Abstract
Xandarellida is a well-defined clade of Lower Palaeozoic non-biomineralized artiopodans that is exclusively known from the early Cambrian (Stage 3) Chengjiang biota of South China. Here we describe a new member of this group, Xandarella mauretanica sp. nov., from the middle Cambrian (Stage 5) Tatelt Formation of Morocco, making this the first non-trilobite Cambrian euarthropod known from North Africa. X. mauretanica sp. nov. represents the youngest occurrence of Xandarellida – extending its stratigraphic range by approximately 10 million years – and expands the palaeobiogeographic distribution of the group to the high southern palaeolatitudes of West Gondwana. The new species provides insights into the lightly sclerotized ventral anatomy of Xandarellida, and offers stratigraphically older evidence for a palaeobiogeographic connection between Burgess Shale-type euarthropod communities in North Africa and South China, relative to the (Tremadocian) Fezouata biota.
Similar content being viewed by others
Introduction
The Xandarellida Chen et al.1 (sensu2,3) are an enigmatic group of non-biomineralized artiopodan euarthropods whose distinctive features include the possession of stalked ventral eyes, a posterior extension of the cephalon covering the anterior trunk tergites, and the occurrence of dorsoventral segmental mismatch on the trunk1,2,3,4,5,6,7. Xandarellida consists of three taxa that are exclusively known from the Chengjiang Konservat-Lagerstätte (Cambrian Stage 3) in South China, namely Xandarella spectaculum4, Cindarella eucalla1, and Luohuilinella rarus6 (Fig. 1).
Topology follows Ortega-Hernández et al.48.
Within the diverse Palaeozoic clade Artiopoda Hou and Bergström2, xandarellids have been regarded as members of a more inclusive group known as the Petalopleura Hou and Bergström2 (Table 1), which also includes the lower Cambrian forms Sinoburius lunaris4 from Chengjiang, and (potentially) Phytophilaspis pergamena8 from the (Stage 4) Sinsk Formation in Siberia9,10. Unlike other monophyletic groups in Artiopoda, a clade that includes trilobites as its most familiar members (Fig. 1), the spatial distribution and temporal occurrence of xandarellids suggests a high degree of endemicity. Indeed, the group is conspicuously absent from Laurentia (North America) despite the intense study of numerous Cambrian Lagerstätten in this region11. Here, we describe an artiopodan interpreted as a xandarellid with appendicular preservation from the Cambrian (lowermost Stage 5) of the western High Atlas in Morocco. The new taxon represents the youngest stratigraphic occurrence of Petalopleura, the first palaeobiogeographic record of Xandarellida outside South China, and clarifies the organization of the lightly sclerotized ventral morphology in this poorly known group of non-biomineralized euarthropods.
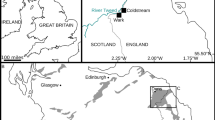
Geographic and Geological Setting
The Tatelt Formation (also referred to as the ‘Asrir’ Formation12) is exposed in the High Atlas Mountains of Morocco in the Lemdad Syncline (Fig. 2) and further south in the Anti-Atlas range. This unit is part of the Early Palaeozoic cover sequence deposited onto basement rocks of the Pan-African Orogen on the margin of West Gondwana13,14,15. The Tatelt Formation thickens southwards, from ca. 13–18 m in the Lemdad Syncline to ca. 55 m in exposures in the Anti-Atlas Mountains, with a concomitant transition from proximal to more distal facies15,16. The more proximal, High Atlas, succession is dominated by fine- to coarse-grained sandstones with intercalated grey-green tuff and ash beds, but also includes shale and conglomeratic layers14,15,16. The upper part of the Tatelt Formation in the Lemdad Syncline includes bidirectional trough cross-stratified layers and is interpreted as being deposited in a near-shore subtidal environment, with occasional intervals of deeper, or more quiescent, deposition14,15.
(A) Map of the locality within Morocco. (B) Geological sketch map of the Lemdad Syncline area. Redrawn, adapted and simplified from Geyer and Landing15. (C) Generalised sedimentary log through the lower – middle Cambrian (Tata – Feijas internes Group) transition in the eastern Lemdad Syncline; compiled from data in Geyer and Landing14,15. Maps (A,B) drafted by T.W.H. using Adobe Illustrator CC 2015.3 (http://www.adobe.com/uk/products/illustrator.html).
There is a well-established trilobite biostratigraphy for the Cambrian of southern Morocco17,18 within which the Tatelt Formation spans the Sectigena, Hupeolenus and Morocconus notabilis Biozones (Fig. 3). Unfortunately, this biostratigraphy is not well constrained by radiometric ages or chemostratigraphy, and a high degree of trilobite endemism has hindered correlation beyond the Iberian Peninsula and, to some extent, Avalonia19,20. A single radiometric age from the upper Lemdad Formation, Antatlasia guttapluviae Zone (Fig. 3), of 515.56 ± 1.16 Ma21 (recalculated from 517.0 ± 1.5 Ma22) provides a lower age boundary in this section. However, the Tatelt Formation is generally considered to straddle the lower – middle Cambrian (Series 2–3) boundary in Morocco, with the upper part deposited in Stage 515,16,20.
Strata in the Lemdad Syncline correlated to the international geological timescale using the Moroccan regional biostratigraphic framework and shown alongside the relative ages of other Cambrian Lagerstätte with Burgess Shale-type faunas. Compiled from Geyer and Landing15, Geyer and Vincent18, Geyer and Malinky19, and Van Roy et al.54. [a]Radiometric age for the upper Antatlasia guttapluviae Zone in the Lemdad Syncline recalculated21 after 517 ± 1.5 Ma22; note the discrepancy between this age and the international age (ca. 510 Ma) of Cambrian Series 2 Stage 4 to which the A. guttapluviae Zone has been biostratigraphically correlated15,17,19. Correlation of early and middle Cambrian strata in Morocco with the international timescale would greatly benefit from additional radiometric constraints.
