Abstract
Early detection and surgery represent the mainstay of treatment for superficial melanoma, but for high risk lesions (Breslow’s thickness >0.75 mm) an effective adjuvant therapy is lacking. Vitamin D insufficiency plays a relevant role in cancer biology. The biological effects of 1α hydroxycholecalciferol on experimental melanoma models were investigated. 105 melanoma patients were checked for 25-hydroxycholecalciferol (circulating vitamin D) serum levels. Human derived melanoma cell lines and in vivo xenografts were used for studying 1α-hydroxycholecalciferol-mediated biological effects on cell proliferation and tumor growth. 99 out of 105 (94%) melanoma patients had insufficient 25-hydroxycholecalciferol serum levels. Interestingly among the six with vitamin D in the normal range, five had a diagnosis of in situ/microinvasive melanoma. Treatment with 1α-hydroxycholecalciferol induced antiproliferative effects on melanoma cells in vitro and in vivo, modulating the expression of cell cycle key regulatory molecules. Cell cycle arrest in G1 or G2 phase was invariably observed in vitamin D treated melanoma cells. The antiproliferative activity induced by 1α-hydroxycholecalciferol in experimental melanoma models, together with the discovery of insufficient 25-hydroxycholecalciferol serum levels in melanoma patients, provide the rationale for using vitamin D in melanoma adjuvant therapy, alone or in association with other therapeutic options.
Similar content being viewed by others
Introduction
Cutaneous melanoma represents 5–7% of all skin malignancies, but it is responsible for about 75% of deaths from skin tumors. The incidence of melanoma is increasing at an alarming rate, with a lifetime risk of developing a melanoma of 1/58 for USA males and 1/25 for Australian males1. Although early detection and surgery represent the mainstay of treatment for localized lesions, no effective therapy for metastatic melanoma is available so far. Furthermore patients with high risk melanomas (Breslow thickness >0.75 mm) are still orphans of an effective adjuvant therapy2,3,4,5,6. Vitamin D, also worded D-hormone, has pleiotropic effects of relevance to cancer, which includes regulation of cell growth and differentiation, induction of apoptosis and regulation of tumor/immune-system interactions7,8,9,10,11.
Interestingly a strong association between low serum levels of 25-hydroxycholecalciferol (25-OH-vitD3) and increased cancer incidence and cancer–related mortality has been demonstrated in large population studies12,13. Several clinical studies and meta-analysis of case-control and cohort studies also support this picture14,15,16. Furthermore, preclinical studies indicate that active metabolites of vitamin D or their synthetic derivatives have potential anticancer activity and could be used to potentiate the anticancer effects of several cytotoxic and antiproliferative drugs17,18,19,20. Vitamin D-mediated-biological effects require the expression of vitamin D receptor (VDR) on the target cells and the integrity of downstream effectors, among which 1α-hydroxylase (1α-OHase) and 25-hydroxylase (25-OHase) are necessary for the synthesis of vitamin D active metabolites, whereas 24-hydroxylase (24-OHase) regulates vitamin D inactivation and catabolism (Supplementary Figure S1). Alterations of VDR receptor and/or downstream enzymatic repertoire may potentially impair vitamin D responsivity of the target cells21,22. The aim of this experimental study was to analyze the biological effects induced by 1α-hydroxycholecalciferol (1α-OH-vitD3) in malignant melanoma models in vitro and in vivo. A panel of primary melanoma cell lines derived from patients with metastatic disease were found to constitutively express functional VDR, 25-OHase and 1α-OHase, indicating a potential vitamin D sensitivity. Treatment of melanoma cells with 1α-OH-vitD3 strongly impaired cell proliferation in vitro and tumor growth in vivo. Moreover low serum levels of 25-OH-vitD3 (10–30 ng/mL) were discovered to occur in the large majority melanoma patients (all stages) at time of first diagnosis23.
All together these findings open a clinical relevant question on the possibility to use vitamin D or its active metabolites in melanoma adjuvant therapy, in particular for high risk lesions, for which an observational approach is currentlty adopted.
Results
Insufficient 25-OH-vitamin-D3 Serum Levels is a Common Feature of Melanoma Patients at time of first diagnosis
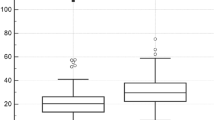
Evaluation of 25-OH-vitD3 serum levels was performed on 105 melanoma patients (unselected) before surgery. Surprisingly, 99 out of 105 patients (94%) showed deficient or insufficient 25-OH-vitD3 serum levels, with values ranging from 7 to 30 ng/mL. Normal serum levels of 25-OH-vitD3 (30–76 ng/mL) were detected in 6 patients only (6% of the cases), 5 of which bearing in situ or micro-invasive melanoma and one a nodular melanoma with lymph node metastasis (Supplementary Table S1 and Fig. 1a,b). Data regarding basal serum levels of 25-OH-vitD3 in 101 matched (for sex and age) blood donors without evidence of neoplastic or chronic diseases were obtained from the Sant’Andrea Hospital data bank and used as comparative control. In this cohort, 55 subjects out of 101 (54%) had 25-OH-vitD3 serum levels in the normal range. The observed difference was statistically significant (p < 0.005).
(a) Comparative evaluation of 25-OH-vitD serum levels registered in 105 melanoma patients at time of surgery ( ) and in 101 matched (for sex and age) healthy individuals (∆). (b) The area between 30–76 ng/mL, which represents 25-OH-vitD sufficiency, is marked by a gray line. Standard deviation is shown (Statistical analysis using t-test: ***p value < 0.005).
) and in 101 matched (for sex and age) healthy individuals (∆). (b) The area between 30–76 ng/mL, which represents 25-OH-vitD sufficiency, is marked by a gray line. Standard deviation is shown (Statistical analysis using t-test: ***p value < 0.005).
The expression of vitamin D receptor (VDR) could be a critical event for vitamin D sensitivity. As demonstrated in RT-PCR the panel of melanoma cell lines considered in this study invariably expressed VDR and the complete repertoire of transcripts that is required for vitamin D activation and catabolism, namely 25-hydroxylase (gene CYP27A1), 1α-hydroxylase (gene CYP27B1) and 24-hydroxylase (gene CYP24A1) (see supplementary Figure S2A). The expression of VDR was also confirmed in western blot and immunocytochemistry for those melanoma cells used for establishing in vitro and in vivo experimental models to work with (Supplementary Figure S2B and data not shown).
