric stabilities and fluorine chemical shifts?
Calculations were carried out on the Gly-FHis-Gly tripeptides in different dielectric environments, using CPCM-modeled acetonitrile and tetrahydrofuran (THF), to see the effect on tautomeric stabilities and fluorine chemical shifts. The results are provided in Table 1. Moving from an aqueous environment to a less polar environment slightly shifted the tautomeric equilibrium of 2-fluorohistidine toward the π-tautomer, with a relative stability of ~1.35 kcal/mol higher than the τ-tautomer in acetonitrile and THF (compared to 1.70 kcal/mol in water). These relatively low energy differences, in combination with a hydrogen-bonding environment that favors stabilization of the π-tautomer over the τ-tautomer, are supported by experimental results. In the crystal structure of 2-fluorohistidine labeled anthrax protective antigen, the π-tautomer of 2-fluorohistidine is indicated to be more stable in some interior sites (i.e. 2-FHis616 in the hydrophobic core of domain 4, stabilized by a hydrogen bond to N3)27. This is similar to histidine, which favors the π-tautomer in some protein environments. In less polar environments, the 19F chemical shifts of both tautomers of 2-fluorohistidine becomes slightly more shielded, with changes on the order of 0.6 ppm.
For 4-fluorohistidine, the relatively hydrophobic environment of THF shifts the tautomeric equilibrium slightly towards the τ-tautomer (3.91 kcal/mol higher than the π); acetonitrile shows a small change (4.75 kcal/mol, relative to 4.85 kcal/mol in water). Dielectric is seen to have very little influence on the 19F chemical shift of the τ-tautomer (less than 0.1 ppm), while less polar environments deshield the 19F nucleus of the 4-FHis π-tautomer from 19.21 ppm in water to 19.31 in acetonitrile and 20.16 ppm in THF. Such a “reverse” or abnormal fluorine chemical shift is discussed more in depth in our recent publication22.
Effects of intramolecular hydrogen bonding on fluorine chemical shifts and tautomeric stabilities
“Capped” histidine (N-acetyl-FHis-O-methyl ester or NAc-FHis-OMe), which has no intramolecular hydrogen bonds with the imidazole ring, was compared to zwitterionic histidine, for which the lowest-energy conformation in implicitly-modeled solvent has hydrogen bonds between the imidazole ring (side chain) and amino or carboxylate moieties of the amino acid. Figure S1 of Supplementary Information shows the lowest-energy optimized geometries of all tautomers of the capped and zwitterionic fluorohistidines. The relative energies and 19F chemical shifts for these species are presented in Table 2.
For 2-fluorohistidine, the effects of intramolecular hydrogen bonding are minor: the fluorine chemical shifts are affected more in the τ-tautomer (deshielded ~0.4 ppm with hydrogen bond) than the π-tautomer (increased shielding of ~0.1 ppm with hydrogen bond). Meanwhile, the relative tautomeric stabilities of 2-fluorohistidine change by <0.2 kcal/mol with the intramolecular hydrogen bond. In contrast, 4-fluorohistidine appears to be more sensitive to hydrogen bond effects: the fluorine chemical shift of the stable π-tautomer has a difference of >3 ppm between forms, with hydrogen-bonded zwitterionic 4-fluorohistidine being more shielded at 16.89 ppm than the non-hydrogen-bonded capped 4-fluorohistidine at 20.21 ppm. This is another “reverse” effect in the fluorine chemical shift of 4-FHis, since hydrogen bonding would be expected to inductively shift electron density away from the fluorine. Considering tautomeric stability, hydrogen bonding contributes 0.9 kcal/mol stabilization to the τ-tautomer of 4-fluorohistidine; yet the π-tautomer remains substantially more stable, by ~4 kcal/mol.
Finally, substitution at C5 of the fluoroimidazole was used to determine the sensitivity of tautomeric stabilities and fluorine chemical shifts to structure. C5 substitution can probe the sensitivity of fluorine chemical shifts to changes in primary and secondary structure, both of which give rise to changes in the wave function near the nucleus of interest. This is described by Oldfield and co-workers as a short-range contribution to chemical shifts, which propagates through the bonding framework55. In this case, the tripeptide was compared with 2F- and 4F-(5-methyl)-imidazole and -(5-trifluoromethyl)-imidazole, providing information about electron-donating and –withdrawing effects, respectively. The relative tautomeric stabilities and fluorine chemical shifts for this series are presented in Table 3.
As can be seen in Table 3, the relative stabilities of tautomers are dramatically affected by substitution at C5. In the case of 2-fluoro-(5-methyl)-imidazole, electron-donating substitution substantially stabilizes the π-tautomer, resulting in isoenergetic tautomers, while the electron-withdrawing CF3 group strongly destabilizes the π-tautomer, relative to the τ-tautomer. The effect is opposite for 4-fluorohistidine: electron-donating substituents further stabilize the π-tautomer, while electron-withdrawing leads to stabilization of the τ-tautomer. However, the energy differences between 4-fluoroimidazole tautomers always remain such that the τ-tautomer remains <1% population in equilibrium at biological or room temperatures.
The fluorine chemical shifts of 4-fluorohistidine are shown to depend dramatically on C5 substitution: electron donation causes an upfield shift (~5 ppm for both tautomers), while electron-withdrawing shifts the signal downfield (>15 ppm). The 19F NMR spectra are not as dramatically influenced by C5 substitution for 2-fluorohistidine, however. The differences between tripeptide and (5-methyl)-imidazole are slight (<0.2 ppm), and CF3, a rather large perturbation on electronic structure, shifts the signal 3 ppm and 6.7 ppm for the τ- and π-tautomers, respectively.