The specimen was recovered from a medium-bedded well-indurated fine sand- to siltstone unit with simple trace fossils near the top of the Tatelt Formation in the Lemdad Syncline (Fig. 2B), in the Morocconus notabilis Zone (Fig. 3). This has been correlated to near the base of Cambrian Stage 5, possibly contemporaneous with Iberian Bilbilian/Leonian boundary and the Lapworthella Limestone (Ad) of the British Comley Series18,19,20.
Results
Systematic Palaeontology
Artiopoda Hou and Bergström2 (sensu23).
Remarks
The new taxon can be assigned to Artiopoda based on the preserved ventral morphology23, notably the antenniform first appendage pair attached at either side of a strongly sclerotized ventral hypostome, followed by numerous pairs of homonomous walking legs that gradually decrease in size along the body, and the possession of hourglass-shaped sternites. In particular, hourglass-shaped sternites rule out comparisons with non-artiopodan Cambrian euarthropods – which lack sternites altogether – such as fuxianhuiids24,25, bivalved stem-group euarthropods26,27, megacheirans28,29, and marrellomorphs30,31. Although the presence of a first pair of antenniform limbs is symplesiomorphic within Deuteropoda Ortega-Hernández32 (i.e. upper stem-group Euarthropoda + crown-group Euarthropoda)33,34, the combination of this character with post-oral limbs that gradually decrease in size and become differentiated into a caudal region (e.g. pygidium) are exclusive to Artiopoda.
Petalopleura Hou and Bergström2.
Xandarellida Chen et al.1.
Remarks
Hou and Bergström2 and Ramsköld et al.3 independently proposed definitions of Xandarellida that differ somewhat in their emphasis on particular morphological characters. We follow the diagnosis provided by Ramsköld et al.3 as this is based on a more comprehensive consideration of the organization of the cephalic appendages, hypostome morphology, and presence of segmental mismatch between the trunk limbs and tergites.
The similar style of preservation observed on the limbs and the hypostome strongly suggest that the new taxon lacked a biomineralized exoskeleton, and thus rules out potential affinities with Trilobita. The fossil is recognized as a member of Xandarellida based on similarities in limb morphology and possession of a natant hypostome associated with frontal organs (see detailed discussion below).
Xandarella Hou et al.4.
Constituent taxa
Xandarella spectaculum Hou et al.4 Cambrian (Stage 3) Chiungchussu Formation, Chengjiang, South China (type species); Xandarella mauretanica sp. nov., Cambrian (Stage 5) Tatelt Formation, High Atlas, Morocco.
Emended diagnosis
Semicircular head shield with small sessile eyes placed laterally. Suture or unfused overlap between shield portions extending from eye to lateral margin. Natant hypostome with elongate suboval outline. Head with antennae and up to six appendage pairs under large head shield; endopod of first post-antennal appendage reduced. Each tergite on anterior half of trunk covers a single pair of biramous appendages. Each succeeding tergite on posterior half of trunk covers an increasing number of appendage pairs, ranging from two to twelve (or more). Endopods slender, with up to a dozen podomeres. Pygidium with median posterior spine.
Remarks
The diagnosis of Xandarella has been revised from Hou et al.4 and Hou and Bergström2 to reflect the variability in the ventral morphology in this taxon, in light of the new specimen from Morocco (Fig. 4). The post-antennal endopods of most artiopodans possess up to seven podomeres (e.g. Cheloniellon35; Triarthrus36; Cindarella3; Phacops sp.37; Kuamaia and Saperion5; Misszhouia and Naraoia38; Emeraldella23; Sidneyia39; Arthroaspis40) as also expressed in several extant representatives, and generally resolved as the ancestral state for crown-group Euarthropoda41. By contrast, X. spectaculum and the new species described here are typified by the presence of post-antennal endopods with approximately 12 podomeres, leading us to propose this character as a diagnostic feature of Xandarella. A high podomere count is not exclusive to Xandarella among Palaeozoic euarthropods, however, as a similar condition is also known in some early Cambrian forms, such as megacheirans (e.g. Fortiforceps2), fuxianhuiids (e.g. Fuxianhuia24; Chengjiangocaris25), and bivalved stem-group euarthropods (e.g. Jugatacaris42). The substantial phylogenetic distance between these euarthropods and Xandarella (see topology in Legg et al.43), however, suggests that endopods with more than seven podomeres are a symplesiomorphy of Deuteropoda29,33,34,41, and its occurrence within Xandarellida is most likely a result of homoplasy. Thus, the taxonomic utility of this character for Xandarella is only applicable within the context of Artiopoda.
Xandarella mauretanica sp. nov. Figs 4 and 5.
Etymology
From the Latin mauretanicus, in reference to Mauretania, a historical region that corresponds to part of North Africa, including the Mediterranean coast of Morocco.
Diagnosis
Xandarellid with robust antennae, and a prominent hypostome with paired frontal organs located medially. At least 22 pairs of post-antennal limbs are present along the body. Endopod of first pair of post-antennal limbs half the length relative to that of the succeeding appendages.
Material, locality, horizon
A single specimen MHNM-HA-TT-CA-1A preserved as a ventral impression (Fig. 4), collected from the middle Cambrian (Stage 5) upper Tatelt Formation (Morocconus notabilis Zone) of Morocco.