The presence of VDR polymorphism was also investigated in a panel of melanoma cell lines used for experimental procedures (Supplementary Table S2). As expected, the identified polymorphisms did not impair VDR function. In fact all melanoma cell lines used in this study showed an antiproliferative response to 1α-OH-vitD3 in vitro (see below).
1α-Hydroxycholecalciferol and Vitamin D2 Active Derivative Paricalcitol Inhibit Melanoma Cell Proliferation in Vitro
Six well-characterized melanoma cell lines were used for studying the biological effects induced by 1α-OH-vitD3 on melanoma growth in vitro. Cells were cultured in the presence or absence of 1α-OH-vitD3 20 ng/mL (5 × 10−8 M). Two different pharmaceutical preparations of this active 1α-OH-vitD3 metabolite (Diseon ® and Dediol ®) were used for these experiments. 1α-OH-vitD3 consistently impaired melanoma cell proliferation in vitro and such a biological effect was measurable after 72 hrs of treatment (Fig. 2a).
Melanoma cells proliferation as determined by colorimetric tetrazolium salt assay (MTS assay) in the presence of (a) 20 ng/mL (5 × 108 M) 1α-hydroxycholecalciferol (two different pharmaceutical preparations) and (b) vitamin-D2 active derivative paricalcitol at concentration ranging from 80 ng/mL to 800 ng/mL. Six metastatic melanoma cell lines were used in this experiment. The assay was performed after 72 hours of vitamin-D treatment. Untreated cells (CTR) and cells incubated with ethanol (EtOH 0.1%) were used as controls. (Experiments in triplicate. S.D. is reported on the top of each column. Statistical analysis using ANOVA and Bonferroni post-hoc test: ns = not significant; *p < 0.05; **p < 0.01; ***p < 0.001).
Similar experiments were performed with the synthetic active derivative of vitamin D2 named paricalcitol. In this case a concentration of 0.8 μg/mL was required to consistently inhibit melanoma cell proliferation in vitro (Fig. 2b). Tumorigenic melanoma cell lines VAG, IR6 and 1007 were then selected for establishing melanoma xenografts for a new set of experiments in vivo. Cells were first cultured in the presence or absence of 1α-OH-vitD3 20 ng/mL (5 × 10−8 M) for 9 days. Once again inhibition of melanoma cell proliferation was observed after few days of 1α-OH-vitD3 treatment (Fig. 3a). To better understand whether the vitamin D-dependent inhibition of melanoma cell growth was a reversible event, a longer vitamin D treatment (18 days) was considered and a proliferation assay was finally performed after 1α-OH-vitD3 withdrawal. As shown in Fig. 3b cell proliferation was recoverable up to 6 days of treatment, while for longer treatment (>9 days) melanoma cells failed to be recovered in a vitamin D –free medium. This biological effect was statistically significant (p < 0.001).
(a) Melanoma cell proliferation analyzed by cell counting after 3, 6 and 9 days of 1α-OH-D3 treatment, is strongly inhibited in the presence of vitamin-D3. This effect was not observed in melanoma control cells growing in standard vitD3 –free medium (solvent alone). (b) Proliferation assay was performed after 1α-OH-vitD3 withdrawal at 6 and 9 days. As showed in the right panel, cell proliferation was rescued up to 6 days of treatment. For longer vitD3 treatment (>9 days) all the melanoma cells failed to be recovered. (Experiments in triplicate. Mean ± S.D. is reported. Statistical analysis using ANOVA and Bonferroni post-hoc test: **p < 0.01; ***p < 0.001).: ** p < 0.01; ***p < 0.001.
Interestingly, melanoma cells exposed to vitamin-D failed to growth efficiently in vitro, but this effect was not related to cell death. Morphological changes consisting in cytosol enlargement with appearance of small dendritic processes were invariable observed in vitamin D treated melanoma cells at the end of the experiments, together with an increased expression of E-cadherin (Fig. 4a–c). The slight differences in the mobility of E-chaderin molecular species observed in western blot analysis are likely due to cell-specific post-translational modifications and/or to the occurence of alternative splicing (expression of different isoforms) as already reported in the literature24. These findings support the evidence that 1α-OH-vitD3 impairs melanoma cell proliferation and triggers differentiation7.
(a) Morphological aspects of melanoma cell differentiation observed after 18 days of vitamin-D3 treatment. Melanoma cells cultured in conventional vitamin-D3 free medium (CRT) and medium supplemented with solvent alone (0.1% ethanol) were used as comparative controls. Note single scattered IR6, VAG and 1007 melanoma cells with enlarged cytoplasm and small dendritic processes after 18 days of vitamin-D3 treatment (scale bar = 100 μm). (b) E-cadherin increased expression in vitamin-D3 treated cells, as evaluated in western blot analysis by using a specific mAb; The slight differences in MW of the observed bands are likely due to the expression of cell-specific isoforms of the protein. β-actin was used as loading control (figure derived from cropped gel/blot for semplification). (C) E-chaderin expression at immunohistochemical level on vitamin-D3 treated and untreated representative melanoma xenografts (post-autopsy) (indirect immunoperoxidase; scale bar = 200 μm).
To deeply investigate the anti-proliferative effects induced by 1α-OH-vitD3 on melanoma cells in vitro, DNA content and cell cycle distribution of 1α-OH-vitD3-treated and untreated cell lines were analysed. Cytofluorimetric analysis was then performed after treatment of the cell lines with 1α-OH-vitD3 20 ng/ml (5 × 10−8 M) at 3, 6 and 9 days.
1α-OH-vitD3 induced cell cycle arrest in all melanoma cell lines, acting at different levels. IR6 melanoma cells were blocked in G1-phase (60% vs 38% of the control cells) after 72 hrs of treatment. The effect was persistent at day 9 (79% vs 54%) indicating that melanoma cells were unable to recover in the presence of 1α-OH-vitD3 (Fig. 5a).