The origin of pKa differences between fluorohistidine isomers
The measured pKa values for aqueous fluorohistidine isomers indicate that 2-fluorohistidine has a lower pKa than 4-fluorohistidine (~2 for 4-FHis and ~1 for 2-FHis)21. The differences in relative tautomeric stabilities between 2-fluorohistidine and 4-fluorohistidine give rise to an entropic factor in the differences in pKa. Neutral 2-FHis is entropically stabilized by its thermally-accessible (tautomeric) states, whereas protonated imidazoliums and netural 4-fluorohistidine are not. Using the Boltzmann definition of entropy, the molar free energy of neutral fluorohistidine is reduced by RT ln 2 (drop in pKa of 0.3) when the two tautomers are isoenergetic (degeneracy = 2). In other cases, the closer the tautomers are in energy (and, hence, the closer their “microscopic” or individual pKa values)47,56, the greater the reduction in observed pKa value relative to microscopic pKa. In summary, the pKa of 2-fluorohistidine is predicted to be reduced by tautomerism, whereas the same effect is not predicted in 4-fluorohistidine.
Next, structural contributions to pKa differences between the most stable tautomers of 2-FHis and 4-FHis were considered. The origins are not immediately obvious by considering resonance and inductive effects. To study this computationally, the gas phase energies of capped 2-fluorohistidine and 4-fluorohistidine, and their conjugate acids were calculated and are presented in Table S2 of SI. The gas phase basicities (Econjugate acid − Eneutral) of the two isomers were found to be virtually identical: within 0.2 kcal/mol of one another. Therefore, solvation appears to be a driving force for the differences in pKa. Indeed, calculations confirm this, with protonation of aqueous 4-fluorohistidine being more favorable than protonation of 2-fluorohistidine by a difference of 1.4 kcal/mol. This is in very good agreement with the pKa difference of ~1–2 reported for the isomers21, since a pKa difference of 1 corresponds to a free energy difference of 1.37 kcal/mol at 300 K.
Solute polarity is a favorable driving force for solvation in water. Considering the apparent polarity of fluorohistidines, by examining their dipole moments (Table 4), it would appear there is a greater solvation driving force for protonation of 2-fluorohistidine than 4-fluorohistidine. In fact, if approximating solvation energy by the Onsager model, in which ΔGsolvation is proportional to μ2 within the same solvation environment57, one would predict the solvated protonation of 2-fluorohistidine to be 1.5 times more favorable. This would give rise to a higher pKa for 2-fluorohistidine than 4-fluorohistidine, the opposite of what is observed and calculated. If the dipole moments cannot explain trends in solvation, the charge distributions must hold the answer.
The key to understanding how solvation energies affect pKa lies in the unexpectedly favorable solvation energy of neutral 2-fluorohistidine, which is very similar to ΔEsolv of 4-fluorohistidine (given in Table S2 of Supplementary Information), in spite of dissimilar dipole moments. Of course, the value of ΔEsolv depends on the charge distribution within the molecule. The charges for the imidazole side chains of protonated and neutral fluorohistidine tripeptides are presented in Fig. 4. The charges are colour-coded, with green negative charges, red positive charges, and dark shading for near-neutral atoms.
Calculated charges for imidazole side chains in Gly-FHis-Gly peptides for (a) τ-tautomer of 2-fluorohistidine (b) protonated form of 2-fluorohistidine (c) π-tautomer of 2-fluorohistidine; (d) τ-tautomer of histidine (e) protonated form of histidine (f) π-tautomer of histidine; (g) π-tautomer of 4-fluorohistidine (h) protonated form of 4-fluorohistidine (i) τ-tautomer of 4-fluorohistidine.
The salient difference between 2-fluorohistidine (τ-tautomer) and all other species is the polarization of the C4-H4 bond, which has a carbon charge of −0.35 and hydrogen charge of +0.24. This charge difference is somewhat less, but on the order of, the N-H bond polarity seen in both fluorohistidine imidazole N-H bonds. The other charges, in the lone pair nitrogens, C-F bonds, and N-H bonds, are fairly similar among all species. Thus, the favourable solvation of the additional polar bond (C4-H4) in the imidazole ring (τ-tautomer only) of neutral 2-fluorohistidine appears to reduce the driving force in ΔEsolv for protonation, making 2-fluorohistidine more acidic than 4-fluorohistidine.
It is interesting to note that calculations with the same level of theory on canonical histidine indicate that the properties of 2-fluorohistidine track very well with those of histidine. The C4-H4 bond of histidine is also polar (charges of −0.275 for C and +0.201 for H) in the τ-tautomer, while the same bond in the π-tautomer is nonpolar (like 2-fluorohistidine). Recently, Kubarych, Pecoraro, and co-workers reported on the polarity of the Cδ2-H bond (on a Zn-bound histidine residue) in a de novo peptide, pointing out that the QM results are substantiated by microwave spectroscopy of imidazole58,59. It is seen below that the higher charge density of the C4 (Cδ2) carbon corresponds with a more shielded 13C NMR shift for this carbon in the τ-tautomer imidazole ring in both histidine and 2-fluorohistidine.
Spectroscopic signals of the tautomers of 2-fluorohistidine
The results presented above indicate the π-tautomer of 4-fluorohistidine is so much more stable than the τ-tautomer that the τ-tautomer is not predicted to be observed (<1%). However, calculations indicate small relative energy differences between the τ- and π-tautomers of 2-fluorohistidine, and both tautomers of 2-fluorohistidine have been observed in the crystal structure of labelled anthrax protective antigen27. Therefore, the discussion below focuses primarily on distinguishing among the two tautomers of 2-fluorohistidine in solution. 13C NMR chemical shifts for 2-fluorohistidine and 4-fluorohistidine in the Gly-FHis-Gly tripeptide are presented in Table 5.