Description
The holotype is an articulated, and almost complete, individual with a total length of 21 mm (sagittal; including the antennae), and maximum width of 8 mm (transverse) (Fig. 4). The specimen represents an exceptionally preserved impression of the lightly sclerotized underside of the body, including appendages and ventral exoskeletal elements. Details of the dorsal exoskeleton, including cephalon and trunk tergite morphology, are entirely absent. The cephalic region incorporates a prominent hypostome with an elongate suboval outline (ca. 4 mm length, sag.; 1.8 mm maximum width, trans.), expressed as a deep concave impression, and typified by a medial transverse constriction that conveys an approximately lemniscate appearance. Two convex rounded structures (ca. 0.9 mm diameter) are associated with the medial constriction; these features are tentatively interpreted as a pair of frontal organs, simple ocelli-like structures found on the anterior region of various artiopodans5,44. The frontal organs superficially bisect the hypostome. The anterior half has a slightly acute anterior margin and a smooth texture. The posterior half has a rounded posterior margin, and evinces ornamentation consisting of a single transverse and broad, crescentic ridge located medially, followed posteriorly by six longitudinal ridges arranged in a parallel series. The suboval outline of the hypostome indicates a natant attachment to the underside of the head, as concomitant hypostomes invariably possess a blunt anterior edge that matches the cephalic margin, or extends from the cephalic doublure (examples discussed below) (Fig. 6B,C).
(A) Campanamuta mantonae, Cambrian (Stage 3) Sirius Passet, North Greenland50. (B) Triarthrus eatoni, Upper Ordovician, Beecher’s Trilobite Bed, USA36. (C) Emeraldella brocki, ROM-61148, Cambrian (Stage 5) Burgess Shale, British Columbia23 (photograph courtesy of Jean-Bernard Caron, Royal Ontario Museum). Abbreviations: ant, antennae; hyp, hypostome.
A pair of multiannulated antennae attach at either side of the hypostome, immediately posterior to the position of the frontal organs. The antennae are both longer (5 mm, sag.) and wider (0.75 mm, trans.) than any of the other preserved appendages, and demonstrate a distinctive sigmoidal flexure towards the anterior end of the body. Fine morphological details are mostly indistinct, but faint segmental boundaries (ca. 0.25 mm length, sag.) suggest that the antennae are composed of at least 20 podomeres or annuli; the length of the annuli is consistently shorter than their width along the preserved extent of the antenna. The holotype preserves a total of 22 pairs of slender concave impressions of variable length, which correspond to the endopods of the post-antennal appendages. The 1st pair of post-antennal appendages originates in close proximity to the posterior margin of the hypostome, and curves anteriorly until reaching the same level as the posterior border of the frontal organs. These delicate appendages are both shorter (ca. 1.3 mm, sag.) and thinner (0.25 mm, trans.) than any other pair on the anterior two thirds of the body. The 2nd and 3rd leg pairs share approximately the same dimensions (ca. 3.5 mm length, sag.; 0.6 mm width, trans.) and are similarly curved anteriad. Faint segmental impressions (ca. 0.25 mm length, sag.) on the distal end of the 2nd leg indicate podomeres that are of subequal length and width, or slightly longer than wide; similar impressions more proximally also indicate podomeres that are slightly longer than wide, suggesting the presence of up to a dozen podomeres per limb, at least on the cephalic region. The 4th leg pair is slightly shorter than the preceding one (ca. 3 mm length, sag.), and is laterally splayed rather than flexed anteriorly. Unlike the 2nd and 3rd legs, the 4th pair evinces a distal curvature towards the posterior end, which becomes more accentuated in the subsequent appendages. The subtle decrease in size and shift in appendage orientation suggest that the 4th leg pair could correspond to the last set of cephalic appendages; if correct, this interpretation would imply that the head includes the antennae and four pairs of (arguably) biramous appendages. This interpretation may be supported by the fact that the 2nd to 4th legs display a regular separation of 1 mm (sag.), whereas the spacing between the succeeding pairs progressively decreases towards the posterior end of the specimen. The 5th to 15th legs have the same overall construction, consisting of a laterally oriented concave impression with a small posterior curvature at the distal end, and only differ slightly in their dimensions. Although the 5th leg has an approximate length of 2.4 mm (sag.) and width of 0.6 mm (trans.), the same measurements are 1.6 mm (sag.) and 0.4 mm (trans.) for the 15th leg, reflecting a gentle decrease in overall size towards the posterior end. This organization gives the appearance of a narrowed trunk relative to the anterior cephalic region, although this is mostly applicable to the proximal parts of the limbs, and thus there is no reason to assume that the dorsal exoskeleton would necessarily follow this morphology. The 16th to 22nd legs show a more drastic decrease in size resulting in a triangular tapering of the body; although the 16th leg is only slightly shorter than the preceding limb (ca. 1.3 mm, sag.), the 22nd leg is only expressed as a suboval impression of approximately 0.15 mm in length (sag.). This sharp change in appendage length may reflect the fusion of posterior segments into a discrete pygidium. The caudal termination of the trunk is not observed.
The only exoskeletal elements preserved on the ventral side – other than the hypostome – correspond to the sternite (i.e. ventral sclerotized plate) series, which occupies a longitudinal axial position between the limb pairs. Unlike the appendages, the sternite series is differentially preserved as a convex impression that reflects the pattern of segmentation through an alternating series of light and dark bands of sediment. The light bars are aligned with the appendage impressions and generally possess a narrower profile (trans.) relative to the dark bands; this disposition indicates that the bands represent the sternites and tendinous bars (i.e. intersegmental arthrodial membrane) respectively. The length of the individual sternites mirrors the spacing of the appendage pairs along the body, and thus the longest (sag.) are located between the 3rd and 5th legs, and become progressively shorter towards the rear termination of the body. Despite the gradual reduction in appendage size, the width of the sternite series remains relatively invariant throughout the length of the body – as informed by the separation between the leg impressions – with a maximum and minimum width (trans.) of 1 mm (at the 4th leg) and 0.8 mm (at the 15th leg) respectively.