(a) Fluorescence-activated cell sorting (FACS) of IR6, VAG and 1007 cell lines treated for 3, 6, and 9 days with (1α-OH-vitD3) or without (CTR) 1α-OH-vitD3 20 ng/ml (5 × 10−8 M), Cell cycle perturbation and redistribution of IR6, VAG and 1007 melanoma cells in presence of vitamin-D3 is demonstrated. Row data regarding cell distribution (%) in G1, S, and G2 phases are shown in detail. (b) Western blot analysis to support FACS data, showing modulation of cell-cycle key regulatory molecules expected to be specifically involved in G1 and G2 blocks, performed on total cell lysates from IR6, VAG and 1007 melanoma cells after 72 h of vitamin-D3 treatment. In the bottom of panel B the lack of procaspase-3 cleavage is shown to demonstrate the absence of apoptosis in vitamin-D3 treated cells. (All the experiments were performed in triplicate; figure derived from a cropped gels/blots for semplification) (c) Densitometric analysis of cyclins (D1, A,B), p27 and p21 protein bands shown in panel B. Each band intensity was first normalized to the corresponding band of internal control (β-actin). Bands obtained from vitamin-D treated and untreated cells were compared each other, normalizing those derived from untreated cells to 1. The band intensity is represented in the graph as relative densitometric unit (rdu). Data are means ± SD of the relative band intensity from three independent WB experiments. Standard deviation is shown (Statistical analysis using t-test: ns = not significant; *p < 0.05; **p < 0.01).
VAG melanoma cells were blocked in G2 phase. Such a perturbation of the cell-cycle was observed after 72 hrs of treatment (32% vs 5%) but it was much more evident on day 9 (66% vs 6%) (Fig. 5a).
In 1007 melanoma cell line 1α-OH-vitD3 induced cell accumulation in the proliferative compartments (S plus G2 phase) at days 6 and 9, compared to the control cells. This was associated with a loss of cells in G1 phase (55% vs 68% at day 6 and 40% vs 72% at day 9) (Fig. 5a).
The sub-G1 peak (apoptosis peak) was not observed in these experiments. Lack of apoptosis was also demonstrated by the absence of PARP-1 and procaspase-3 cleveage in western blot analysis of cell lysates from 1α-OH-vitD3 treated melanoma cells (data not shown and Fig. 5b). Altogether these findings, suggest that 1α-OH-vitD3 Induces cell-cycle perturbation and arrest of melanoma cell proliferation in G1 or G2 phase, whereas apoptosis (or cell death for toxicity) was not observed in this experimental condition.
1α-OH-vitD3 Modulates the Expression of Cell-Cycle Regulatory Molecules
To further analyze the cell-cycle perturbation induced by 1α-OH-vitD3 in melanoma cells, the expression of candidate cell-cycle regulatory molecules was evaluated by western blot analysis (Fig. 5b,c). Cell-cycle arrest in G1 phase, induced by 1α-OH-vitD3 in IR6 melanoma cell line correlated with an increased expression of the cyclin-dependent kinase inhibitors p21 and p27, and down-regulation of cyclin-D1. On the other side, the arrest in G2 phase observed in VAG was associated to a slight decrease in cyclin-B1 expression level (Fig. 5b,c). In 1007 melanoma cell line 1α-OH-vitD3 induced an increased expression of cyclin-A1 with slight up-regulation of p21 and p27 according to the accumulation of melanoma cells in the proliferative compartment (S plus G2 phase) (Fig. 5b,c). These findings indicate that cell cycle perturbation induced by 1α-OH-vitD3 in melanoma cells involves the recruitment of regulatory molecules and pathways, which are specific for each melanoma cell line. However, the observed vitaminD-mediated effects on cell cycle, invariably converge toward an impairment of melanoma cell proliferation.
Interestingly the observed cell cycle inhibition seems to be independent by the BRAF status (see Supplementary Table S2 also).
Long-term Systemic Administration of 1α-OH-vitD3 Inhibits Melanoma Cell Growth in Vivo
In vivo experimental models of human melanoma xenografts were finally used for studying the long term effects induced by 1α-OH-vitD3 on melanoma growth. Daily administration of 1α-OH-vitD3 (see Material and Methods section for detail) markedly inhibited melanoma growth in vivo. The biological effect observed after 42 days from melanoma cells injection was statistically significant (Fig. 6a). The antiproliferative effect of 1α-OH-vitD3 on melanoma xenografts was finally confirmed at autopsy. The final histological evaluation of residual explanted tumours showed large areas of tumor necrosis with dystrophic calcifications in melanoma xenografts derived from vitamin D treated mice, but not in tumors explanted from control animals (Fig. 6b). A slight hypercalcemia with 5–10% body weight loss was observed in all of 1α-OH-vitD3 treated mice at the end of the experiment but not in the control animals (data not shown). These findings might suggest the occurrence of potentially harmful side effects of long-term administration of 1α-OH-vitD3. However, at the end of the experiment all the animals were in good health. No histological alterations in kidney, liver, lung or other organs were observed at the final histological examination (data not shown).
(a) Tumor mass variations at the end of vitamin-D3 treatment in vivo (day 42 from melanoma cells injection). Each point represents the average of values of four xenografted melanomas. Values at day 42 were directly determined at autopsy. (S.D. is reported on the top of each point. Statistical analysis at 42 days using ANOVA and Bonferroni post-hoc test: saline vs treated mice ***p < 0.001). (b) Representative panel of histological slides showing tumor necrosis and dystrophic calcifications in the vitamin-D3 treated residual melanoma xenografts collected at autopsy. Melanoma xenografts from animals injected with saline solution alone, were used as comparative controls. (Formalin-fixed and paraffin embedded histological preparations; haematoxylin-eosin staining; scale bar = 200 μm).
Interestingly, 1α-OH-vitD3-mediated modulation of expression of cell cycle regulatory molecules, previously observed in vitro, was also detected in vivo at immunohistochemical level (Supplementary Figure S3 and data not shown). These findings further support the notion that vitamin D active metabolites may trigger inhibitory signals in melanoma cells, which impair cell proliferation and tumor growth.