Previous work by Oldfield46, Scheraga41, and co-workers have shown that the tautomeric forms of canonical histidine depend strongly on peptide environment (as seems to be the case for 2-fluorohistidine). Moreover, in their work, theoretical methods used to predict tautomeric fractions (relative stabilities) and the 13C NMR chemical shifts of each tautomer have been shown to be reliable. Oldfield and co-workers used a large data set of histidine-containing peptides and provided method-dependent scaling factors for each of the carbons in the histidine imidazole ring.
As is seen in canonical histidine, the C2 chemical shift of 2-fluorohistidine and 4-fluorohistidine are invariant with tautomeric form, while the C4 and C5 chemical shifts distinguish tautomers41,46. The C2 shift for both tautomers of 2-fluorohistidine is ~157 ppm: 157.4 ppm for the τ-tautomer and 156.9 ppm for the π-tautomer. The values of the C4 and C5 chemical shifts exhibit a wide spread for the τ-tautomer, with chemical shifts at 118.3 (C4/Cδ2) and 142.8 (C5/Cϒ) ppm. In contrast, the C4 and C5 chemical shifts are closer together in the π-tautomer, at 129.7 and 133.5 ppm, respectively. This range of chemical shifts between the 2-fluorohistidine tautomers (~11 ppm for C4 and ~10 ppm for C5) is on the order of the range observed for canonical histidine tautomers in a Gly-His-Gly peptide studied by Scheraga and co-workers41.
The range of chemical shifts for the carbons in the two tautomers of 2-fluorohistidine directly reflect the charge distribution on the carbons. The C4 carbon of the τ-tautomer is significantly more shielded (118.3 ppm) than the other carbons, corroborating its calculated negative charge (−0.4). As expected the C2 carbon bonded to fluorine is the most deshielded (~157 ppm for both tautomers), reflecting the polarity of the C-F bond. The polarization of C4 in the τ-tautomer coincides with polarization of the adjacent carbon, C5, which a relatively deshielded shift at 142.8 ppm (reflected in the calculated charge of +0.40). In contrast, there appears to be little polarization of C4 and C5 in the imidazole ring of the less polar π-tautomer, with chemical shifts that are close together, at 129.7 (C4) and 133.5 ppm (C5).
An experimental 13C NMR spectrum was acquired in deionized water for zwitterionic 2-fluorohistidine in water, and the imidazole ring shifts are as follows: 127.9 ppm, 113.6 ppm, 150.0 ppm for C5, C4 and C2 carbons respectively. The C5, C4, and C2 shifts for capped 2-fluorohistidine (close in structure to the aqueous zwitterion), provided in Table S3 of Supplementary Information, are calculated to be: 141.4 ppm, 118.9 ppm, 157.1 ppm respectively. Using a scaling factor approach in the same vein as Oldfield and co-workers, recommended scaling factors for BHandHLYP/6–311 ++ G(d, p) calculated 13C chemical shifts of fluorohistidines are as follows: 1.11, 1.05, 1.05 (C5, C4, C2). This is quite good agreement, indicating that the BHandHLYP functional may be a fairly reliable method for 13C chemical shifts (as it is for 19F).
Although 13C chemical shifts should not be needed to differentiate the tautomers of 4-fluorohistidine, given that the τ-tautomer is considerably more unstable, it is worth remarking on the predicted spectra. The π-tautomer of 4-fluorohistidine exhibits a broader spread in carbon chemical shifts than the τ-tautomer. We have recently published the experimental 13C spectrum of aqueous 4-fluorohistidine, and calculated values of the π-tautomer are in good agreement60. The C5 carbon has a relatively shielded nucleus at 101 ppm, and C4 (attached to fluorine) is the most deshielded at 152 ppm. The carbon chemical shifts again correlate with polarity and calculated charges: the C-F bond in the π-tautomer is more polar than the τ-tautomer, with a positive charge on the C4 carbon of +0.60 (−0.29 on F). The most shielded carbon (C5) is upfield at 101 ppm, corroborating a calculated charge of −0.10.
Discussion
It has been observed that typically, biosynthetic incorporation of 2-fluorohistidine is more successful than biosynthetic incorporation of 4-fluorohistidine23,29,30. Although it has been argued that 4-FHis may not be a good substrate for the HisRS of E. coli for steric reasons30, the results presented above indicate that the stabilities and the molecular properties (charge distribution, C-H bond polarity) of the tautomers of 2-fluorohistidine are remarkably similar to canonical histidine, making its substitution into biomolecules more amenable than 4-fluorohistidine. Of course, the pKa of 2-fluorohistidine is dramatically altered, providing a useful mechanistic probe for proton transfer steps in biological processes.
Studying Tautomeric Form in Proteins
For 2-fluorohistidine, the τ-tautomer is more stable in most environments, as it is for native histidine. In the crystal structure of 2-fluorohistidine-labelled anthrax protective antigen, a mixture of τ- and π-tautomers were observed, with the identified π-tautomer (His616) buried in the hydrophobic core of domain 4. This is consistent with our findings that lower dielectric environments shift the tautomeric equilibrium of 2-fluorohistidine toward the π-tautomer. Furthermore, hydrogen bonding was shown in our results to shift the equilibrium toward the π-tautomer, which also corroborates with the hydrogen-bond stabilized His616 in π form. So it is anticipated that local hydrogen bonds and dielectric environment will dictate tautomeric form of 2-fluorohistidine in proteins, as is often the case for canonical histidine in proteins. The introduction of the heavy atom (fluorine) in 2-fluorohistidine provides a distinctive marker in X-ray crystallography, allowing for histidine tautomeric form to be distinguished with low ambiguity in solved structures.