Remarks
X. mauretanica sp. nov. differs from X. spectaculum in that the 1st leg pair in the former is distinctively reduced (Figs 4 and 5), whereas in the latter species these appendages follow a more gentle gradation in size with the succeeding cephalic legs2,45. X. spectaculum also differs in featuring up to 36 pairs of post-antennal leg pairs2, compared to the 22 leg pairs preserved in the new taxon. Whether this difference may be attributed to the preservation of X. mauretanica sp. nov., or if it reflects actual interspecific variability, remains uncertain. It is also possible that the different number of post-antennal legs can be attributed to ontogeny, as the holotype X. mauretanica sp. nov. is significantly smaller (length 21 mm, sag.) than type material of X. spectaculum (e.g. holotype, length 51 mm length, sag.2). Clarification on these issues will require the input of additional material of X. mauretanica sp. nov., or studies on the ontogeny of X. spectaculum.
Discussion
Phylogenetic affinities
Although only details of the ventral anatomy are preserved in the available material, the similarities between X. mauretanica sp. nov. and X. spectaculum support their close phylogenetic affinities, and offers new insights on the morphological variability within Xandarellida. The new taxon confirms the presence of hourglass-shaped sternites connected by intersegmental tendinous bars – previously suggested2 or inferred3 for members of Xandarellida – similarly to the ventral exoskeletal anatomy of other artiopodans36,38,46,47. The appendicular organization in X. mauretanica sp. nov. and X. spectacullum share various symplesiomorphies of Artiopoda, most notably the presence of antennae at either side of a sclerotized hypostome, the homonomous construction of the post-oral appendages, and the progressive reduction in size of the legs towards the posterior end of the body. However, the presence of post-antennal endopods with approximately 12 podomeres is unique to Xandarella within the evolutionary context of Artiopoda48.
These comparisons are further strengthened by the presence of a natant hypostome in X. mauretanica sp. nov. and X. spectaculum. Several Cambrian artiopodans possess a concomitant hypostome that is widely attached to the anterior margin of the cephalon, and may be expressed as an extension of the cephalic doublure with or without a suture (e.g. Triarthrus36; Emeraldella23; Squamacula49; Aglaspis48) (Fig. 6B,C) or occur in association with an anterior sclerite (e.g. Conciliterga2,44; see also char. 12 in Edgecombe and Ramsköld5). By contrast, the natant hypostome of Xandarellida is situated further back, in a position closer to the sagittal midline of the head2,3. Although the natant hypostome is also known in various non-trilobite artiopodans (e.g. Cheloniellon35; Nektaspida10,38; Campanamuta50; Arthroaspis40) (Fig. 6A), none of these taxa combine this character with the presence of endopods with more than seven podomeres as observed in Xandarella.
The ventral anatomy of X. mauretanica sp. nov. is broadly comparable to that of Cindarella eucalla in terms of overall appendage organization. However, these taxa differ in that the 1st leg pair of the latter is not reduced3, and the hypostome of the former is more elongate and bears the paired frontal organs (Figs 4 and 5); the endopods of C. eucalla also differ in evidently having archetypal endopods with seven podomeres3. Comparisons with the recently described xandarellid Luohuilinella rarus are problematic because this taxon is only known from the dorsal exoskeleton6. However, the posterior end of the body in X. mauretanica sp. nov. and L. rarus exhibits a sharp decrease in width that produces a distinctive subtriangular caudal tapering, which is otherwise not observed in either X. spectaculum or C. eucalla2,3, or other non-trilobite artiopodan groups (e.g. Cheloniellida35; Conciliterga51; Nektaspida38; Xenopoda23).
Outside the Xandarellida, the morphology of X. mauretanica sp. nov. shares similarities with the petalopleuran Sinoburius lunaris4. With the exception of the 1st leg pair in X. mauretanica sp. nov., the cephalic appendages of both taxa are noticeably longer than those in the trunk region; however, the possibility that this appearance in the new taxon may be a taphonomic artefact cannot be entirely discarded at present given the preferential preservation of the proximal portion of the appendages (Fig. 4). S. lunaris may further resemble X. mauretanica sp. nov., in the possession of four legs in the head region, yet again, assuming that the extrapolation of the cephalic shield based on the anatomy of the anterior appendages is correct. The preservation of S. lunaris only reveals the outline of the endopods, and thus the number of constituent podomeres is uncertain. Less cryptically, the paired frontal organs in the hypostome of X. mauretanica sp. nov. draw a parallel to similar structures in S. lunaris2,5, although the natant hypostome in the latter taxon is comparatively smaller and has a subtriangular outline. The frontal organs of S. lunaris appear to be located in a slightly anterior position relative to the hypostome, however, and thus it is uncertain if they reflect an identical organization to that observed in X. mauretanica sp. nov. The pygidial segmentation of S. lunaris also displays a sharp decrease in limb size, comparable to that of X. mauretanica sp. nov. (Fig. 4) and L. rarus6.
Taphonomic implications
The preservation of X. mauretanica sp. nov. is noteworthy in comparison to other xandarellid specimens, and indeed to other soft-bodied Cambrian euarthropods. Previously described xandarellid fossils from the Chengjiang biota are expressed as pyritized carbonaceous compressions in shale, as is typical for non-trilobite euarthropods52. The holotype of X. mauretanica sp. nov. is three-dimensionally preserved in a fine sand- to siltstone (Fig. 4) and was probably pyritized during early diagenesis, which would account for the exceptional preservation of the lightly sclerotized ventral morphology; however, the appearance of the fossil strongly suggests a more recent exposure to oxidation and intense weathering. This peculiar style of three-dimensional preservation contrasts with that of Burgess Shale-type Cambrian deposits in Laurentia, consisting of flattened carbonaceous films53, and also with the non-biomineralized euarthropods from the Early Ordovician Fezouata biota of Morocco54. Thus, the taphonomy of the fossils at the Tatelt Formation requires further investigation, but demonstrates that soft-bodied Burgess Shale-type euarthropods can be found in atypical sedimentological settings.