Discussion
Vitamin D has pleiotropic effects of relevance to cancer with potential therapeutical implications in oncology7,8,9,10. By using experimental models of human melanoma we demonstrate that 1α-OH-vitD3 consistently impairs melanoma cell proliferation and tumor growth in vitro and in vivo. The possibility to take advantage of the vitamin D-mediated biological effects for treating melanoma patients is further supported by epidemiological and clinical evidence: i) Large observational studies show a significant association between low serum levels of 25-OH-vitD3 and increased cancer incidence11,12,13,14,15,16. Meta-analysis of case-control and cohort studies demonstrate that individuals with 25-OH-vitD3 ≥ 33 ng/mL (82 nmol/L) had a 50% lower incidence of colorectal cancer14; ii) Interestingly and in line with our work hypothesis, patients with early stage non small cell lung carcinomas with normal 25-OH-vitD3 serum levels and high vitamin D intake, had improved overall and recurrence-free survival16 iii) Preclinical data also indicate that active metabolites of vitamin D play as potential anticancer agents in different tumor models10,17,18,19,20,25,26,27. We demonstrate here that malignant melanoma represents a potentially responsive target to vitamin D. Although mutational analysis of VDR gene, CYP27A1, CYP27B1 and CYP24 A1 genes may be required before to consider a vitamin D treatment, relevant mutational events, which impair melanoma cells sensitivity to D-hormone, seem to be sporadic. The scenario of vitamin D –mediated biological effects is very complex to be analyzed in detail. Vitamin D active metabolites regulate important biological processes via genomic and non-genomic effects. Non genomic effects are in part mediated by the increase of free cytosolic calcium levels, whereas genomic effects require the binding to VDR, the predominant nuclear receptor protein expressed on target cells and tissues. VDR binds vitamin-D ligand with high affinity, resulting in heterodimerization with retinoid X receptor (RXR) and in zinc-finger-mediated binding to vitamin D responsive elements (VDREs), which modulate transcription of the target genes26,27,28,29,30. As a general feature all the patients-derived melanoma cell lines we tested were sensitive to 1α-OH-vitD3, although the occurrence of vitamin D3 resistant melanoma cell lines has been reported31. Vitamin D3 mediated-arrest of cell proliferation and modulation of the expression repertoire of p21, p27, cyclin-D1 and likely of other cell-cycle key regulatory molecules, seem to be common biological events in melanoma experimental models used in this study. Interestingly this scenario has been observed also in different cell systems26,27,28,29,30. It is noteworthy that the promoter regions of specific cell cycle regulatory genes posses VDREs, which modulate gene transcription in response to vitamin-D/VDR binding10,32. This mechanism, which demonstrates a direct functional role of vitamin D in regulating the cell cycle, corroborates our findings.
By using in vitro experimental models we show that 1α-OH-vitD3 induces cell cycle arrest of IR6 melanoma cells in G1 phase, whereas VAG melanoma cells were blocked in G2 phase and 1007 melanoma cells accumulate in the proliferative compartment (S plus G2 phases). Such a cell-type specific perturbation of cell cycle was recoverable up to 6 days of vitD3 treatment, after that time melanoma cells proliferation was definitively impaired. Early VitD3 removal by the culture medium, infact, restored melanoma cell proliferation. This effect was quite evident in all melanoma cell lines considered in this study, with differences related to the cell-proliferation rates and/or cell-type specific biological features. A detailed analysis of modulation of cell cycle regulatory molecules in these experimental conditions represents an interesting field of research to be pursued in the near future.
Furthermore we finally demonstrate that a relatively long systemic treatment with 1α-OH-vitD3 also inhibits melanoma growth in vivo.
The differences in 1α-OH-vitD3 mediated-biological effects observed in melanoma cell lines may be explained, at least in part, by the presence of specific molecular alterations occurring in each specific tumor (i.e BRAF and KIT mutations, p53 status etc.). In each specific case, in fact, 1α-OH-vitD3 could activate different pathways of cell response with different functional consequences on cell-cycle33. Interestingly, the melanoma cell lines considered in this study included tumors bearing BRAFV600E mutation (IR6 and FOR) and BRAF wild-type (VAG, 1007, MUL, LOJ), but the observed 1α-OH-vitD3 mediated-biological effects on cell proliferation and tumor growth occurred independently by the BRAF status. The potential functional role of vitamin-D in triggering tumor cell differentiation and/or senescence also deserves consideration. It is well known that normal melanocytes have a dendridic shape and that a E-chaderin plays a pivotal role in melanocyte-keratinocyte interaction in normal conditions. After neoplastic transformation this phenotype is generally lost. In the present study we observed that 1α-OH-vitD3 induced morphological changes (dendritic processes) and E-chaderin expression in melanoma cells, suggesting a pro-differentiative activity. This interesting effect has been already observed in a different cell system7. Further experiments will be carried out to deeply investigate this important issue.
Finally, although the value of normal serum level of 25-OH-vitD3 has been recently revisited, the effective vitamin D intake for tumor prevention and therapy are not defined yet23,34,35. The discovery of insufficient 25-OH-vitD3 serum levels (10–30 ng/mL) in almost all melanoma patients analyzed in this study, opens the question on the effects that restored serum levels of D-hormone may potentially have on the overall and recurrence-free survival and eventually on melanoma prevention. Although a direct correlation between 25-OH-vitD3 serum levels and clinical-pathological prognostic parameters could not be found in our cohort of patients, because of the low number of cases in each specific subgroup, it is intriguing that 5 out of 6 melanoma patients with sufficient vitamin-D serum levels at time of surgery had in situ/microinvasive melanomas. Moreover, a direct correlation between 25-OH-vitD3 serum levels and melanoma thickness has been reported in some clinical studies that fully support our work hypothesis36,37,38,39.
In conclusion this preclinical study provides the biological and clinical rationale for using vitamin D in melanoma adjuvant therapy.
What remains to be defined is if restored 25-OH-vitD serum levels (30–76 ng/mL) will be sufficient to improve the overall and disease-free survival of melanoma patients with high risk of tumor progression.
The intriguing possibility to use D-hormone in association with the most recently proposed molecular targeted therapies for treating metastatic melanoma, also deserves consideration. Anti-BRAF and anti-KIT inhibitors, in fact, finally produce functional impairment of down-stream molecules that regulate cell proliferation (cell cycle) and survival, some of which are direct targets of vitamin D10,40,41,42.
Targeting the aforementioned oncogenic molecules has been reported to induce important objective responses in patients with advanced melanoma. However the occurrence of a relatively rapid drug resistence, as well as the paradoxical activation of downstream molecules provides the rationale for exploring a combined therapeutic approach43,44,45. In this context the demonstrated vitamin D-mediated antiproliferative effects may be oncologically relevant.
Concluding, the antiproliferative activity of 1α-OH-vitD3 in experimental melanoma models in vitro and in vivo, together with the discovery of 25-OH-vitD3 insufficiency in almost all melanoma patients at time of surgery, provide the biological and clinical rationale for using vitamin D in melanoma adjuvant therapy, alone or in association with other therapeutic options. Long-term randomized clinical trials will be necessary for demonstrating, definitively, the real impact of vitamin D on melanoma patients’ survival.
