Abstract
A range of chemistries were explored for the efficient covalent conjugation of DNA to poly(N-isopropylacrylamide) (poly(NIPAM)) in organic solvents. Amide coupling and thiol–ene Michael addition were found to be ineffective for the synthesis of the desired products. However, the inverse electron-demand Diels–Alder (DAinv) reaction between tetrazine (Tz) and norbornene (Nb) was found to give DNA–polymer conjugates in good yields (up to 40%) in organic solvents (N,N-dimethylformamide, N,N-dimethylacetamide and N-methyl-2-pyrrolidone), and without the need for a catalyst. Methods for the synthesis of Tz-and Nb- functionalised DNA were developed, along with a post-polymerisation functionalisation strategy for the production of Tz-functionalised polymers.
Similar content being viewed by others
Introduction
Since the introduction by Seeman, Rothemund and others of the concept of DNA origami, the use of nucleic acids in nanoscale materials has blossomed1,2. DNA’s unique ability to form perfectly-controlled higher order structures makes it very appealing to materials scientists. Highly complex objects can now be fabricated on the nanoscale, including a lockable DNA box, structures with three dimensional curvature, tetrahedra, prisms and cubes3,4,5,6,7. It is difficult to envisage how these structures could have been formed by any other currently available technology.
The utility of DNA can be further enhanced by direct conjugation to synthetic polymers, which bring an added dimension in terms of their solvophilicity, functionality and potential to respond to external stimuli. For example, Mirkin et al. attached poly(styrene) (PS) to a short segment of single stranded DNA (ssDNA)8. Upon resuspension in water the PS-ssDNA amphiphile assembled into well-defined micelles. The ssDNA located in the corona of the micelle was then used as a functional handle - in this case, hybrid materials were created when the micelles were mixed with gold nanoparticles bearing the complementary DNA strand through the formation of a DNA duplex. Herrmann et al. used a similar approach to form poly(propylene oxide) (PPO)-ssDNA micelles, and these were used to perform DNA-templated synthesis9, synergistically deliver anti-cancer therapeutics10, and to create structures whose morphology could be tuned by the addition of different ssDNA strands11. Gianneschi et al. also exploited the use of DNA hybridisation in combination with enzymatic tools to create ‘programmable shape-shifting micelles’12. The elegant work of Sleiman et al. further demonstrates the unique properties that DNA-polymer conjugates can possess. They used DNA to control the precise placement of polymer domains within hybrid structures, and demonstrated the ability of polymers conjugated to DNA to form discrete nanocube structures, which predictably changed the polymer’s self-assembly behaviour, as well as introducing a new driving force for the assembly of DNA nanostructures in the form of hydrophobic interactions13,14,15,16. Our own group also made the first report of a DNA-polymer conjugate in which the DNA was used to form part of a discrete nanostructure, in this case a DNA tetrahedron17. The polymer used here was temperature-responsive, leading to the formation of dynamic higher order DNA–polymer structures in solution.
All of this work demonstrates the potential of DNA-polymer conjugates to provide access to highly functional materials. However, a significant barrier to greater uptake of the technology is the inefficiency with which DNA–polymer conjugates are produced. Where yields are reported in the literature they are typically below 20%, and conjugation with more hydrophobic polymers rarely exceeds 10% yield. This is problematic given the high cost of functional DNA as a starting material. Many reported syntheses also rely on the use of a solid support8,9,10,18,19,20, which usually implies the use of a DNA synthesizer, an expensive piece of equipment to which many polymer synthesis groups do not have access. It would be ideal therefore to find a method for creating DNA–polymer conjugates meeting the following criteria: i) Highly efficient; ii) Proceeds in a range of common organic solvents (to facilitate conjugation of more hydrophobic polymers); iii) Uses DNA starting materials that are commercially available and/or straightforward to produce; iv) Works in solution without the need for a solid support.
We previously reported that correct catalyst selection could make the copper-catalysed azide-alkyne cycloaddition (CuAAC) reaction a highly efficient chemistry for DNA–polymer conjugation in organic solvents17. We therefore set out to explore whether other macromolecular coupling strategies could be applied to DNA–polymer conjugation. In this work, the use of amide coupling chemistry, the thiol–ene reaction and the inverse electron demand Diels–Alder (DAinv) reaction were surveyed as potential routes to DNA–polymer conjugates. All of these chemistries have previously been used successfully for macromolecular coupling in solution in the absence of a solid support. For example, amide coupling has been successfully used for the synthesis of DNA–polymer conjugates in water, with yields of up to 90%21,22,23,24,25,26. We could find only one reported yield for the use of this reaction to produce conjugates in organic solution (DMSO in this case), which was tantalisingly high at 75%27. The use of the thiol Michael addition for synthesis of diblock copolymers of poly(N-isopropylacrylamide) (Poly(NIPAM))and PS was reported by Sumerlin et al. and the use of this reaction in polymer chemistry has been extensively reviewed28,29. Kataoka et al. reported DNA conjugates with poly(ethylene glycol) (PEG) in yields up to 89% in water, while Herrmann et al. synthesised DNA–PS conjugates with yields of around 10%30,31,32,33,34. Hansell et al. used the DAinv reaction between tetrazine (Tz) and norbornene (Nb) to couple polymers synthesised via different routes with high efficiency under ambient conditions35. While these examples illustrate that these chemistries hold promise, to our knowledge no systematic study exists of their applicability to the synthesis of DNA–polymer conjugates.
Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerisation was used for this work as it provides good control over molecular weight for a wide range of monomers36. Poly(NIPAM) was chosen as a model polymer as it is organic- and water-soluble but also exhibits temperature-responsive behaviour, a potentially useful characteristic for nanotechnology applications37.
Results and Discussion
The properties of the polymers used in these studies are given in Table 1. A summary of the coupling conditions tested is presented in Table 2. Structures of small molecules are given in Fig. 1.
Amide Coupling
Since the thiocarbonyl thio groups present in RAFT chain transfer agents (CTAs) degrade in the presence of amines, it has always been difficult to incorporate this group into polymers synthesised using the RAFT technique38. By contrast, carboxylic acid groups are stable under RAFT polymerisation conditions and can be incorporated into the CTA. 2-(Dodecylthiocarbonothioylthio)-2-methylpropionic acid (DDMAT, 1) is a widely used CTA, which has been shown to control the polymerisation of a large variety of monomers, and which contains a free carboxylic acid group in its structure (Fig. 1). It was envisaged that polymers produced using this CTA could be coupled to amine-functionalised DNA.