Stratigraphic and palaeobiogeographic significance
The discovery of X. mauretanica sp. nov. from the middle Cambrian Tatelt Formation in the High Atlas Mountains of Morocco provides the youngest known record of xandarellids, extending their stratigraphic range by approximately 10 million years. More significantly, this finding substantially expands the palaeobiogeographic and palaeolatitudinal range of Xandarellida out of tropical South China (Chengjiang biota) and onto polar Gondwana at high southern latitudes during the Cambrian (Fig. 7). Given that xandarellids are not known from the early Cambrian (Stage 4) Emu Bay Shale in South Australia11,55, X. mauretanica sp. nov. represents the only direct connection between communities of non-biomineralized artiopodans in continental Gondwana (North Africa) and South China (Chengjiang biota) during the Cambrian (Fig. 7). The close palaeobiogeographical links between Morocco and South China only become evident later on during the latest Tremadocian (Lower Ordovician) thanks to the euarthropod diversity preserved in the Fezouata biota, which includes representatives of several typically Cambrian groups such as marrellomorphs, leanchoiliids, mollisoniids, nektaspids and aglaspidids54,56,57 (Table 2). This raises the possibility that Burgess Shale-type euarthropod communities in Gondwana are not necessarily restricted to the Emu Bay Shale in South Australia, but that they may also extend to the early Cambrian of Morocco.
Xandarella mauretanica sp. nov. represents the only member of Xandarellida reported outside South China, and expands the distribution of this clade to the South Hermisphere. References for Cambrian localities: Laurentia23,39,51,58,59,60,61; North Greenland40,62; Morocco (this study); South China2,63,64,65; South Australia9,10,66; Tasmania67. References for Ordovician localities: Baltica68; Wales69,70; Morocco54,56,71; South Africa72. Palaeocontinental reconstructions redrawn, adapted and simplified by J.O.-H. from Torsvik and Cocks73 (Figs 2.8, 2.11) using Adobe Illustrator CC 2015.3 (http://www.adobe.com/uk/products/illustrator.html).
The discovery of X. mauretanica sp. nov. draws attention to the absence of xandarellids from several Cambrian Burgess Shale-type faunas in North America (Fig. 7) (Table 2). Given the intense efforts invested in the systematic description of non-biomineralized Cambrian euarthropods from Laurentia over the last 50 years, the absence of xandarellids from this palaeocontinent may reflect a real palaeobiogeographic signal, rather than an artefact of taphonomic or collection bias. Future work on the Tatelt Formation offers great potential for the discovery of additional exceptionally preserved fossils in the middle Cambrian of Morocco that will help further refine the palaeobiogeographic and stratigraphic distribution of Burgess Shale-type faunas.
Materials and Methods
A single available specimen collected from the upper Tatelt Formation (Morocconus notabilis Zone) of Morocco. Specimen MHNM-HA-TT-CA-1A corresponds to the external mould of the ventral side of the body in dorsoventral view, preserved in a medium-bedded well-indurated mudstone and sandstone unit. Photographs were taken with a Nikon 3100 DSLR. The material is housed at the MHNM (Natural Museum History of Marrakesh).
Additional Information
How to cite this article: Ortega-Hernández, J. et al. A xandarellid artiopodan from Morocco – a middle Cambrian link between soft-bodied euarthropod communities in North Africa and South China. Sci. Rep. 7, 42616; doi: 10.1038/srep42616 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
09 May 2017
A correction has been published and is appended to both the HTML and PDF versions of this paper. The error has not been fixed in the paper.
09 May 2017
Scientific Reports 7: Article number: 42616; published online: 17 February 2017; updated: 09 May 2017 This Article contains errors in Figure 7. The correct Figure 7 appears below as Figure 1 with a corrected Figure legend and additional references:
References
Chen, J. Y., Zhou, G. Q., Zhu, M. & Yeh, K. Y. The Chengjiang biota. A unique window of the Cambrian Explosion. Taichung, Taiwan. Natl. Mus. Nat. Sci. (1996).
Hou, X. & Bergström, J. Arthropods of the Lower Cambrian Chengjiang fauna, southwest China. Foss. Strata 45, 1–116 (1997).
Ramsköld, L., Chen, J., Edgecombe, G. D. & Zhou, G. Cindarella and the arachnate clade Xandarellida (Arthropoda, Early Cambrian) from China. Earth Environ. Sci. Trans. R. Soc. Edinb. 88, 19–38 (1997).
Hou, X., Ramsköld, L. & Bergström, J. Composition and preservation of the Chengjiang fauna – a Lower Cambrian soft-bodied biota. Zool. Scr. 20, 395–411 (1991).
Edgecombe, G. D. & Ramsköld, L. Relationships of Cambrian Arachnata and the systematic position of Trilobita. J. Paleontol. 73, 263–287 (1999).
Zhang, X., Fu, D. & Dai, T. A new xandarellid arthropod from the Chengjiang Lagerstätte, Lower Cambrian of Southwest China. Geobios 45, 335–338 (2012).
Ortega-Hernández, J. & Brena, C. Ancestral Patterning of tergite formation in a centipede suggests derived mode of trunk segmentation in trilobites. PLoS One 7, e52623 (2012).
Ivantsov, A. Y. Trilobite-like arthropod from the Lower Cambrian of the Siberian Platform. Acta Palaeontol. Pol. 44, 455–466 (1999).
Paterson, J. R., García-Bellido, D. C. & Edgecombe, G. D. New artiopodan arthropods from the Early Cambrian Emu Bay Shale Konservat-Lagerstätte of South Australia. J. Paleontol. 86, 340–357 (2012).