Methods
Melanoma Patients, Valuation of 25-OH-vitamin-D3 Serum Levels
105 melanoma patients were checked for 25-OH-vitD3 (the active measurable vitamin D metabolite) serum levels at time of diagnosis. Patients were 43 male and 62 female, age ranging from 28 to 87 years (median age 53.7), 49 of them had high risk cutaneous melanoma (Breslow’s thickness >0.75 mm).
Data regarding the basal serum levels of 25-OH-vitD3 in 101 matched (for sex and age) healthy individuals (blood donors) from the same community, were anonymously obtained from the blood bank at Saint’ Andrea Hospital and used as comparative control. The clinical features of melanoma patients object of this study are summarized in Supplementary Table S1. 25-OH-vitD serum levels were determined by using the fully automated chemiluminescence method Liaison 25OHD assay system (Dia-Sorin, Saluggia VC, Italy) and classified according to the recent literature23. Briefly 25-OH-vitD3 serum levels <10 ng/mL were considered ‘deficient’; >10 to 30 ng/mL ‘insufficient’; and >30 to 76 ng/mL (75 to 190 nmol/L) were considered ‘normal’.
Melanoma Cell lines, phenotipic and molecular characterization
Metastatic melanoma - derived cell lines (VAG, FOR, IR6, 1007, MAR, and LOJ) were established and phenotypically characterized at the Immunology Laboratory, National Cancer Institute Regina Elena of Rome, Italy. Most of these cell lines were previously described46,47,48,49,50,51. Cell lines were regularly checked immunocytochemically for the expression of specific melanoma markers (Mart-1, HMB-45, S-100) before each experimental procedure (data not shown). The expression of vitamin D receptor (VDR) was analyzed immunohistochemically as reported below and the occurence of VDR polymorphism was also investigated. Briefly, three major restriction fragment lenghts polymorphism (RFLP) were investigated, in the exon 2 and 3′-region, respectively the TaqI, BsmI and FokI polymorphisms52,53. The FokI polymorphism results in the production of a VDR protein that is three amino acids longer with an increase of transcription levels54. The BsmI is a RFLP in intron 8 at the 3′ end of the VDR gene, which may influence VDR messenger RNA stability55. TaqI polymorphism is a RFLP at codon 352 in exon 9 of the VDR gene, that leads to a silent codon change from ATT to ATC, which both result in an isoleucine at codon 35256.
Melanoma cell lines (VAG, FOR, IR6, 1007, MAR, and LOJ) were also tested for the presence of BRAF (exon 15 V600E) mutations. Genomic DNA was extracted using standard protocol (Roche) and subjected to amplification of the target gene using the following primers: BRAF-F: 5′-CCTAAACTCTTCATAATGCTT-3′ and BRAF-R: 5′-ATAGCCTCAATTCTTACCAT-3′. Nucleotide sequence analyses were performed using BigDye Terminator Cycle Sequencing Ready reaction Kit (Applied Biosystems) and an ABI PRISM 310 Genetic Analyzer (Applied Biosystems). The phenotypical and molecular features of melanoma cell lines used in this study are summarized in Supplementary Table S2.
RT-PCR, Western Blot and Immunohistochemical Analysis
Total RNA extraction and cDNA synthesis were previously described55. PCR reactions were performed in PCR buffer (10 mM Tris-HCl pH 8.4; 500 mM KCl; 50 mM MgSO4) with 0.2 μmol primers and 2U Platinum Taq DNA Polymerase (Invitrogen) in a final volume of 50 μl for 35 cycles (denaturation at 95 °C for 30 seconds, annealing at 48 °C–52 °C, according to different pair of primers, for 30 seconds and extension at 72 °C for 1 minute). The following human oligonucleotides were used as primers: Vitamin D Receptor (VDR) for 5′-CCAGTTCGTGTGAATGATGG-3′, rev 5′-GTCGTCCATGGTGAAGGA-3′; CYP27A1 (25-OHase) for: 5′-GGCAAGTACCCAGTACGG-3′, rev: 5′-AGCAAATAGCTTCCAAGG-3; CYP27B1 (1α-OHase) for: 5′-TGTTTGCATTTGCTCAGA-3′, rev: 5′-CCGGGAGAGCTCATACAG-3′; CYP24A1 (24-OHase) for: 5′-GCAGCCTAGTGCAGATTT-3′, rev: 5′-ATTCACCCAGAACTGTTG-3′. Integrity and equal loading of cDNA in the PCR reactions were checked by quantification of 18S mRNA levels.
Mouse monoclonal antibodies (mAbs) to p21, p27, Cyclin-D1, E-cadherin, and human VDR NR1/1 (Space Import & Export, Milan, Italy); goat polyclonal antiserum to Cyclin-B1, rabbit polyclonal antiserum to Cyclin-A1 (Space Import & Export), mouse mAbs to β-actin (Santa Crutz Biotechnology, CA, USA) and rabbit polyclonal procaspase-3 antibody sc-7148 (Santa Cruz Biotechnology) were purchased and used in immunochemical assays according to the manufacturer’s instruction. Immunohistochemical analysis on formalin fixed and paraffin embedded histological samples, as well as western blot analysis, were previously described57,58,59.
Melanoma Cell Cultures and Vitamin D Treatment in Vitro
For in vitro experiments 1α-hydroxycho-lecalciferol (1α-OH-vitD3) (Dediol® Sanofi-Aventis SpA, Milan, Italy, or Diseon® Teva Italia Srl, Milan, Italy) was added to the culture medium at final concentration of 20 ng/mL (5 × 10−8 M). Drug concentration was selected after preliminary dose-response experiments in vitro (data not shown). Briefly, 3 × 105 cells were seeded in six-well plate (Falcon Labware, Oxnard, CA USA) in complete RPMI-1640 culture medium, supplemented with 2% FBS. After 24 hours 1α-OH-vitD3 or solvent alone (0.1% ethanol) were added to the culture medium. The medium was replaced every 48 hours (in both treated and control cell lines) to guarantee a constant presence of fresh vitamin D. Cells were finally collected at 3, 6, 9 and 18 days of treatment (depending by the experiment) and used as targets for proliferation assays and FACS analysis as reported below. Similar experiments were performed by using the synthetic active derivative of vitamin-D2 worded paricalcitol (Zemplar ® Abbott Srl, Latina, Italy) at concentration ranging from 0.08 μg/mL to 0.8 μg/mL17. Tumorigenic melanoma cell lines (1007, IR6, VAG) were finally selected for creating melanoma xenografts in vivo as reported below.