Poly(NIPAM) of varying molecular weights was thus synthesised using RAFT polymerisation, with 1 as the CTA. As outlined in Table 1, good control over molecular weight was achieved, with all polymers having a low dispersity (i.e. below 1.2). It was thought that the trithiocarbonate end group might interfere with the DNA coupling reaction, so, using a previously reported method39, this group was substituted with a hydrogen atom (Supplementary Figure S1), effectively removing all functionality from this end of the polymer. Successful end group removal was confirmed by comparing the SEC UV traces at 309 nm (characteristic of the trithiocarbonate group) before and after the reaction (Supplementary Figure S1). 1H NMR spectroscopic analysis in d6-DMSO confirmed that the carboxylic acid group survived the end group removal process (Supplementary Figure S2).
With the carboxylic acid-functionalised polymers in hand, coupling of amine-functionalised DNA (s0-NH2) was attempted. The DNA strand s0-NH2 was designed to have no secondary structure at room temperature and was 22 nucleotides in length (see Supplementary Figure S3 for sequence).
Since the primary aim of this work was to find an accessible way of conjugating polymers to DNA, it was decided to work at DNA concentrations not above 10 μM. This meant that reactions could be carried out on a practical scale (i.e. 10 μL or above) without using too much material – an important consideration given the high cost of functional DNA. Coupling to the carboxylic acid-functionalised poly(NIPAM) (P1a) synthesised above was attempted using a variety of solvents and coupling agents as summarised in Table 2.
The coupling agents and polymer (see Supplementary Figure S4 for structures) were added in a 10- to 100-fold excess relative to the amine group and the reaction left overnight to proceed. Each reaction mixture was then diluted with 5× glycerol loading buffer and water, and analysed by 15% native poly(acrylamide) gel electrophoresis (PAGE).
Tables S1–3 give the details of all the coupling reactions attempted. No product was observed by PAGE when EDCI, DCC, PyBOP or PyBroP were used as the coupling agent (Supplementary Figures S5-6). However, when HBTU or HATU were used (popular coupling reagents often recommended for ‘difficult’ coupling reactions)40 a slow-moving band attributed to the polymer–DNA conjugate was observed when DMF was used as the reaction solvent (Supplementary Figure S6). Analysis of the PAGE results using densitometry (Supplementary Figure S7) gave an approximate yield of the conjugate of 25%. The product bands were excised and MS analysis (ESI and MALDI-ToF) attempted, however it did not prove possible to obtain mass information about the extracted products. Instead, PAGE control experiments were performed to eliminate the possibility that these bands were caused by other species such as degradation products, dimers, or non-specific association of s0 with poly(NIPAM) (Supplementary Figure S8). A large number of previous reports also assign a broad, slow-moving band to a conjugation product8,10,11,12,13,14,15,16,17,21,22,23,24,25,26,27,30,31,32,33,34.
Experiments were also carried out to determine whether the degree of polymerisation (DP) – and therefore the molecular weight – of the polymer used had any effect on the efficiency of the coupling reaction. To this end, the synthesis of conjugates containing poly(NIPAM) with DPs of 97 and 196 (P1b and P1c) was attempted using the conditions identified above, with HATU or HBTU as the coupling agent and DMF as the solvent. However, in no cases were the conjugates observed (Supplementary Figure S9). It also proved very difficult to repeat the synthesis of the lower DP conjugate, despite increasing the purity of the solvent and DNA. The best yield obtained in the repeat experiments was 5% (as assessed by PAGE densitometry), but only for the lower DP polymer P1a.
Amide Coupling Using Pre-Activated Acids
Recently, pentafluorophenyl (PFP) esters have been used for the functionalisation of polymers produced by RAFT polymerisation41,42,43,44. The more established NHS esters have also been shown to be very useful in polymer functionalisation45. Previous work in our group has shown that pre-activated species such as these may be more effective for modification of s0-NH2 with small molecules than generating the activated ester in situ. It is hypothesised that this is because the formation of the activated ester results in the production of by-products, which later inhibit the attack of the amine and/or cause its degradation. It was therefore reasoned that conjugation of s0-NH2 to polymers containing a pre-activated acid group may be more efficient than the coupling agent-mediated process described above.
Two activated ester-functionalised CTAs (2 and 3) were thus synthesised. Both were used to polymerise NIPAM with good control over molecular weight and dispersity (see Table 1, P2-3). The presence of the PFP group was confirmed by 19F NMR spectroscopy by observing the appearance of broad signals at −152.9, −158.0 and −162.3 ppm attributable to the PFP group attached to the polymer.
By introducing an external standard with both fluorine and hydrogen NMR peaks (in this case trifluorotoluene), it was possible to estimate the degree of PFP functionalisation – around 80-95% (Supplementary Figure S10). The presence of the NHS group was confirmed by 1H NMR spectroscopy by comparing the integral of the signal at 2.85 ppm (attributable to the two CH2 groups on the succinimide group) to that of the signal at 3.30 ppm (due to the CH2 group adjacent to the trithiocarbonate group at the opposite end of the polymer). To prevent unwanted side reactions, the trithiocarbonate groups were removed from the termini of P2 and P3 using AIBN and LPO (Supplementary Figures S11–14)46.
To confirm the reactivity of the PFP group at the polymer chain end, a study using a small molecule amine was undertaken. P3 was dissolved in one of three NMR solvents − d7-DMF, d6-DMSO or d8-THF – and a small excess of benzylamine was added. The reactions were left overnight at room temperature. 1H NMR analysis showed the appearance of a new peak around 5 ppm (the exact value was solvent-dependent) due to the CH2 group adjacent to the newly-formed amide group, and 19F NMR showed the complete absence of any PFP ester peaks, concomitant with the appearance of new peaks due to the pentafluorophenol formed during the reaction (Supplementary Figure S15). It was therefore concluded that the PFP end group was present and reactive under mild conditions towards primary amines.