In contrast to 2-fluorohistidine, this work indicates that equilibrium tautomerism of 4-fluorohistidine is unfavorable in all environments. In all cases, the π-tautomer is favored by ~4 kcal/mol. This preference for the π-tautomer helps explain why 4-fluorohistidine could be successfully incorporated into the RNase A protein in work by Wells and co-workers29. Of the two active site histidines, His12 is indicated to be 100% π-tautomer37, while the tautomeric form of His119 during proton transfer is unknown (see our recent letter for further discussion)60. Having a viable enzyme with 4-FHis substitution may indicate the π-tautomer is the active form during mechanistic steps. Overall, these results indicate that fluorohistidines may be poised for studying the role of histidine tautomerization or protonation state in biological mechanisms, as the τ-tautomer of 2-fluorohistidine is more stable, but tautomeric form can easily be identified by crystallography in labeled proteins and via 19F and 13C NMR. Meanwhile the π-tautomer is strongly favored in 4-fluorohistidine, and the effect of this tautomeric stability perturbation on protein function can be evaluated.
19F NMR Spectra Reflect Protein Environment
Fluorine chemical shifts are noted for their high sensitivity to local environment. Differences in 1° and 2° structure in proteins may give rise to conformational diversity and different inductive effects on the imidazole side chain, that, in combination with varying dielectric and hydrogen bonding environments, result in the range of 19F signals observed for folded and unfolded proteins. For 4-fluorohistidine, differences in 1° and 2° structure in proteins may well be the primary source of changes in fluorine chemical shifts in the folded protein environment. The π-tautomer of 4-fluorohistidine appears to be quite sensitive to hydrogen bonding and inductive effects in the imidazole ring, exhibiting changes upward of 4 ppm for each perturbation. However, dielectric environment has a relatively minor effect on 4-fluorohistidine chemical shift (up to 1 ppm). All environmental effects result in much smaller fluorine chemical shift changes for both tautomers of 2-fluorohistidine: on the scale of 0.1–0.5 ppm per environmental change. This is in line with the findings of Gerig and co-workers, who postulated that diversity in fluorine chemical shifts in denatured/folded proteins arises from a sum of changes in chemical shift due to factors such as van der Waals interactions, hydrogen bonds, electric fields, and local magnetic anisotropies61.
Probing Proton Transfer in Proteins
The altered pKa values of fluorohistidines can be used to determine whether histidines act as general acids and bases in enzymes29. The differences in pKa between the two isomers, with 2-fluorohistidine being less basic than 4-fluorohistidine, can be attributed primarily to solvation effects. Calculated gas phase pKa values are quite similar, but the aqueous pKa of 4-fluorohistidine is higher than 2-fluorohistidine. This arises from favorable solvation of the polar charge distribution of 2-fluorohistidine (τ-tautomer) that reduces the driving force for protonation. The thermally-accessible tautomeric equilibrium of 2-fluorohistidine also lowers the pKa value of this isomer somewhat (up to 0.3 pKa units, when tautomers are isoenergetic). Since tautomerism is not favored by 4-fluorohistidine, the pKa value for this isomer is not lowered by tautomeric equilibrium. In RNase A, the pKa of 4-fluorohistidines in the active site were indicated to be over 3.5 (cf. ~2 in aqueous solution), showing the influence of protein environment29.
Conclusions
Fluorohistidines can contribute to biophysical studies of proteins in numerous ways. They have the potential to be powerful probes of tautomeric state and the contributions of tautomeric form to protein stability and enzyme mechanism. In crystallography, the fluorine atoms’ high electron density in the imidazole ring allows for definitive assignment of carbons and nitrogens, facilitating assignment of tautomeric form as well. The tautomeric stability of 2-fluorohistidine is predicted to shift with environment, with low dielectric environment shifting the tautomeric equilibrium toward the π-tautomer. While tautomeric form and tautomerism are acknowledged to be important factors in histidine’s chemical versatility and role in protein structure-function, identification of tautomeric form (at equilibrium and in rate-determining steps) has proven difficult in large biomolecular systems. 4-fluorohistidine is poised to serve as a powerful probe of the role of tautomeric form in structure and catalytic steps. In addition, its altered pKa provides a tool for determining how proton transfer affects catalysis, and at pH values that are less acidic than those required for 2-fluorohistidine labeled proteins.
Additionally, biosynthetic incorporation of either fluorohistidine isomer introduces a valuable spectroscopic label for 19F NMR studies. Fluorine NMR spectra provide vastly simplified information relative to traditional protein NMR spectra, with the ability to report on site-specific dynamics62,63 and ligand-binding events64. While the origin of changes in fluorine chemical shifts are not systematically understood, this work provides some insight on how environment influences chemical shift. The 19F signal of 2-fluorohistidine is shown to behave “normally”—in the way that would be predicted for 1H chemical shifts. Deshielding is observed with hydrogen bonds, higher dielectric, and nearby electron withdrawing groups. In contrast, the 19F chemical shifts of 4-fluorohistidine are “reverse”, with environmental changes that normally deshield nuclei (such as hydrogen bonding, higher dielectric) resulting in shielding of the fluorine nucleus. Fluorohistidines are poised to serve as powerful biophysical probes of protein structure-dynamics-mechanisms. This work can serve as a guide for interpretation of mechanistic experiments and spectroscopic data with fluorohistidine-labeled biomolecules.