Determination of Melanoma Cells Growth Rate In Vitro
Melanoma cells were seeded at concentration of 3 × 105 cells/well in a six-well plate and maintained in complete RPMI 2% FBS with or without 1α-OH-vitD3 as reported above. Cell proliferation was determined by cell counting in a Burker camera after 3, 6, 9 and 18 days of vitamin D treatment, depending by the experiment.
To achieve melanoma cell cultures for 18 days, melanoma cells growing at confluence were split at appropriate time (according to the doubling time of each cell line: TdIR6 = 36 hrs, TdVAG = 48 hrs and Td1007 = 72 hrs) by using a PBS solution containing trypsin 0.05% and EDTA 0.02% (Gibco). All the experiments were performed in triplicate. In a different set of experiments the MTS assay (colorimetric tetrazolium salt assay) was also used for evaluating cell growth rate in vitro. Cells were cultured at density of 5 × 104 cells/well in flat-botton 96-well plates. Vitamin-D treatment in vitro was performed with 1α-OH-vitD3 at final concentration of 20 ng/mL (5 × 10−8 M) or paricalcitol at concentration ranging from 0.08 μg/mL to 0.8 μg/mL. After 3 days of treatment CellTiter 96® Aqueous One Solution Reagent (Promega, Madison, WI, USA) was added to each well according to the manufacturer’s instructions. Cell viability was determined by measuring the absorbance at 490 nm using a 550 BioRad plate-reader (Bio-Rad, Hertfordshire, UK).
Cell cycle profile analysis by flow cytometry (FACS analysis)
Melanoma cells adjusted to reach 60–70% confluence at the time of FACS analysis were collected after 3, 6, and 9 days of 1α-OH-vitD3 treatment, washed in phosphate buffered saline solution (PBS) pH 7.4 and fixed in 50% acetone/methanol (1:4 v/v) in PBS for 1 hour. Cells were re-suspended in a DNA staining solution containing propidium iodide (10 mg/ml−1) and RNAse (1.8 units μl−1) for 30 minutes and finally analyzed on a FACScan (Becton Dickinson, Immunocytomety System, Mountain View, CA, USA). Twenty thousand events per sample were registered. All the experiments were performed in triplicate.
Melanoma Xenografts and 1α-OH-vitD3 Treatment in Vivo
36 pathogen-free Balb-c nude (nu/nu) mice, 4–5-weeks-old (Charles River Breeding Laboratories, USA) and tumorigenic human melanoma cells lines 1007, IR6 e VAG were used for establishing melanoma xenografts in vivo47,48. 36 mice were organized in 9 groups in cages of 4 animals each (12 mice for each melanoma cell line) and fed with standard diet and water ad libitum. One week after subcutaneous injection of melanoma cells (5 × 106 cells/mouse) mice were treated every other day, for a total of six weeks, with 1α-hydroxycholecalciferol via gavage, according to the following protocol: Groups 1 control mice: saline solution only (200 μl/mouse); Groups 2: 1α-OH-vitD3 4 I.U. (0.1 μg/200 μl volume/mouse); Groups 3: 1α-OH-vitD3 12 I.U. (0.3 μg/200 μl volume/mouse). Mice in groups 2 received a total of 80 I.U. of 1α-OH-vitD3 in 42 days of treatment, whereas mice in groups 3 got 240 I.U. Tumor size was measured every 3 days and tumor weight (gm) was calculated as previously described47. Animal health conditions were monitored daily all along the experiment.
Calcium serum level was measured in each animal at the beginning and at the end of the experiment. Mice were sacrificed after 6 weeks of treatment. Explanted organs and residual tumors were processed for conventional histology and immunohistochemistry.
Statistical Analysis
In vitro experiments have been repeated 3 times and the results obtained are presented as means ± standard deviation (SD). Significant changes were assessed by using Student’s t-test for unpaired data, two-way Anova and Bonferroni post-hoc tests for time and concentration effects (GraphPad Prism Software, version 5.0) a p values < 0.05 were considered significant.
Ethics statement
Melanoma cell lines used in this study were established at the Immunology Laboratory, National Cancer Institute Regina Elena of Rome, according to the Institutional ethical guidelines provided by the Italian Ministry of Public Health (IMPH). In vivo animal experiments were performed at the Animal Facility of the National Cancer Institute Regina Elena of Rome, under the direct control of an independent veterinary staff. Animal experiments were performed according to the guidelines provided by the IMPH. The study was approved by the Institutional Animal Care and Use Committee and by the multidisciplinary Board of Physicians of the Saint’ Andrea Melanoma Working Group (SAMWG).
Blood samples and tissues from humans were collected at Saint Andrea University Hospital and used anonymously in full agreement with the guidelines provided by the Institutional Review Board (Prot. CE no. 8391/2013) and Helsinki Declaration. Dermatologists from SAMWG, which have the responsibility for patients’ follow-up, collected a written informed consent from all the melanoma patients considered in this study.
Additional Information
How to cite this article: Spath, L. et al. Antiproliferative Effects of 1α-OH-vitD3 in Malignant Melanoma: Potential Therapeutic implications. Sci. Rep. 7, 40370; doi: 10.1038/srep40370 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Siegel, R., Naishadham, D. & Jemal, A. Cancer statistics, 2012. CA. Cancer J. Clin. 62, 10–29 (2012).
Tsao, H., Atkins, M. B. & Sober, A. J. Management of cutaneous melanoma. N Engl J Med 351, 998–1012 (2004).
Cummins, D. L. et al. Cutaneous malignant melanoma. Mayo Clin. Proc. 81, 500–507 (2006).
Korn, E. L. et al. Meta-analysis of phase II cooperative group trials in metastatic stage IV melanoma to determine progression-free and overall survival benchmarks for future phase II trials. J. Clin. Oncol. 26, 527–534 (2008).
Kudchadkar, R. & Sondak, V. Adjuvant interferon: recommit or move on? Lancet Oncol. 12, 112–113 (2011).
Hansson, J. et al. Two different durations of adjuvant therapy with intermediate-dose interferon alfa-2b in patients with high-risk melanoma (Nordic IFN trial): a randomized phase 3 trial. Lancet Oncol. 12, 144–152 (2011).