Having confirmed that the activated esters remained intact following end group removal, coupling to s0-NH2 was then attempted under various reaction conditions (summarised in Table 2, detailed conditions in Supplementary Table S4). However, under none of these conditions was the expected product observed. It is clear that, under these conditions, neither of the activated esters used were reactive enough for the DNA conjugate to form. Instead, hydrolysis and/or degradation occurred before the attack of the amine. Given the low yields and irreproducible nature of the conjugation reactions, it was concluded that amide coupling was not effective for the production of DNA-polymer conjugates in organic solvents.
Thiol–Ene Michael Addition
DNA–polymer conjugation was previously reported using the thiol–maleimide32 and thiol–acrylate31 Michael addition reactions, but only ever in an aqueous environment. Yields were good (around 90% in the case of the maleimide reaction) but required a relatively high DNA concentration of 0.1 mM, and the use of water as the reaction solvent precluded the conjugation of hydrophobic polymers. Given that polymer–polymer coupling in organic solvents has been achieved using thiol Michael addition chemistry, it was reasoned that an exploration of reaction conditions could yield some interesting results in the area of DNA–polymer conjugation. DNA strands functionalised with a methacrylamide, acrylamide or maleimide group were therefore targeted in the following work, and tested for their reaction with thiol-terminated polymers.
Thiol-terminated polymers were synthesised by stirring P1a-c with sodium borohydride and purified by dialysis. Successful removal of the trithiocarbonate end group was confirmed by SEC (Supplementary Figure S16). Some disulfide coupling was observed, but the presence of the free thiol was confirmed using Ellman’s assay (see Table 1 for a summary of the thiol contents)47,48. DNA functionalised with a methacrylamide group (Supplementary Figure S17) was commercially available, so the s0 sequence was purchased with this modification at the 5′ end (s0-MAAm). Conjugation to thiol-terminated P4a-c was then attempted. However, despite testing many different catalyst and solvent combinations (Table 2 and Tables S5-8), negligible product was formed (Figs S18-20).
It was thought that formation of the thiol group should be attempted in situ, so poly(NIPAM) containing a terminal dithioester group (P5, synthesised using alkyne-containing CTA 4) was mixed with s0-MAAm in the presence of hexylamine and TEA, which should have cleaved the dithioester to release a thiol – these reagents also served to catalyse the thiol Michael addition reaction itself. Again, several solvents were tested as outlined in Table 2. However, no product was observed.
Having achieved only very low yields using the methacrylamide-functionalised DNA, the use of the more reactive acrylamide group was explored. Acrylamide-functionalised DNA was not commercially available, so it was synthesised from s0-NH2 DNA and acrylic acid using EDCI and HOBt as coupling agents. Purification of the reaction mixture by HPLC (Supplementary Figure S21) afforded the product, which was analysed by LC-MS and had the expected mass. Identical polymer couplings were attempted, but no product was observed in any case. It was decided that a different type of ene compound should be used, so maleimide was investigated as a potentially more active alternative to (meth)acrylamide.
Maleimide-functionalised DNA (s0-Mal) was not commercially available, so the desired product was synthesised using a bifunctional adapter, 5. The product was isolated via HPLC and LC-MS confirmed the expected mass (Supplementary Figure S22). Polymer conjugation reactions were first attempted under aqueous conditions (Supplementary Table S9) and broad, low-mobility bands were observed (Supplementary Figure S23), with estimated yields of around 50%.
Having successfully formed the DNA–polymer conjugate in an aqueous environment, the use of organic solvent systems was explored. Five different organic solvents were tested, both with and without an auxiliary base, as summarised in Table 2 (detailed conditions in Supplementary Table S10). No product was observed for any of the solvents trialled (Supplementary Figure S24) and HPLC analysis confirmed that degradation of the maleimide group was taking place (Supplementary Figure S25).
Having attempted DNA–polymer conjugation using methacrylamide-, acrylamide- and maleimide-functionalised DNA in a number of organic solvents and observed only very slight evidence of the formation of a product, it was concluded that the thiol Michael addition reaction was not going to provide a successful route to the desired conjugates.
Tetrazine–Norbornene Coupling
Recent work in our group has revealed that the DAinv reaction between Tz and Nb is efficient for the conjugation of macromolecules in solution35. Nb-functionalised DNA (s0-Nb) was synthesised using an acid-containing Nb and s0-NH2 under standard EDCI/HOBt coupling conditions. After one hour, HPLC analysis indicated the formation of two products in nearly quantitative yield (Supplementary Figure S26-27), which were both found to have the same mass of 6 996 Da, which was the expected mass of s0-Nb; it was therefore concluded that the two products corresponded to the endo- and exo-Nb adducts (the Nb acid starting material was a mixture of the two isomers).
The reactivity of the Nb group on the DNA was tested by mixing each of the s0-Nb isomers with an alcohol-functionalised Tz (6) under conditions analogous to those to be used for the DNA–polymer conjugation. Analysis of the DNA by HPLC before and after addition of 6 revealed a clear peak shift for both isomers (Supplementary Figure S28), indicating that both retained an intact Nb group and that this group was reactive towards Tz. UV-vis spectroscopy also confirmed that the reaction had taken place (Supplementary Figure S29).
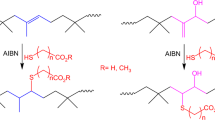
The Tz group is not stable towards radical polymerisation conditions as it reacts with monomers bearing vinyl functionality49. Incorporation of this group into an appropriate CTA was therefore not a viable option for the production of Tz-functionalised polymers by RAFT polymerisation. The alcohol-containing Tz, 6, was therefore used in the post-polymerisation modification of PFP-terminated poly(NIPAM), as above. Functionalisation of P3 was found not to proceed, possibly because the two methyl groups α to the PFP ester were hindering the attack of the alcohol – PFP-methacrylate has been observed to react with alcohols much more slowly (if at all) than the acrylate analogue50. An alternative PFP-containing CTA, 851, was therefore synthesised wherein the PFP group was held at the end of an ethyl linker, minimising steric blocking. Using pentafluorophenyl trifluoroacetate as the source of the PFP group, the yield was dramatically improved over the literature preparation (80% vs. 20%).
Next, the CTA 8 was used to control the RAFT polymerisation of NIPAM. Good control was achieved over molecular weight and dispersity (P6, see Table 1). 1H and 19F NMR analyses confirmed the presence of the end groups in the product (Figs S30–31).