Paterson, J. R., Edgecombe, G. D., García-Bellido, D. C., Jago, J. B. & Gehling, J. G. Nektaspid arthropods from the Lower Cambrian Emu Bay Shale Lagerstätte, South Australia, with a reassessment of lamellipedian relationships. Palaeontology 53, 377–402 (2010).
Hendricks, J. R., Lieberman, B. S. & Stigall, A. L. Using GIS to study palaeobiogeographic and macroevolutionary patterns in soft-bodied Cambrian arthropods. Palaeogeogr. Palaeoclimatol. Palaeoecol. 264, 163–175 (2008).
Álvaro, J. J., Benziane, F., Thomas, R., Walsh, G. J. & Abdelaziz, Y. Neoproterozoic–Cambrian stratigraphic framework of the Anti-Atlas and Ouzellagh promontory (High Atlas), Morocco. J. Afr. Earth Sci. 98, 19–33 (2014).
Destombes, J., Hollard, H. & Willefert, S. In Lower Palaeozoic of North-Western and West Central Africa (ed. Holland, C. H. ) 91–336 (John Wiley & Sons Ltd., 1985).
Geyer, G. & Landing, E. In Morocco’ 95—The Lower–Middle Cambrian standard of western Gondwana (eds Geyer, G. & Landing, E. ) 7–46 (1995).
Geyer, G. & Landing, E. In Morocco 2006. Ediacaran–Cambrian Depositional Environments and Stratigraphy of the Western Atlas Rgions. Explanatory Description and Field Excursion Guide (eds Geyer, G. & Landing, E. ) 9–75 (2006).
Landing, E., Geyer, G. & Heldmaier, W. Distinguishing eustatic and epeirogenic controls on Lower–Middle Cambrian boundary successions in West Gondwana (Morocco and Iberia). Sedimentology 53, 899–918 (2006).
Geyer, G. Revised Lower to lower Middle Cambrian biostratigraphy of Morocco. Newsl. Stratigr. 22, 53–70 (1990).
Geyer, G. & Vincent, T. The Paradoxides puzzle resolved: the appearance of the oldest paradoxidines and its bearing on the Cambrian Series 3 lower boundary. Paläontol. Z. 89, 335–398 (2015).
Geyer, G. & Malinky, J. M. Middle Cambrian fossils from Tizi N’Tichka, the High Atlas, Morocco. Part 1. Introduction and trilobites. J. Paleontol. 71, 620–637 (1997).
Álvaro, J. J., Elicki, O., Geyer, G., Rushton, A. W. A. & Shergold, J. H. Palaeogeographical controls on the Cambrian trilobite immigration and evolutionary patterns reported in the western Gondwana margin. Palaeogeogr. Palaeoclimatol. Palaeoecol. 195, 5–35 (2003).
Maloof, A. C. et al. Constraints on early Cambrian carbon cycling from the duration of the Nemakit-Daldynian–Tommotian boundary δ13C shift, Morocco. Geology 38, 623–626 (2010).
Landing, E. et al. Duration of the Early Cambrian: U–Pb ages of volcanic ashes from Avalon and Gondwana. Can. J. Earth Sci. 35, 329–338 (1998).
Stein, M. & Selden, P. A. A restudy of the Burgess Shale (Cambrian) arthropod Emeraldella brocki and reassessment of its affinities. J. Syst. Palaeontol. 10, 361–383 (2012).
Bergström, J., Hou, X., Zhang, X. & Clausen, S. A new view of the Cambrian arthropod Fuxianhuia. GFF 130, 189–201 (2008).
Yang, J., Ortega-Hernández, J., Butterfield, N. J. & Zhang, X. Specialized appendages in fuxianhuiids and the head organization of early euarthropods. Nature 494, 468–471 (2013).
García-Bellido, D. C. et al. The bivalved arthropods Isoxys and Tuzoia with soft‐part preservation from the Lower Cambrian Emu Bay Shale Lagerstätte (Kangaroo Island, Australia). Palaeontology 52, 1221–1241 (2009).
Legg, D. A. & Caron, J.-B. New middle Cambrian bivalved arthropods from the Burgess Shale (British Columbia, Canada). Palaeontology 57, 691–711 (2014).
Haug, J. T., Briggs, D. E. G. & Haug, C. Morphology and function in the Cambrian Burgess Shale megacheiran arthropod Leanchoilia superlata and the application of a descriptive matrix. BMC Evol. Biol. 12, 162 (2012).
Aria, C., Caron, J.-B. & Gaines, R. R. A large new leanchoiliid from the Burgess Shale and the influence of inapplicable characters on stem arthropod phylogeny. Palaeontology 58, 629–660 (2015).
García-Bellido, D. C. & Collins, D. H. A new study of Marrella splendens (Arthropoda, Marrellomorpha) from the Middle Cambrian Burgess Shale, British Columbia, Canada. Can. J. Earth Sci. 43, 721–742 (2006).
Kühl, G. & Rust, J. Re-investigation of Mimetaster hexagonalis: a marrellomorph arthropod from the Lower Devonian Hunsrück Slate (Germany). Paläontol. Z. 84, 397–411 (2010).
Ortega-Hernández, J. Making sense of ‘lower’ and ‘upper’ stem-group Euarthropoda, with comments on the strict use of the name Arthropoda von Siebold, 1848. Biol. Rev. Camb. Philos. Soc. 91, 255–273 (2016).
Waloszek, D., Maas, A., Chen, J. & Stein, M. Evolution of cephalic feeding structures and the phylogeny of Arthropoda. Palaeogeogr. Palaeoclimatol. Palaeoecol. 254, 273–287 (2007).
Waloszek, D., Chen, J., Maas, A. & Wang, X. Early Cambrian arthropods—new insights into arthropod head and structural evolution. Arthropod Struct. Dev. 34, 189–205 (2005).