Methods
Optimized geometries, frequencies, and NMR chemical shifts were calculated for Gly-FHis-Gly tripeptides and the following analogues, in order to simulate effects of peptide environment on fluoroimidazole ring properties: 2-fluorohistidine (2-FHis), 4-fluorohistidine (4-FHis), 2-fluoro-(5-methyl)-imidazole (2F-MeIm), 4-fluoro-(5-methyl)-imidazole (4F-MeIm), N-acetyl-2FHis-O-methyl ester (NAc-2FHis-OMe), N-acetyl-4FHis-O-methyl ester (NAc-4FHis-OMe), 2-fluoro-(5-trifluoromethyl)-imidazole (2F-CF3Im), 4-fluoro-(5-trifluoromethyl)-imidazole (4F-CF3Im). All quantum chemical calculations were performed using Gaussian 09 package65 with calculations set up in GaussView 566. BHandHLYP67 with 6–311++G(d, p) basis set is considered for all calculations. Initially, all the geometries were optimized to get a stable conformation, and frequencies were calculated to verify that geometries are at local energy minima. These geometries were used for calculating absolute shielding constants, energies and electrostatic potential charge distribution (by ChelpG method)68 at the same level of theory. Free energies were obtained from frequency calculations.
Solvation was modeled with the Self Consistent Reaction Field (SCRF) method, using the conductor polar continuum model (CPCM)69 to study the effect of dielectric environment/solvation on spectra and tautomeric stabilities. To estimate chemical shifts and molecular properties of buried residues (versus solvent-exposed side chains), aqueous (ε = 78.3) calculations are compared to those in tetrahydrofuran (THF, ε = 7.6) and acetonitrile (ε = 35.7) to see effects of less polar environments.
NMR shielding constants were calculated using Gauge Independent Atomic Orbital (GIAO)70,71 method, and are reported for 13C and 19F nuclei. The NMR chemical shifts (δ) reported for the compounds are calculated by the formula δcompound = σref − σcompound from the shielding value (σ) of the compound and shielding of the reference compound: C6F6 for 19F and tetramethylsilane (TMS) for 13C. Shielding tensors for the reference compound are calculated with BHandHLYP/6–311++G(d, p) method after optimizing the molecule with the same method. Experimental 13C NMR data for 2-fluorohistidine in water and variable-temperature 19F NMR data for 4-fluorohistidine was collected on a Varian Mercury VX 300 MHz NMR equipped with a Varian 300 SW/BB probe.
Additional Information
How to cite this article: Kasireddy, C. et al. The Biophysical Probes 2-fluorohistidine and 4-fluorohistidine: Spectroscopic Signatures and Molecular Properties. Sci. Rep. 7, 42651; doi: 10.1038/srep42651 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Shimba, N. et al. Quantitative identification of the protonation state of histidines in vitro and in vivo . Biochemistry-Us 42, 9227–9234, doi: 10.1021/bi0344679 (2003).
Salwiczek, M., Nyakatura, E. K., Gerling, U. I. M., Ye, S. J. & Koksch, B. Fluorinated amino acids: compatibility with native protein structures and effects on protein-protein interactions. Chem Soc Rev 41, 2135–2171, doi: 10.1039/c1cs15241f (2012).
Klein, D. C. et al. 2-Fluoro-L-Histidine, an Inhibitor of Enzyme-Induction. Mol Pharmacol 12, 720–730 (1976).
Grimsley, G. R., Scholtz, J. M. & Pace, C. N. A summary of the measured pK values of the ionizable groups in folded proteins. Protein Sci 18, 247–251, doi: 10.1002/pro.19 (2009).
Edgcomb, S. P. & Murphy, K. P. Variability in the pKa of histidine side-chains correlates with burial within proteins. Proteins 49, 1–6, doi: 10.1002/prot.10177 (2002).
Kirk, K. L. & Cohen, L. A. Photochemical Decomposition of Diazonium Fluoroborates - Application to Synthesis of Ring-Fluorinated Imidazoles. J Am Chem Soc 93, 3060–3061 (1971).
Kirk, K. L., Nagai, W. & Cohen, L. A. Photochemistry of Diazonium Salts. 2. Synthesis of 2-Fluoro-L-Histidine and 2-Fluorohistamine, and Halogen Lability of 2-Fluoroimidazoles. J Am Chem Soc 95, 8389–8392, doi: 10.1021/Ja00806a031 (1973).
Kirk, K. L. & Cohen, L. A. Photochemistry of Diazonium Salts. 1. Synthesis of 4-Fluoroimidazoles, 4-Fluorohistamine, and 4-Fluorohistidine. J Am Chem Soc 95, 4619–4624, doi: 10.1021/Ja00795a026 (1973).
Hajduch, J., Cramer, J. C. & Kirk, K. L. An enantioselective synthesis of (S)-4-fluorohistidine. J Fluorine Chem 129, 807–810, doi: 10.1016/j.jfluchem.2008.04.011 (2008).
Lipunova, G. N., Nosova, E. V. & Charushin, V. N. Fluoroimidazoles and their Heteroannelated Derivatives: Synthesis and Properties (Review). Chem Heterocycl Com+ 49, 1691–1714, doi: 10.1007/s10593-014-1422-6 (2014).
Gerig, J. T. Fluorine Nmr of Proteins. Prog Nucl Mag Res Sp 26, 293–370, doi: 10.1016/0079-6565(94)80009-X (1994).