Alcohol 6 was then mixed with P6 and DMAP and stirred for twenty-four hours in anhydrous THF under an atmosphere of nitrogen. The polymer was then separated from the excess alcohol by preparatory SEC in DMF. Removal of the solvent under high vacuum yielded a pink solid (P7) which was studied by UV-vis spectroscopy and found to have the same peaks as the starting material, suggesting that the Tz group had been incorporated successfully (Supplementary Figure S32).1H NMR spectroscopy also showed the presence of new peaks due to the Tz group and the CH2 group adjacent to the newly-formed ester (Supplementary Figure S33), confirming the incorporation of the Tz group in approximately 50% yield. The polymer was also analyzed by DMF SEC with an in-line UV-vis detector set to 540 nm (the wavelength of one of the characteristic peaks of the Tz group). The results (Fig. 2) showed both that the molecular weight and dispersity of the polymer remained unchanged, and that the Tz group had been successfully incorporated at the polymer chain end, by the appearance of a peak at 540 nm.
Further confirmation was provided by running THF SEC analysis using a photodiode array (PDA) detector. This collected a full UV-vis spectrum for every retention time point. The full Tz peak at 540 nm was observed to elute at the same time as the main polymer peak (Supplementary Figure S34). Importantly, both 1H NMR spectroscopy and SEC indicated that no small molecule Tz was present in the sample.
With the Tz-functionalised polymer (P7) in hand, coupling to s0-Nb was attempted under the conditions summarised in Table 2. The reaction mixtures were analysed by 15% native PAGE (Fig. 3), which showed that the conjugate was formed in moderate yield in water by the clear appearance of a low-mobility band attributed to the product. When DMF or DMAc was used as the reaction solvent, yields were below 10%, and in NMP and DMSO no reaction was observed. It should be noted at this point that in most cases PAGE analysis showed the retention of some DNA-containing material in the wells. Control experiments confirmed that this was due to the high concentration of free polymer in the loaded samples and did not effect the yield estimates – the higher concentration images are presented here as the product bands are easier to discern.
15% native PAGE analysis of the conjugation of P7 to s0-Nb.
The DNA-polymer conjugate was visible as a slow-migrating band (blue box) when water, DMF or DMAc were used as the reaction solvent (lanes i-iii), but not with NMP (lane iv) or DMSO (lane v). Left: Densitometry plot of lane i showing the band assigned to the DNA-polymer conjugate - the yield was calculated by comparing the areas under the conjugate and free s0-Nb peaks.
In an effort to improve the yields, the positions of the Tz and Nb groups were switched. In order to obtain Tz-functionalised DNA, it was first necessary to synthesise an adapter containing the Tz group. This was obtained by activating the Tz-acid, 9, with N-hydroxysuccinimide (NHS) to give Tz-NHS, 10. 1H (Supplementary Figure S35) and 13C NMR spectroscopy both showed the expected peaks, including the four protons from the NHS group.
Next, 10 was reacted with s0-NH2 in 1:1 DMF/PBS solution at 40 °C, with a DNA concentration of 100 μM and a one thousand-fold excess of the small molecule. Purification of the reaction mixture yielded a peak which exhibited the characteristic Tz UV-vis absorbance at around 330 nm (Supplementary Figure S36), and had the expected mass (Supplementary Figure S37).
To confirm the reactivity of s0-Tz, a small molecule test reaction was carried out in both water and DMF. Commercially available 5-norbornene-2-exo,3-exo-dimethanol was mixed with s0-Tz at a concentration of 50 μM and the reaction followed by HPLC. A clear peak shift was observed in both solvents (Supplementary Figure S38), confirming that the Tz retained its reactivity once conjugated to the DNA strand. The yield of the reaction was estimated by comparison of the areas under the peaks due to the Tz-Nb coupling product and unreacted DNA and found to be approximately 70% and 40% in water and DMF respectively. The difference in yield can possibly be attributed to degradation of the Tz group by free amines present in the DMF used.
Next, DNA–polymer conjugation was attempted using poly(NIPAM) end-capped with a Nb group (P8)52. After reacting with 10 μM s0-Tz DNA overnight at room temperature in DMF, DMAc or NMP (see Table 2), 15% native PAGE analysis revealed that the conjugate had been formed in up to 50% yield (by densitometry) – see Fig. 4. It should be noted that a significant shift in the migration distance of the starting material band was observed in these gels. This was thought to be due to degradation of the Tz group under the reaction conditions, and inspection of the HPLC analysis in Supplementary Figure S38 confirmed that this was likely the case: as well as the product peak a large number of smaller peaks appeared, which were attributed to degradation products. The significant difference in yield compared to the s0-Nb/P7 reaction was attributed to the different structures of the Tz groups used, in line with recent reports, which show that in general electron-donating groups (such as the methyl group attached at the 6 position to the Tz in P7) increase the energy of the LUMO, making the reaction less favorable, while electron-withdrawing groups (such as the pyridyl groups of s0-Tz) lower the LUMO’s energy, increasing the favorability of the reaction53,54. The Tz-Nb method represented by far the most efficient DNA–polymer conjugation chemistry tested and is a novel, catalyst-free approach for the production of DNA–polymer conjugates in good yield in organic solution. Future studies will focus on elucidating the relationship between Tz structure and the efficiency of the reaction in different solvent systems in order to optimize yields.
15% native PAGE analysis showing the formation of a DNA-polymer conjugate from P8 and s0-Tz using the Tz-Nb DAinv reaction.
Left: Densitometry plot of lane ix showing the band assigned to the DNA-polymer conjugate - the yield was calculated by comparing the areas under the conjugate and free s0-Tz peaks. The solvents used were DMF (lanes i-iii), DMAc (lanes iv-vi) and NMP (lanes vii-ix), with either 1 (lanes i, iv and vii), 10 (lanes ii, v and viii) or 100 (lanes iii, vi and ix) equivalents of the polymer.
Conclusions
This work has demonstrated that the conjugation of polymers to DNA strands in organic solvents is not straightforward. At moderate DNA concentrations and in free solution both the thiol–ene reaction and the amide coupling route failed to yield significant amounts of the desired conjugate when organic solvents were used, despite their efficacy in water. While it was not possible to produce conjugates using these reactions a number of novel DNA and polymer modifications were developed which it is hoped will be of use in other areas of materials science, including acrylamide-functionalised DNA. In contrast, it was shown that the DAinv reaction between a norbornene and a tetrazine can lead to the production of DNA–polymer conjugates in organic solution, at practical (i.e. reasonably low) DNA concentrations and without the need for a solid support. With further development of the precise structures of the norbornene and tetrazine derivatives used this technique has the potential to become a mild and high-yielding route to DNA–polymer conjugates.