Palmer, H. G. et al. Vitamin D promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and inhibition of beta-catenin signaling. J. Cell. Biol. 154, 369–387 (2001).
Jiang, F., Li, P., Nicosia, S. V. & Bai, W. Induction of ovarian cancer cell apoptosis by 1-, 25-dihydroxyvitamin D3 through the down-regulation of telomerase. J. Biol. Chem. 279, 53213–53221 (2004).
Schwartz, G. G. & Skinner, H. G. Vitamin D status and cancer: new insight. Curr. Opin. Clin. Nutr. Metab. Care. 10, 6–11 (2007).
Deeb, K. K., Trump, D. L. & Johnson, C. S. Vitamin D signalling pathways in cancer: potential for anticancer therapeutics. Nature Rev. Cancer 7, 684–697 (2007).
Feldman, D., Krishnan, A. V., Swami, S., Giovannucci, E. & Feldman, B. J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 14, 342–357 (2014).
Garland, C. F. et al. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet 2, 1176–1178 (1989).
Bertone-Johnson, E. R. et al. Plasma 25-hydroxyvitamin D and 1, 25-dihydroxyvitamin D and risk of breast cancer. Cancer Epidemiol. Biomarkers Prev. 14, 1991–1997 (2007).
Gorham, E. D. et al. Vitamin D and prevention of colorectal cancer. J Steroid Biochem. Mol. Biol. 97, 179–194 (2005).
Giovannucci, E. et al. Prospective study of predictors of vitamin D status and cancer incidence and mortality in men. J. Natl. Cancer Inst. 98, 451–459 (2006).
Zhou, W. et al. Circulating 25-hydroxyvitamin D levels predict survival in early stage non-small-cell-lung cancer patients. J. Clin. Oncol. 25, 479–485 (2007).
Kumagai, T., O’Kelly, J., Said, J. W. & Koeffler, H. P. Vitamin D2 analog 19-nor-1, 25 dihydroxyvitamin D2: antitumor activity against leukaemia, myeloma and colon cancer cells. J. Natl. Cancer Inst. 95, 896–905 (2003).
Yu, W. D. et al. Enhancement of 1α, 25-dihydroxyvitamin D3-mediated antitumor activity with dexamethasone. J. Natl. Cancer Inst. 90, 134–141 (1998).
Peehl, D. M., Seto, E., Hsu, J. Y. & Feldman, D. Preclinical activity of ketoconazole in combination with calcitriol or the vitamin D analogue EB1089 in prostate cancer cells. J. Urol. 168, 1583–1588 (2002).
Beer, T. M. et al. Double–blinded randomized study of high-dose calcitriol plus docetaxel compared with placebo plus docetaxel in androgen-independent prostate cancer: a report from the ASCENT investigators. J. Clin. Oncol. 25, 669–674 (2007).
Ingles, S. A. et al. Association of prostate cancer risk with genetic polymorphisms in vitamin D receptor and androgen receptor. J. Natl. Cancer Inst. 89, 166–170 (1997).
Albertson, D. G. et al. Quantitative mapping of amplicon structure by array CGH identifies CYP24 as a candidate oncogene. Nature Genet. 25, 144–146 (2000).
Rosen, C. J. Vitamin D Insufficiency. N. Engl. J. Med. 364, 248–254 (2011).
Slusser, A. et al. Cadherin expression, vectorial active transport, and metallothionein isoform 3 mediated EMT/MET responses in cultured primary and immortalized human proximal tubule cells. PLoS One 10(3), e0120132 (2015).
Eisman, J. A., Barkla, D. H. & Tutton, P. J. M. Suppression of in vivo growth of human cancer solid tumor xenografts by 1, 25-dihydroxyvitamin D3 . Cancer. Res. 47, 21–25 (1987).
Zhuang, S. H. & Burnstain, K. L. Antiproliferative effect of 1alpha, 25-dihydroxyvitamin D3 in human prostate cancer cell line LNCaP involves reduction of cyclin-dependent kinase 2 activity and persistent G1 accumulation. Endocrinology 139, 1197–1207 (1998).
Artaza, J., Sirad, F., Ferrini, M. G. & Norris, K. C. 1, 25(OH)2 vitamin D inhibits cell proliferation by promoting cell cycle arrest without inducing apoptosis and modifies cell morphology of mesenchymal multipotent cells. J. Steroid Biochem. Mol. Biol. 119, 73–83 (2010).
Kawa, S. et al. Vitamin D analogues up-regulate p21 and p27 during growth inhibition of pancreatic cancer cell lines. Br. J. Cancer 76, 884–889 (1997).
Dai, X. et al. Keratinocyte G2/M growth arrest by 1, 25-dihydroxyvitamin D3 is caused by Cdc2 phosphorylation through Wee1 and Myt1 regulation. J. Invest. Dermatol. 122, 1356–1364 (2004).
Li, P. et al. p27 Kip1 stabilization and G1 arrest by 1, 25 dihydroxyvitamin D3 in ovarian cancer cells mediated through down-regulation of cyclin E/cyclin-dependent kinase 2 and Skp1-cullin-F-box protein/Skp2 Ubiquitin ligase. J. Biol. Chem. 279, 25260–25267 (2004).
Reichrath, J. et al. In vivo comparison of the vitamin D endocrine system in 1, 25(OH)2D3-responsive and –resistant melanoma cells. Cancer Biol. Ther. 6, 48–55 (2007).
Murayama, A., Kim, M. S., Yanagisawa, J., Takeyama, K. & Kato, S. Transrepression by a liganded nuclear receptor via a bHLH activator through co-regulator switching. EMBO J. 23, 1598–1608 (2004).
Stambolsky, P. et al. Modulation of the vitamin D3 response by cancer associated Mutant p53. Cancer Cell 17, 273–285 (2010).
Garland, C. F., French, C. B., Baggerly, L. L. & Heaney, R. P. Vitamin D supplement doses and serum 25-hydroxyvitamin D in the range associated with cancer prevention. Anticancer Res. 31, 607–611 (2011).
Holick, M. F. et al. Guidelines for preventing and treating vitamin D deficiency and insufficiency revisited. J Clin Endocrinol Metab 97, 1153–1158 (2012).
Newton-Bishop, J. A. et al. Serum 25-hydroxyvitamin D3 levels are associated with Breslow thickness at presentation and survival from melanoma. J. Clin. Oncol. 27, 5439–5444 (2009).