Danielson, M. A. & Falke, J. J. Use of F-19 NMR to probe protein structure and conformational changes. Annu Rev Bioph Biom 25, 163–195, doi: 10.1146/Annurev.Bb.25.060196.001115 (1996).
Chen, H., Viel, S., Ziarelli, F. & Peng, L. F-19 NMR: a valuable tool for studying biological events. Chem Soc Rev 42, 7971–7982, doi: 10.1039/c3cs60129c (2013).
Sternberg, U., Klipfel, M., Grage, S. L., Witter, R. & Ulrich, A. S. Calculation of fluorine chemical shift tensors for the interpretation of oriented F-19-NMR spectra of gramicidin A in membranes. Phys Chem Chem Phys 11, 7048–7060, doi: 10.1039/b908236k (2009).
Pomerantz, W. C. et al. Profiling the Dynamic Interfaces of Fluorinated Transcription Complexes for Ligand Discovery and Characterization. Acs Chem Biol 7, 1345–1350, doi: 10.1021/cb3002733 (2012).
Urick, A. K. et al. Dual Screening of BPTF and Brd4 Using Protein-Observed Fluorine NMR Uncovers New Bromodomain Probe Molecules. Acs Chem Biol 10, 2246–2256, doi: 10.1021/acschembio.5b00483 (2015).
Li, C. G. et al. Protein F-19 NMR in Escherichia coli . J Am Chem Soc 132, 321–327, doi: 10.1021/ja907966n (2010).
Campos-Olivas, R., Aziz, R., Helms, G. L., Evans, J. N. S. & Gronenborn, A. M. Placement of F-19 into the center of GB1: effects on structure and stability. Febs Lett 517, 55–60, doi: Pii S0014-5793(02)02577-2 doi: 10.1016/S0014-5793(02)02577-2 (2002).
Isley, W. C., 3rd, Urick, A. K., Pomerantz, W. C. & Cramer, C. J. Prediction of (19)F NMR Chemical Shifts in Labeled Proteins: Computational Protocol and Case Study. Molecular pharmaceutics 13, 2376–2386, doi: 10.1021/acs.molpharmaceut.6b00137 (2016).
Wang, G. F., Li, C. G. & Pielak, G. J. F-19 NMR studies of alpha-synuclein-membrane interactions. Protein Sci 19, 1686–1691, doi: 10.1002/pro.449 (2010).
Yeh, H. J. C., Kirk, K. L., Cohen, L. A. & Cohen, J. S. F-19 and H-1 Nuclear Magnetic-Resonance Studies of Ring-Fluorinated Imidazoles and Histidines. J Chem Soc Perk T 2, 928–934, doi: 10.1039/P29750000928 (1975).
Kasireddy, C., Bann, J. G. & Mitchell-Koch, K. R. Demystifying fluorine chemical shifts: electronic structure calculations address origins of seemingly anomalous F-19-NMR spectra of fluorohistidine isomers and analogues. Phys Chem Chem Phys 17, 30606–30612, doi: 10.1039/c5cp05502d (2015).
Wimalasena, D. S. et al. Effect of 2-fluorohistidine labeling of the anthrax protective antigen on stability, pore formation, and translocation. Biochemistry-Us 46, 14928–14936, doi: 10.1021/bi701763z (2007).
Klein, D. C., Weller, J. L., Kirk, K. L. & Hartley, R. W. Incorporation of 2-Fluoro-L-Histidine into Cellular Protein. Mol Pharmacol 13, 1105–1110 (1977).
Rajapaksha, M. et al. pH effects on binding between the anthrax protective antigen and the host cellular receptor CMG2. Protein Sci 21, 1467–1480, doi: 10.1002/pro.2136 (2012).
Harris, M. E., Piccirilli, J. A. & York, D. M. Integration of kinetic isotope effect analyses to elucidate ribonuclease mechanism. Bba-Proteins Proteom 1854, 1801–1808, doi: 10.1016/j.bbapap.2015.04.022 (2015).
Wimalasena, D. S. et al. Evidence That Histidine Protonation of Receptor-Bound Anthrax Protective Antigen Is a Trigger for Pore Formation. Biochemistry-Us 49, 6973–6983, doi: 10.1021/bi100647z (2010).
Hu, L. et al. Comparison of the structural stability and dynamic properties of recombinant anthrax protective antigen and its 2-fluorohistidine-labeled analogue. J Pharm Sci-Us 101, 4118–4128, doi: 10.1002/jps.23294 (2012).
Jackson, D. Y. et al. A Designed Peptide Ligase for Total Synthesis of Ribonuclease-a with Unnatural Catalytic Residues. Science 266, 243–247, doi: 10.1126/Science.7939659 (1994).
Eichler, J. F., Cramer, J. C., Kirk, K. L. & Bann, J. G. Biosynthetic incorporation of fluorohistidine into proteins in E-coli: A new probe of macromolecular structure. Chembiochem 6, 2170–2173, doi: 10.1002/cbic.200500249 (2005).
Reynolds, W. F., Peat, I. R., Freedman, M. H. & Lyerla, J. R. Determination of Tautomeric Form of Imidazole Ring of L-Histidine in Basic Solution by C-13 Magnetic-Resonance Spectroscopy. J Am Chem Soc 95, 328–331, doi: 10.1021/Ja00783a006 (1973).
Karlin, S. & Zhu, Z. Y. Classification of mononuclear zinc metal sites in protein structures. Proc Natl Acad Sci USA 94, 14231–14236, doi: 10.1073/Pnas.94.26.14231 (1997).