Methods
For materials and methods information and comprehensive experimental details please see the Supporting Information. Syntheses of key compounds are presented below.
Synthesis of PFP-DDMAT, 2
PFP-DDMAT, 2, was synthesised as follows. 1 (0.500 g, 1.37 mmol) was added to an oven-dried schlenk flask, which was then evacuated and refilled with nitrogen three times. Anhydrous DMF (7.5 mL) was added via syringe and the flask cooled to 0 °C with an ice bath. DIPEA (354 μL, 2.74 mmol) was then added via syringe, followed by dropwise addition of pentafluorophenyl trifluoroacetate (283 μL, 1.65 mmol). After one hour stirring at 0 °C, the flask was opened to the air and diethyl ether (30 mL) was added, followed by a 1 M solution of HCl (30 mL). The organic layer was collected and washed with water (2 × 30 mL) and brine (30 mL). The solvent was removed in vacuo to give a yellow oily residue, which was then purified by silica gel column chromatography, eluting with a mixture of ethyl acetate and pet. ether 40-60 (gradient from 5-10% ethyl acetate). The fractions containing the product (Rf = 0.81) were combined and the solvent removed in vacuo to yield CTA 2 as a yellow oil (0.686 g, 94%). 1H NMR (400 MHz, CDCl3) δ 3.31 (t, J = 7 Hz, 2H, SCH2), 1.86 (s, 6H, C(CH3)2), 1.69 (quint, J = 7 Hz, 2H, SCH2CH2), 1.40 (m, 2H, CH2CH3), 1.26 (br s, 16H, SCH2CH2(CH2)8), 0.88 (t, J = 7 Hz, 3H, CH2CH3) ppm. 13C NMR (150 MHz, CDCl3) δ 219.9 (C = S), 169.6 (C = O), 142.1 (t), 140.4 (t), 138.7 (t), 137.0 (t) (PFP Cs), 55.4 (C(CH3)2), 37.2 (SCH2), 31.9, 29.6, 29.5, 29.4, 29.3, 29.1, 29.0, 28.9, 27.8, 25.4 (C(CH3)2), 22.7 (CH2CH3), 14.1 (S(CH2)11CH3) ppm. 19F NMR (375 MHz, CDCl3) −151.5 (d, 2F, ortho F), −157.7 (t, 2F, para F), −162.3 (t, 2F, meta F) ppm. IR (νmax/cm−1): 2925, 2854, 1779, 1517, 1079, 992, 815. ESI HR MS calcd. for C23H31F5O2S3 [M + H]+ 531.1486 Da; observed 531.1480 Da.
Synthesis of NHS-DDMAT, 3
NHS-DDMAT, 3, was synthesised as follows45. 1 (0.500 g, 1.37 mmol), NHS (0.158 g, 1.37 mmol) and DCC (0.283 g, 1.37 mmol) were dissolved in dichloromethane and the mixture stirred for 48 hours. The cloudy mixture was filtered through a 0.45 μm PTFE syringe filter and the retentate washed with CH2Cl2 (5 mL). This filtration process was then repeated. The solvent was removed in vacuo and the residue dissolved in a small amount of ethyl acetate. The solution was purified by silica gel column chromatography, eluting with a mixture of ethyl acetate and pet. ether 40–60 (1:1). The product fractions (Rf = 0.53) were collected and the solvent removed in vacuo to afford 3 as a yellow solid (0.502 g, 79%). 1H NMR (400 MHz, CDCl3) δ 3.30 (t, J = 7 Hz, 2H, SCH2), 2.81 (br s, 4H, CH2(C = O)N), 1.87 (s, 6H, C(CH3)2), 1.68 (quint, J = 7 Hz, 2H, SCH2CH2), 1.38 (m, 2H, CH2CH3), 1.25 (br s, 16H, SCH2CH2(CH2)8), 0.88 (t, J = 7 Hz, 3H, CH2CH3) ppm. 13C NMR (150 MHz, CDCl3) δ 218.8 (C = S), 169.1 (C = OO), 168.7 (NHS C = O), 54.3 (C(CH3)2), 37.2 (SCH2), 31.9 (SCH2CH2), 29.6, 29.5, 29.4, 29.3, 29.1, 29.0, 27.8, 25.4 (NHS CH2), 25.3 (C(CH3)2), 22.7 (CH2CH3), 14.1 (S(CH2)11CH3) ppm. IR (νmax/cm−1): 2917, 2848, 1777, 1736, 1203, 1074, 811. ESI HR MS calcd. for C21H35NO4S3 [M + Na]+ 484.1626 Da; observed 484.1619 Da.
Synthesis of P2 and P3 using CTAs 2 and 3
Polymerisation of NIPAM with 3 was conducted as follows. NHS-DDMAT, 3, (0.041 g, 0.09 mmol), NIPAM (1.000 g, 8.84 mmol) and AIBN (0.002 g, 0.01 mmol) were dissolved in 1,4-dioxane (1.5 mL) and transferred to an oven-dried ampoule. The mixture was subjected to three freeze-pump-thaw cycles and sealed under an atmosphere of nitrogen. It was then placed in an oil bath preheated to 65 °C. After 2 hours the ampoule was removed and the reaction quenched by opening it to the air and cooling with liquid nitrogen. The solution was poured into pet. ether 40-60 (80 mL) cooled in an ice bath and the precipitant collected by filtration. The product was then dissolved in THF (1 mL) and the process repeated 5 more times. Finally, the isolated solid was dissolved in THF (1 mL) and precipitated into diethyl ether (80 mL) cooled in an ice bath. The product was isolated by filtration, dried in vacuo and isolated as a yellow powder (0.335 g, 36%) and analysed by DMF SEC using PMMA calibration standards (Mn 9 610 Da, Đ 1.10). 1H NMR (400 MHz, CDCl3) δ 7.36–5.55 (br m, PNIPAM NH), 4.00 (br s, PNIPAM CH(CH3)2), 3.33 (br m, 2H, SCH2), 2.86 (br s, 4H, CH2(C = O)N), 2.64–0.80 (br m, PNIPAM backbone H), 0.88 (t, J = 7 Hz, 3H, S(CH2)11CH3) ppm.