Stürmer, W. & Bergström, J. The arthropod Cheloniellon from the Devonian Hunsrück Shale. Paläontol. Z. 52, 57–81 (1978).
Cisne, J. L. Triarthrus eatoni (Trilobita): anatomy of its exoskeletal, skeletomuscular, and digestive systems. Palaeontogr. Am. 9, 99–141 (1981).
Bruton, D. L. & Haas, D. W. The anatomy and functional morphology of Phacops (Trilobita) from the Hunsrück Slate (Devonian). Palaeontogr. Abt. A 253, 29–75 (1999).
Zhang, X.-L., Shu, D.-G. & Erwin, D. H. Cambrian naraoiids (arthropoda): morphology, ontogeny, systematics, and evolutionary relationships. J. Paleontol. 81, 1–52 (2007).
Stein, M. Cephalic and appendage morphology of the Cambrian arthropod Sidneyia inexpectans Walcott, 1911. Zool. Anz. - J. Comp. Zool. 253, 164–178 (2013).
Stein, M., Budd, G. E., Peel, J. S. & Harper, D. A. Arthroaspisn. gen., a common element of the Sirius Passet Lagerstätte (Cambrian, North Greenland), sheds light on trilobite ancestry. BMC Evol. Biol. 13, 99 (2013).
Boxshall, G. A. The evolution of arthropod limbs. Biol. Rev. Camb. Philos. Soc. 79, 253–300 (2004).
Fu, D. & Zhang, X. A new arthropod Jugatacaris agilis n. gen. n. sp. from the Early Cambrian Chengjiang Biota, South China. J. Paleontol. 85, 567–586 (2011).
Legg, D. A., Sutton, M. D. & Edgecombe, G. D. Arthropod fossil data increase congruence of morphological and moleucular phylogenies. Nat. Commun. 4, 2485 (2013).
Ortega-Hernández, J. Homology of head sclerites in Burgess Shale euarthropods. Curr. Biol. 25, 1625–1631 (2015).
Liu, Y., Scholtz, G. & Hou, X. When a 520 million year old Chengjiang fossil meets a modern micro CT – a case study. Sci. Rep. 5, (2015).
Whittington, H. B. Anatomy of the Ordovician trilobite Placoparia . Philos. Trans. R. Soc. Lond. B Biol. Sci. 339, 109–118 (1993).
Ramsköld, L., Jun-Yuan, C., Edgecombe, G. D. & Gui-Qing, Z. Preservational folds simulating tergite junctions in tegopeltid and naraoiid arthropods. Lethaia 29, 15–20 (1996).
Ortega-Hernández, J., Legg, D. A. & Braddy, S. J. The phylogeny of aglaspidid arthropods and the internal relationships within Artiopoda. Cladistics 29, 15–45 (2013).
Zhang, X., Han, J., Zhang, Z., Liu, H. & Shu, D. Redescription of the Chengjiang arthropod Squamacula clypeata Hou and Bergström, from the Lower Cambrian, south-west China. Palaeontology 47, 605–617 (2004).
Budd, G. E. Campanamuta mantonae gen. et sp. nov. and exceptionally preserved arthropod from the Sirius Passet Fauna (Buen Formation, lower Cambrian, North Greenland). J. Syst. Palaeontol. 9, 217–260 (2011).
Whittington, H. B. Tegopelte gigas, a second soft-bodied trilobite from the Burgess Shale, Middle Cambrian, British Columbia. J. Paleontol. 59, 1251–1274 (1985).
Gabbott, S. E., Xian-guang, H., Norry, M. J. & Siveter, D. J. Preservation of Early Cambrian animals of the Chengjiang biota. Geology 32, 901–904 (2004).
Butterfield, N. J., Balthasar, U. & Wilson, L. A. Fossil diagenesis in the Burgess Shale. Palaeontology 50, 537–543 (2007).
Van Roy, P., Briggs, D. E. G. & Gaines, R. R. The Fezouata fossils of Morocco; an extraordinary record of marine life in the Early Ordovician. J. Geol. Soc. 172, 541–549 (2015).
Paterson, J. R., Edgecombe, G. D. & Jago, J. B. The ‘great appendage’ arthropod Tanglangia: biogeographic connections between early Cambrian biotas of Australia and South China. Gondwana Res. 27, 1667–1672 (2015).
Van Roy, P. et al. Ordovician faunas of Burgess Shale type. Nature 465, 215–218 (2010).
Martin, E. L. O. et al. The Lower Ordovician Fezouata Konservat-Lagerstätte from Morocco: Age, environment and evolutionary perspectives. Gondwana Res. 34, 274–283 (2016).
Whittington, H. B. The Middle Cambrian trilobite Naraoia, Burgess Shale, British Columbia. Philos. Trans. R. Soc. B Biol. Sci. 280, 409–443 (1977).
Conway Morris, S. & Robinson, R. A. More soft-bodied animals and algae from the Middle Cambrian of Utah and British Columbia. Univ. Kans. Palaeontol. Contrib. 122, 1–48 (1988).
Briggs, D. E. G., Lieberman, B. S., Hendricks, J. R., Halgedahl, S. L. & Jarrad, R. D. Middle Cambrian arthropods from Utah. J. Paleontol. 82, 238–254 (2008).
Lerosey-Aubril, R., Ortega-Hernández, J., Kier, C. & Bonino, E. Occurrence of the Ordovician-type aglaspidid Tremaglaspis in the Cambrian Weeks Formation (Utah, USA). Geol. Mag. 150, 945–951 (2013).
Budd, G. E. A nektaspid arthropod from the Early Cambrian Sirius Passet fauna, with a description of retrodeformation based on functional morphology. Palaeontology 42, 99–122 (1999).