Chakrabarti, P. Geometry of Interaction of Metal-Ions with Histidine-Residues in Protein Structures. Protein Eng 4, 57–63, doi: 10.1093/Protein/4.1.57 (1990).
Shimahara, H. et al. Tautomerism of histidine 64 associated with proton transfer in catalysis of carbonic anhydrase. J Biol Chem 282, 9646–9656, doi: 10.1074/jbc.M609679200 (2007).
Hass, M. A. S., Hansen, D. F., Christensen, H. E. M., Led, J. J. & Kay, L. E. Characterization of conformational exchange of a histidine side chain: Protonation, rotamerization, and tautomerization of His61 in plastocyanin from Anabaena variabilis. J Am Chem Soc 130, 8460–8470, doi: 10.1021/ja801330h (2008).
Pelton, J. G., Torchia, D. A., Meadow, N. D. & Roseman, S. Tautomeric States of the Active-Site Histidines of Phosphorylated and Unphosphorylated E-Coli-Iiiglc Using Heteronuclear 2d Nmr Techniques. Biophys J 64, A372–A372 (1993).
Walters, D. E. & Allerhand, A. Tautomeric States of the Histidine-Residues of Bovine Pancreatic Ribonuclease-a - Application of C-13 Nuclear Magnetic-Resonance Spectroscopy. J Biol Chem 255, 6200–6204 (1980).
Jewsbury, P. & Kitagawa, T. The Distal Residue Co Interaction in Carbonmonoxy Myoglobins - a Molecular-Dynamics Study of 2 Distal Histidine Tautomers. Biophys J 67, 2236–2250 (1994).
Singer, A. U. & FormanKay, J. D. pH titration studies of an SH2 domain-phosphopeptide complex: Unusual histidine and phosphate pK(a) values. Protein Sci 6, 1910–1919 (1997).
Garrett, D. S., Seok, Y. J., Peterkofsky, A., Clore, G. M. & Gronenborn, A. M. Identification by NMR of the binding surface for the histidine-containing phosphocarrier protein HPr on the N-terminal domain of enzyme I of the Escherichia coli phosphotransferase system. Biochemistry-Us 36, 4393–4398, doi: 10.1021/Bi970221q (1997).
Vila, J. A., Arnautova, Y. A., Vorobjev, Y. & Scheraga, H. A. Assessing the fractions of tautomeric forms of the imidazole ring of histidine in proteins as a function of pH. Proc Natl Acad Sci USA 108, 5602–5607, doi: 10.1073/pnas.1102373108 (2011).
Li, S. H. & Hong, M. Protonation, Tautomerization, and Rotameric Structure of Histidine: A Comprehensive Study by Magic-Angle-Spinning Solid-State NMR. J Am Chem Soc 133, 1534–1544, doi: 10.1021/ja108943n (2011).
Hansen, A. L. & Kay, L. E. Measurement of histidine pK(a) values and tautomer populations in invisible protein states. P Natl Acad Sci USA 111, E1705–E1712, doi: 10.1073/pnas.1400577111 (2014).
Wolff, N. et al. Histidine pK(a) shifts and changes of tautomeric states induced by the binding of gallium-protoporphyrin IX in the hemophore HasA(SM). Protein Sci 11, 757–765, doi: 10.1110/ps.3630102 (2002).
Bhattacharya, S., Sukits, S. F., MacLaughlin, K. L. & Lecomte, J. T. J. The tautomeric state of histidines in myoglobin. Biophys J 73, 3230–3240 (1997).
Cheng, F., Sun, H. H., Zhang, Y., Mukkamala, D. & Oldfield, E. Solid state C-13 NMR, crystallographic, and quantum chemical investigation of chemical shifts and hydrogen bonding in histidine dipeptides. J Am Chem Soc 127, 12544–12554, doi: 10.1021/ja051528c (2005).
Tanokura, M. H-1-Nmr Study on the Tautomerism of the Imidazole Ring of Histidine-Residues.1. Microscopic Pk Values and Molar Ratios of Tautomers in Histidine-Containing Peptides. Biochim Biophys Acta 742, 576–585, doi: 10.1016/0167-4838(83)90276-5 (1983).
Wasylishen, R. E. & Tomlinson, G. Ph-Dependence of C-13 Chemical-Shifts and C-13, H Coupling-Constants in Imidazole and L-Histidine. Biochem J 147, 605–607 (1975).
Shimba, N., Takahashi, H., Sakakura, M., Fujii, I. & Shimada, I. Determination of protonation and deprotonation forms and tautomeric states of histidine residues in large proteins using nitrogen-carbon J couplings in imidazole ring. J Am Chem Soc 120, 10988–10989, doi: 10.1021/Ja982153g (1998).
Wilbur, D. J. & Allerhand, A. Titration Behavior and Tautomeric States of Individual Histidine Residues of Myoglobins - Application of Natural Abundance C-13 Nuclear Magnetic-Resonance Spectroscopy. J Biol Chem 252, 4968–4975 (1977).
Munowitz, M., Bachovchin, W. W., Herzfeld, J., Dobson, C. M. & Griffin, R. G. Acid-Base and Tautomeric Equilibria in the Solid-State - N-15 Nmr-Spectroscopy of Histidine and Imidazole. J Am Chem Soc 104, 1192–1196, doi: 10.1021/Ja00369a007 (1982).
Blomberg, F., Maurer, W. & Ruterjans, H. Nuclear Magnetic-Resonance Investigation of N-15-Labeled Histidine in Aqueous-Solution. J Am Chem Soc 99, 8149–8159, doi: 10.1021/Ja00467a005 (1977).