PFP-DDMAT, 2, was also used to polymerise NIPAM using an identical procedure.
Synthesis of s0-AAm
Acrylamide-functionalised DNA (s0-AAm) was synthesised from amine-functionalised DNA (s0-NH2) as follows. Acrylic acid (16.7 μL, 600 mM in DMF, 10 μmol), EDCI (16.7 μL, 600 mM in DMF, 10 μmol), HOBt (16.7 μL, 600 mM in DMF, 10 μmol) and DIPEA (1.7 μL, 10 μmol) were mixed and incubated at room temperature for thirty minutes. Phosphate buffer pH 8.0 (47.0 μL) and s0-NH2 (3.2 μL, 3.17 mM in water, 10 nmol) were added and the mixture left at room temperature for 24 hours. The excess small molecules and DMF were removed by extraction with dichloromethane (3 × 200 μL). The aqueous layer was isolated and topped up to a final volume of 100 μL with water. The product was isolated by HPLC, with a yield of 20% as quantified by UV-vis spectroscopy using the known extinction coefficient of the starting material DNA at 260 nm.
Synthesis of s0-Mal using the bifunctional adapter, 5
s0-NH2 (1000 μL, 200 μM in water, 200 nmol), 5 (53.6 mg, 200 mmol) and DIPEA (35 μL, 200 mmol) were mixed in DMF (1000 μL) and the reaction shaken overnight at 40 °C. The excess small molecules were then removed using a NAP-10 Sephadex column and the collected solution concentrated in vacuo and purified by HPLC. The product was isolated as a single fraction, with an isolated yield of 63% as quantified by UV-vis spectroscopy using the known extinction coefficient of the starting material DNA at 260 nm.
Synthesis of s0-Nb
EDCI (100 μL, 300 mM in DMF) was mixed with HOBt (100 μL, 300 mM in DMF), 5-norbornene-2-carboxylic acid (100 μL, 300 mM in DMF) and PBS (150 μL) and thoroughly mixed. 75 μL of this solution was mixed with s0-NH2 (25 μL, 200 μM in water) and DIPEA (0.87 μL). After one hour shaking the flask at room temperature the mixture was purified by HPLC and the product isolated as two separate peaks. Both were analysed by MALDI-ToF MS. Expected mass 6 996.3 Da; observed 6 995.8 Da.
Perfluorophenyl 4-cyano-4-(dodecylthiocarbonothioylthio)-pentanoate, 8
Perfluorophenyl 4-cyano-4-(dodecylthiocarbonothioylthio)pentanoate (8) was synthesised as follows51. 7 (0.5 g, 1.24 mmol) was added to an oven-dried flask under nitrogen. Anhydrous DMF (9 mL) was added followed by DIPEA (431 μL, 2.48 mmol). The mixture was cooled using an ice bath and pentafluorophenyl trifluoroacetate (255 μL, 1.49 mmol) was added dropwise with vigorous stirring. The reaction was allowed to proceed for one hour, then diethyl ether (40 mL) was added followed by 1 M HCl (40 mL). The organic layer was separated and washed with water (2 × 40 mL) and brine (40 mL). The solvent was then removed in vacuo and the residue purified by silica gel column chromatography, eluting with a mixture of hexane and diethyl ether (10:1). The product (Rf 0.21) was isolated as an orange viscous liquid (0.563 g, 80%). 1H NMR (300 MHz, CDCl3) δ 3.34 (t, J = 7 Hz, 2H, SCH2), 3.01 (m, 2H, CH2CO2PFP), 2.60 (m, 2H, CH2CH2CO2PFP), 1.93 (s, 3H, SCCH3), 1.70 (quint, J = 7 Hz, 2H, SCH2CH2), 1.40 (m, 2H, SCH2CH2CH2), 1.26 (br s, 16H, S(CH2)3(CH2)8CH3), 0.88 (t, J = 7 Hz, 3H, S(CH2)11CH3) ppm. IR (νmax/cm−1) 2918, 2850, 1797, 1517, 1094, 990, 803. (These values compare well with the literature values above, but are given because a significantly different method was used for the synthesis).
2,5-dioxopyrrolidin-1-yl 5-oxo-5-(6-(6-(pyridin-2-yl)-1,2,4,5-tetrazin-3-yl)pyridin-3-ylamino)pentanoate, 10
The tetrazine DNA adaptor was synthesised as follows. Tetrazine 9 (0.100 g, 0.27 mmol) and NHS (0.032 g, 0.27 mmol) were dissolved in DMF (4 mL) and the solution bubbled with nitrogen for 30 minutes. The mixture was then cooled using an ice bath and DCC (0.057 g, 0.27 mmol) dissolved in DMF (1 mL) was added via syringe. After 30 minutes stirring the ice bath was removed and the reaction stirred under nitrogen for 16 hours. The reaction mixture was adsorbed onto silica which had been previously treated with EtSiCl3. EtSiCl3-treated silica gel column chromatography was then performed, eluting first with DMF, then acetone. The pure fractions were collected and combined, then dried in vacuo to afford the product 10 as a deep red powder (0.048 g, 38%). 1H NMR (400 MHz, d6-DMSO) δ 10.67 (brs, 1H, NHC = O), 9.10 (d, J = 2 Hz, 1H, Tz H1), 8.97 (d, J = 4 Hz, 1H, Tz H7), 8.66 (d, J = 9 Hz, 1H, Tz H4), 8.63 (d, J = 8 Hz, 1H, Tz H3), 8.47 (dd, J = 2, 9 Hz, 1H Tz H2), 8.19 (td, J = 1, 4 Hz, 1H, Tz H6), 7.76 (dd, J = 5, 7 Hz, 1H, Tz H5), 2.87 (m, 6H, CH2(C = O)N and CH2(C = O)NH), 2.63 (t, J = 7 Hz, 2H, CH2(C = O)OSu), 2.03 (quint, J = 7 Hz, 2H, CH2CH2(C = O)NH) ppm. 13C NMR (125 MHz, d6-DMSO) δ 172.5 (C = ONH), 170.7 (NHS C = O), 169.3 (C = OO), 163.5 (Tz N-C = N), 163.2 (Tz N-C = N), 151.1 (Tz C7), 150.7 (Tz C-C = N), 144.3 (Tz C-C = N), 141.8 (Tz C1), 138.9 (Tz C-NHC = O), 138.3 (Tz C5), 127.0, 126.7, 125.4, 124.7 (Tz C2/3/4/6), 35.1 (CH2C = ONH), 30.0 (CH2C = OO), 25.9 (NHS CH2), 20.2 (CH2CH2C = O) ppm. IR (νmax/cm−1): 2895, 1732, 1714, 1543, 1392, 1061. ESI HR MS calcd. for C21H18N8O5 [M + Na]+ 485.1298; observed 485.1293.