Nürnberg, B. et al. Reduced serum 25-hydroxyvitamin D levels in stage IV melanoma patients. Anticancer Res. 29, 3669–3674 (2009).
Hutchinson, P. E., Osborne, J. E. & Pringle, J. H. Higher serum 25-hydroxy vitamin D3 levels at presentation are associated with improved survival from melanoma, but there is no evidence that later prevailing levels are protective. J. Clin. Oncol. 28, 492–503 (2010).
Gambichler, T., Bindsteiner, M., Höxtermann, S. & Kreuter, A. Serum 25-hydroxyvitamin D serum levels in a large German cohort of melanoma patients. Br. J. Dermatol. 10, 1365–2133 (2012).
Robert, C. et al. Ipilimumab plus Dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364, 2517–2526 (2011).
Chapman, P. B. et al. Improved survival with Vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011).
Carvajal, R. D. et al. KIT as a therapeutic target in metastatic melanoma. JAMA 305, 2327–2334 (2011).
Nazarian, R. et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature 468, 973–977 (2010).
Poulikakos, P. I., Zhang, C., Bollag, G., Shokat, K. M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signaling in cells with wild-type BRAF. Nature 464, 427–430 (2010).
Poulikakos, P. I. et al. RAF inhibitor resistance is mediated by dimerization of aberrantly spliced BRAF(V600E). Nature 480, 387–390 (2011).
Ziai, M. R. et al. Analysis with monoclonal antibodies of the molecular and cellular heterogeneity of human high molecular weight melanoma associated antigen. Cancer Res 47, 2474–2480 (1987).
Bartolazzi, A., Barbieri, R., Nastruzzi, C., Natali, P. G. & Gambari, R. Antitumor activity of the proteinase inhibitor Tetra-p-amidino-phenoxy-neopentane, in a nude mouse model of human melanoma. In Vivo 3, 383–388 (1989).
Bartolazzi, A., Peach, R., Aruffo, A. & Stamenkovic, I. CD44-hyaluronate interaction is implicated in the regulation of tumor growth. J. Exp. Med. 180, 53–66 (1994).
Gasbarri, A. et al. CD44s adhesive function spontaneous and PMA-inducible CD44 cleavage are regulated at post-translational level in cells of melanocytic lineage. Melanoma Res. 13, 325–337 (2003).
Giacomini, P. et al. Low prevalence of selective human leukocyte antigen (HLA)-A and HLA-B epitope losses in early-passage tumor cell lines. Cancer Res. 59, 2657–2667 (1999).
Spinella, F. et al. Endothelin-1 and endothelin-3 promote invasive behavior via hypoxia-inducible factor-1alpha in human melanoma cells. Cancer Res. 67, 1725–1734 (2007).
Miyamoto, K. et al. Structural organization of the human vitamin D receptor chromosomal gene and its promoter. Mol. Endocrinol. 11, 1165–1179 (1997).
Whitfield, G. K. et al. Functionally relevant polymorphisms in the human nuclear vitamin D receptor gene. Mol. Cell. Endocrinol. 177, 145–159 (2001).
Gross, C. et al. The vitamin D receptor gene start codon polymorphism: a functional analysis of FokI variants. J. Bone Miner. Res. 13, 1691–1699 (1998).
Hustmyer, F. G., Deluca, H. F. & Peacock, M. Apa1, Bsm1, EcoRV and Taq1 polymorphisms at the human vitamin D receptor gene locus in Caucasians, Blacks and Asians. Hum. Mo.l Genet. 2, 487 (1993).
Uitterlinden, A. G., Fang, Y., Van Meurs, J. B., Pols, H. A. & Van Leeuwen, J. P. Genetics and biology of vitamin D receptor polymorphisms. Gene 338, 143–156 (2004).
Bartolazzi, A. et al. Expression of alpha 3 beta 1 integrin receptor and its ligands in human lung tumors, Int. J. Cancer 64, 248–252 (1995).
Cecchinelli, B. et al. Repression of the antiapoptotic molecule galectin-3 by homeodomain-interacting protein kinase 2-activated p53 is required for p53-induced apoptosis. Mol. Cell. Biol. 26, 4746–4757 (2006).
Martegani, M. P., Del Prete, F., Gasbarri, A., Natali, P. G. & Bartolazzi, A. Structural variability of CD44v molecules and reliability of immunodetection of CD44 isoforms using mAbs specific for CD44 variant exon products. Am. J. Pathol. 154, 291–300 (1999).
Acknowledgements
This study was partially supported by A.I.R.C. (The Italian Association for Cancer Research). L.F. is a research fellow from Abbott. We thank Dr. Francesco Fiorentino (Laboratory Genoma, Rome, Italy) for the analysis of VDR polymorphisms. We thank *S.A.M.W.G. (Sant’Andrea Melanoma Working Group) for the interesting multidisciplinary discussion.
Author information
Authors and Affiliations
Contributions
A.B.: Study concept, study coordinator and supervisor of the experimental activity. M.C., L.F., A.N.: Acquisition of clinical data and melanoma patient management. A.B., L.F., A.N., E.L.: in vivo experiments, melanoma xenografts. L.S., A.U., E.L., L.L.: In vitro experiments, molecular biology, cell culture, data analysis. B.B.: Cell cycle and FACS analysis, interpretation of the results. A.B., M.G.: Histopathology and immunohistochemistry. D.P.: vitamin D expert, support for in vitro and in vivo experiments. A.B.: Drafting of the manuscript. S.S., M.C., A.B.: Critical revision of the manuscript for intellectual content. L.S., M.C., L.F., A.U., A.N., M.G., E.L., L.L., B.B., D.P., S.S. and A.B.: Analysis and interpretation of the results. Final approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Spath, L., Ulivieri, A., Lavra, L. et al. Antiproliferative Effects of 1α-OH-vitD3 in Malignant Melanoma: Potential Therapeutic implications. Sci Rep 7, 40370 (2017). https://doi.org/10.1038/srep40370
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep40370
This article is cited by
-
Systematic vitamin D supplementation is associated with improved outcomes and reduced thyroid adverse events in patients with cancer treated with immune checkpoint inhibitors: results from the prospective PROVIDENCE study
Cancer Immunology, Immunotherapy (2023)
-
Gene expression of programmed cell death ligand-1 (PDL-1) and vitamin D receptor (VDR) with the serum vitamin D3 in lung cancer
The Egyptian Journal of Bronchology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.