Yu, W. et al. Comparison of some representative density functional theory and wave function theory methods for the studies of amino acids. Journal of computational chemistry 30, 589–600, doi: 10.1002/jcc.21091 (2009).
Andra, K. K., Bullinger, J. C., Bann, J. G. & Eichhorn, D. M. 2-Fluoro-L-histidine. Acta Crystallogr E 66, O2713–U1695, doi: 10.1107/S1600536810038663 (2010).
Dedios, A. C., Pearson, J. G. & Oldfield, E. Secondary and Tertiary Structural Effects on Protein Nmr Chemical-Shifts - an Abinitio Approach. Science 260, 1491–1496, doi: 10.1126/Science.8502992 (1993).
Khandogin, J. & Brooks, C. L. Constant pH molecular dynamics with proton tautomerism. Biophys J 89, 141–157, doi: 10.1529/biophysj.105.061341 (2005).
Onsager, L. Electric moments of molecules in liquids. J Am Chem Soc 58, 1486–1493, doi: 10.1021/Ja01299a050 (1936).
Ross, M. R. et al. Histidine Orientation Modulates the Structure and Dynamics of a de Novo Metalloenzyme Active Site. J Am Chem Soc 137, 10164–10176, doi: 10.1021/jacs.5b02840 (2015).
Christen, D., Griffiths, J. H. & Sheridan, J. The Microwave-Spectrum of Imidazole - Complete Structure and the Electron-Distribution from Nuclear-Quadrupole Coupling Tensors and Dipole-Moment Orientation. Z Naturforsch A 36, 1378–1385 (1981).
Kasireddy, C., Ellis, J. M., Bann, J. G. & Mitchell-Koch, K. R. Tautomeric stabilities of 4-fluorohistidine shed new light on mechanistic experiments with labeled ribonuclease A. Chem Phys Lett 666, 58–61, doi: 10.1016/j.cplett.2016.10.072 (2016).
Gerig, J. T. & Lau, E. Y. Origins of fluorine NMR chemical shifts in fluorine-containing protein’s. Biophys J 78, 9a–9a (2000).
Hoeltzli, S. D. & Frieden, C. Stopped-flow NMR spectroscopy: real-time unfolding studies of 6-19F-tryptophan-labeled Escherichia coli dihydrofolate reductase. Proc Natl Acad Sci USA 92, 9318–9322 (1995).
Hoeltzli, S. D. & Frieden, C. F-19 Nmr-Spectroscopy of [6-F-19]Tryptophan-Labeled Escherichia-Coli Dihydrofolate-Reductase - Equilibrium Folding and Ligand-Binding Studies. Biochemistry-Us 33, 5502–5509, doi: 10.1021/Bi00184a019 (1994).
Urick, A. K., Calle, L. P., Espinosa, J. F., Hu, H. & Pomerantz, W. C. Protein-Observed Fluorine NMR Is a Complementary Ligand Discovery Method to 1H CPMG Ligand-Observed NMR. Acs Chem Biol, doi: 10.1021/acschembio.6b00730 (2016).
Frisch, M. J. Gaussian 09, Gaussian, Inc., Wallingford, CT, USA (2009).
Dennington, R., Keith, T. A. & Millam, J. M. GaussView, Version 5, Semichem Inc., Shawnee Mission KS: USA, (2009).
Becke, A. D. A New Mixing of Hartree-Fock and Local Density-Functional Theories. J Chem Phys 98, 1372–1377, doi: 10.1063/1.464304 (1993).
Breneman, C. M. & Wiberg, K. B. Determining atom‐centered monopoles from molecular electrostatic potentials. The need for high sampling density in formamide conformational analysis. J Comput Chem 11, 361–373 (1990).
Barone, V. & Cossi, M. Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J Phys Chem A 102, 1995–2001, doi: 10.1021/Jp9716997 (1998).
Ditchfield, R. Self-Consistent Perturbation-Theory of Diamagnetism.1. Gauge-Invariant Lcao Method for Nmr Chemical-Shifts. Mol Phys 27, 789–807, doi: 10.1080/00268977400100711 (1974).
Wolinski, K., Hinton, J. F. & Pulay, P. Efficient Implementation of the Gauge-Independent Atomic Orbital Method for Nmr Chemical-Shift Calculations. J Am Chem Soc 112, 8251–8260, doi: 10.1021/Ja00179a005 (1990).
Acknowledgements
We are grateful to Dr. Christine Morales for a critical reading of the manuscript. Financial support for the work comes from Wichita State University, Fairmount College of Liberal Arts and Sciences and K-INBRE funds under NIH National Institute of General Medical Sciences, P20 GM103418. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of General Medical Sciences or the National Institutes of Health. Computing resources were funded by the National Science Foundation under Grant No. EIA-0216178 and Grant No. EPS-0236913, with matching support from the State of Kansas and the Wichita State University High Performance Computing Center.
Author information
Authors and Affiliations
Contributions
Research was planned by K.R.M., J.G.B., and C.K. C.K performed calculations. Data analysis was done by C.K. and K.R.M. Data interpretation and manuscript writing were done by C.K., K.R.M., and J.G.B., J.M.E. acquired the 13C and 19F NMR spectra.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kasireddy, C., Ellis, J., Bann, J. et al. The Biophysical Probes 2-fluorohistidine and 4-fluorohistidine: Spectroscopic Signatures and Molecular Properties. Sci Rep 7, 42651 (2017). https://doi.org/10.1038/srep42651
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42651
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.