Synthesis of s0-Tz
300 mM solutions of EDCI, HOBt and 10 were prepared in DMF and then mixed in equal proportions. 100 μL of s0-NH2 (200 μM in water) were added to a 1 mL centrifuge tube and the solvent removed in vacuo. 100 μL of the EDCI/HOBt/10 mixture were added, followed by 100 μL of potassium phosphate buffer (100 mM, pH 8.0). The solution was vortexed to mix and then heated at 40 °C for four hours, after which time small molecules were removed by passing the solution through a NAP-5 sephadex column, eluting with water. The sample was concentrated in vacuo and then purified by HPLC. The product was isolated as a single peak (6%) and analysed by LC-MS. Expected mass 7 223.9 Da; observed 7 223.2 Da.
Additional Information
How to cite this article: Wilks, T. R. and O’Reilly, R. K. Efficient DNA–Polymer Coupling in Organic Solvents: A Survey of Amide Coupling, Thiol–Ene and Tetrazine–Norbornene Chemistries Applied to Conjugation of Poly(N-Isopropylacrylamide). Sci. Rep. 6, 39192; doi: 10.1038/srep39192 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Rothemund, P. W. K. Folding DNA to create nanoscale shapes and patterns. Nature 440, 297, doi: 10.1038/nature04586 (2006).
Seeman, N. C. Nucleic acid junctions and lattices. J. Theor. Biol. 99, 237, doi: 10.1016/0022-5193(82)90002-9 (1982).
Aldaye, F. A. & Sleiman, H. F. Modular access to structurally switchable 3D discrete DNA assemblies. J. Am. Chem. Soc. 129, 13376, doi: 10.1021/ja075966q (2007).
Andersen, E. S. et al. Self-assembly of a nanoscale DNA box with a controllable lid. Nature 459, 73, doi: 10.1038/nature07971 (2009).
Goodman, R. P. et al. Rapid chiral assembly of rigid DNA building blocks for molecular nanofabrication. Science 310, 1661, doi: 10.1126/science.112036 (2005).
Han, D. et al. DNA origami with complex curvatures in three-dimensional space. Science 332, 342, doi: 10.1126/science.1202998 (2011).
He, X., Dong, L., Wang, W., Lin, N. & Mi, Y. Folding single-stranded DNA to form the smallest 3D DNA triangular prism. Chem. Commun. 49, 2906, doi: 10.1039/C3CC39266J (2013).
Li, Z., Zhang, Y., Fullhart, P. & Mirkin, C. A. Reversible and chemically programmable micelle assembly with DNA block-copolymer amphiphiles. Nano Lett. 4, 1055, doi: 10.1021/nl049628o (2004).
Alemdaroglu, F. E., Ding, K., Berger, R. & Herrmann, A. DNA-templated synthesis in three dimensions: Introducing a micellar scaffold for organic reactions. Angew. Chem. Int. Ed. 45, 4206, doi: 10.1002/anie.200600524 (2006).
Alemdaroglu, F. E., Alemdaroglu, N. C., Langguth, P. & Herrmann, A. DNA block copolymer micelles - a combinatorial tool for cancer nanotechnology. Adv. Mater. 20, 899, doi: 10.1002/adma.200700866 (2008).
Ding, K., Alemdaroglu, F. E., Börsch, M., Berger, R. & Herrmann, A. Engineering the structural properties of DNA block copolymer micelles by molecular recognition. Angew. Chem. Int. Ed. 46, 1172, doi: 10.1002/anie.200603064 (2007).
Chien, M.-P., Rush, A. M., Thompson, M. P. & Gianneschi, N. C. Programmable shape-shifting micelles. Angew. Chem. Int. Ed. 49, 5076, doi: 10.1002/anie.201000265 (2010).
Carneiro, K. M. M. et al. Stimuli-responsive organization of block copolymers on DNA nanotubes. Chem. Sci. 3, 1980, doi: 10.1039/C2SC01065H (2012).
McLaughlin, C. K. et al. Three-dimensional organization of block copolymers on “DNA-minimal” scaffolds. J. Am. Chem. Soc. 134, 4280, doi: 10.1021/ja210313p (2012).
Serpell, C. J., Edwardson, T. G. W., Chidchob, P., Carneiro, K. M. M. & Sleiman, H. F. Precision polymers and 3D DNA nanostructures: Emergent assemblies from new parameter space. J. Am. Chem. Soc. 136, 15767, doi: 10.1021/ja509192n (2014).
Chidchob, P., Edwardson, T. G. W., Serpell, C. J. & Sleiman, H. F. Synergy of two assembly languages in DNA nanostructures: Self-assembly of sequence-defined polymers on DNA cages. J. Am. Chem. Soc. 138, 4416, doi: 10.1021/jacs.5b12953 (2016).
Wilks, T. R. et al. “Giant surfactants” created by the fast and efficient functionalization of a DNA tetrahedron with a temperature-responsive polymer. ACS Nano 7, 8561, doi: 10.1021/nn402642a (2013).
Ayaz, M. S. et al. Synthesis of DNA block copolymers with extended nucleic acid segments by enzymatic ligation: cut and paste large hybrid architectures Chem. Commun. 47, 2243, doi: 10.1039/C0CC04746E (2011).
Hurst, S. J. et al. Synthetically programmable DNA binding domains in aggregates of DNA-functionalized gold nanoparticles Small 5, 2156, doi: 10.1002/smll.200900568 (2009).
He, P. & He, L. Synthesis of surface-anchored DNA-polymer bioconjugates using reversible addition-fragmentation chain transfer polymerization Biomacromolecules 10, 1804, doi: 10.1021/bm9002283 (2009).
Jeong, J. H., Kim, S. W. & Park, T. G. A new antisense oligonucleotide delivery system based on self-assembled ODN-PEG hybrid conjugate micelles. J. Control. Rel. 93, 183, doi: 10.1016/j.jconrel.2003.07.002 (2003).