Zhang, X., Han, J. & Shu, D. New occurrence of the Burgess Shale arthropod Sidneyia in the Early Cambrian Chengjiang Lagerstätte (South China), and revision of the arthropod Urokodia . Alcheringa Australas. J. Palaeontol. 26, 1–8 (2002).
Lerosey-Aubril, R., Ortega-Hernández, J. & Zhu, X. The first aglaspidid sensu stricto from the Cambrian of China (Sandu Formation, Guangxi). Geol. Mag. 150, 565–571 (2013).
Zhang, X., Zhao, Y., Yang, R. & Shu, D. The Burgess Shale arthropod Mollisonia (M. sinica New Species): new occurrence from the Middle Cambrian Kaili Fauna of Southwest China. J. Paleontol. 76, 1106–1108 (2002).
Edgecombe, G. D., Paterson, J. R. & García-Bellido, D. C. A new aglaspidid-like euarthropod from the lower Cambrian Emu Bay Shale of South Australia. Geol. Mag. (2016).
Ortega-Hernández, J., Braddy, S. J., Jago, J. B. & Baillie, P. W. A new aglaspidid arthropod from the Upper Cambrian of Tasmania. Palaeontology 53, 1065–1076 (2010).
Rak, Š., Bergström, J., Fatka, O. & Budil, P. The Upper Ordovician arthropod Zonozoe drabowiensis Barrande (Libeň and Letná formations, Sandbian, Barrandian area, Czech Republic). Bull. Geosci. 185–188, doi: 10.3140/bull.geosci.1026 (2009).
Fortey, R. A. & Rushton, A. W. A. A new aglaspidid arthropod from the Lower Ordovician of Wales. Palaeontology 46, 1031–1038 (2003).
Legg, D. A. & Hearing, T. W. A late surviving xenopod (Arthropoda) from the Ordovician Period, Wales. Geol. Mag. 152, 942–948 (2015).
Ortega-Hernández, J., Van Roy, P. & Lerosey-Aubril, R. A new aglaspidid euarthropod with a six-segmented trunk from the Lower Ordovician Fezouata Konservat-Lagerstätte, Morocco. Geol. Mag. 153, 524–536 (2016).
Fortey, R. A. & Theron, J. N. A new Ordovician arthropod, Soomaspis, and the agnostid problem. Palaeontology 37, 841–861 (1995).
Torsvik, T. H. & Cocks, L. R. M. In Early Palaeozoic Biogeography and Palaeogeography (eds Harper, D. A. T. & Servais, T. ) 38, 5–24 (Geological Society, London, 2013).
Hesselbo, S. P. Aglaspidida (Arthropoda) from the upper Cambrian of Wisconsin. J. Paleontol. 66, 885–923 (1992).
Yang, J., Ortega-Hernández, J., Lan, T., Hou, J. & Zhang, X. A predatory bivalved euarthropod from the Cambrian (Stage 3) Xiaoshiba Lagerstätte, South China. Sci. Rep. 6, 27709 (2016).
Briggs, D. E. G. Bivalved arthropods from the Cambrian Burgess Shale of British Columbia. Palaeontology 20, 595–621 (1977).
Legg, D. A. An acercostracan marrellomorph (Euarthropoda) from the Lower Ordovician of Morocco. Sci. Nat. 103, 21 (2016).
Liu, Q. The first discovery of Marrella (Arthropoda, Marrellomorpha) from the Balang Formation (Cambrian Series 2) in Hunan China. J. Paleontol. 87, 391–394 (2013).
Chen, J., Waloszek, D. & Maas, A. A new ‘great-appendage’ arthropod from the Lower Cambrian of China and homology of chelicerate chelicerae and raptorial antero-ventral appendages. Lethaia 37, 3–20 (2004).
Liu, Y., Hou, X. & Bergström, J. Chengjiang arthropod Leanchoilia illecebrosa (Hou, 1987) reconsidered. GFF 129, 263–272 (2007).
Edgecombe, G. D., García-Bellido, D. C. & Paterson, J. R. A new leanchoiliid megacheiran arthropod from the lower Cambrian Emu Bay Shale, South Australia. Acta Palaeontol. Pol. 56, 385–400 (2011).
Walcott, C. D. Cambrian Geology and Paleontology II. Middle Cambrian Branchiopoda, Malacostraca, Trilobita and Merostomata. Smithson. Misc. Collect. 57, 145–229 (1912).
Acknowledgements
JOH is supported by a Herchel-Smith Postdoctoral Fellowship held at the Department of Zoology, University of Cambridge (UK). TWH is supported by NERC (CENTA) Studentship NE/L0022493/1, and a BGS University Funding Initiative CASE partnership (BUFI S266) which provided field work funding. Philip Wilby and Matthew Saker-Clark are thanked for their assistance in the field.
Author information
Authors and Affiliations
Contributions
T.W.H., T.H.P.H., A.A. and K.E.H. arranged the field work and field permissions; T.W.H. found and excavated the specimen, and A.A. assisted with field work; J.O.-H., T.W.H. and G.D.E. wrote the manuscript with contributions from A.A., T.H.P.H., A.H. and K.E.H.; A.A. and K.E.H. arranged specimen curation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ortega-Hernández, J., Azizi, A., Hearing, T. et al. A xandarellid artiopodan from Morocco – a middle Cambrian link between soft-bodied euarthropod communities in North Africa and South China. Sci Rep 7, 42616 (2017). https://doi.org/10.1038/srep42616
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42616
This article is cited by
-
The Souss lagerstätte of the Anti-Atlas, Morocco: discovery of the first Cambrian fossil lagerstätte from Africa
Scientific Reports (2021)
-
The appendicular morphology of Sinoburius lunaris and the evolution of the artiopodan clade Xandarellida (Euarthropoda, early Cambrian) from South China
BMC Evolutionary Biology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.