Jeong, J. H., Kim, S. W. & Park, T. G. Novel intracellular delivery system of antisense oligonucleotide by self-assembled hybrid micelles composed of DNA/PEG conjugate and cationic fusogenic peptide. Bioconj. Chem. 14, 473, doi: 10.1021/bc025632k (2003).
Jeong, J. H., Kim, S. H., Kim, S. W. & Park, T. G. In vivo tumor targeting of ODN-PEG-folic acid/PEI polyelectrolyte complex micelles. J. Biomater. Sci. Polym. Ed. 16, 1409, doi: 10.1163/156856205774472335 (2005).
Jeong, J. H., Kim, S. H., Kim, S. W. & Park, T. G. Polyelectrolyte complex micelles composed of c-raf antisense oligodeoxynucleotide-poly(ethylene glycol) conjugate and poly(ethylenimine): Effect of systemic administration on tumor growth. Bioconj. Chem. 16, 1034, doi: 10.1021/bc0497315 (2005).
Fong, R. B., Ding, Z., Long, C. J., Hoffman, A. S. & Stayton, P. S. Thermoprecipitation of streptavidin via oligonucleotide-mediated self-assembly with poly(N-isopropylacrylamide). Bioconj. Chem. 10, 720, doi: 10.1021/bc980151f (1999).
Costioli, M. D., Fisch, I., Garret-Flaudy, F., Hilbrig, F. & Freitag, R. DNA purification by triple-helix affinity precipitation. Biotechnol. Bioeng. 81, 535, doi: 10.1002/bit.10497 (2003).
Jeong, J. H. & Park, T. G. Novel polymer-DNA hybrid polymeric micelles composed of hydrophobic poly(D,L-lactic-co-glycolic acid) and hydrophilic oligonucleotides. Bioconj. Chem. 12, 917, doi: 10.1021/bc010052t (2001).
Li, M., De, P., Gondi, S. R. & Sumerlin, B. S. End group transformations of RAFT-generated polymers with bismaleimides: Functional telechelics and modular block copolymers. J. Polym. Sci. Polym. Chem. 46, 5093, doi: 10.1002/pola.22837 (2008).
Kempe, K., Krieg, A., Becer, C. R. & Schubert, U. S. “Clicking” on/with polymers: A rapidly expanding field for the straightforward preparation of novel macromolecular architectures. Chem. Soc. Rev. 41, 176, doi: 10.1039/C1CS15107J (2012).
Oishi, M., Sasaki, S., Nagasaki, Y. & Kataoka, K. pH-responsive oligodeoxynucleotide (ODN)-poly(ethylene glycol) conjugate through acid-labile β-thiopropionate linkage: Preparation and polyion complex micelle formation. Biomacromolecules 4, 1426, doi: 10.1021/bm034164u (2003).
Oishi, M., Nagasaki, Y., Itaka, K., Nishiyama, N. & Kataoka, K. Lactosylated poly(ethylene glycol)-siRNA conjugate through acid-labile β-thiopropionate linkage to construct pH-sensitive polyion complex micelles achieving enhanced gene silencing in hepatoma cells. J. Am. Chem. Soc. 127, 1624, doi: 10.1021/ja044941d (2005).
Oishi, M., Nagatsugi, F., Sasaki, S., Nagasaki, Y. & Kataoka, K. Smart polyion complex micelles for targeted intracellular delivery of PEGylated antisense oligonucleotides containing acid-labile linkages. ChemBioChem 6, 718, doi: 10.1002/cbic.200400334 (2005).
Safak, M., Alemdaroglu, F. E., Li, Y., Ergen, E. & Herrmann, A. Polymerase chain reaction as an efficient tool for the preparation of block copolymers. Adv. Mat. 19, 1499, doi: 10.1002/adma.200700240 (2007).
Sowwan, M. et al. Polarizability of DNA block copolymer nanoparticles observed by electrostatic force microscopy. Macromol. Rapid Commun. 31, 1242, doi: 10.1002/marc.200900904 (2010).
Hansell, C. F. et al. Additive-free clicking for polymer functionalization and coupling by tetrazine-norbornene chemistry. J. Am. Chem. Soc. 133, 13828, doi: 10.1021/ja203957h (2011).
Moad, G., Rizzardo, E. & Thang, S. H. Living radical polymerization by the RAFT process: A third update. Aust. J. Chem. 65, 985, doi: 10.1071/CH12295 (2012).
Schild, H. G. Poly(N-isopropylacrylamide): experiment, theory and application. Prog. Polym. Sci. 17, 163, doi: 10.1016/0079-6700(92)90023-r (1992).
Willcock, H. & O’Reilly, R. K. End group removal and modification of RAFT polymers. Polym. Chem. 1, 149, doi: 10.1039/B9PY00340A (2010).
Chong, Y. K., Moad, G., Rizzardo, E. & Thang, S. H. Thiocarbonylthio end group removal from RAFT-synthesized polymers by radical-induced reduction. Macromolecules 40, 4446, doi: 10.1021/ma062919u (2007).
Valeur, E. & Bradley, M. Amide bond formation: beyond the myth of coupling reagents. Chem. Soc. Rev. 38, 606, doi: 10.1039/B701677H (2009).
Gibson, M. I., Fröhlich, E. & Klok, H.-A. Postpolymerization modification of poly(pentafluorophenyl methacrylate): Synthesis of a diverse water-soluble polymer library. J. Polym. Sci. Polym. Chem. 47, 4332, doi: 10.1002/pola.23486 (2009).
Roth, P. J., Haase, M., Basché, T., Theato, P. & Zentel, R. Synthesis of heterotelechelic α, ω dye-functionalized polymer by the RAFT process and energy transfer between the end groups. Macromolecules 43, 895, doi: 10.1021/ma902391b (2009).
Singha, N. K., Gibson, M. I., Koiry, B. P., Danial, M. & Klok, H.-A. Side-chain peptide-synthetic polymer conjugates via tandem “ester-amide/thiol-ene” post-polymerization modification of poly(pentafluorophenyl methacrylate) obtained using ATRP. Biomacromolecules 12, 2908, doi: 10.1021/bm200469a (2011).
Wiss, K. T. & Theato, P. Facilitating polymer conjugation via combination of RAFT polymerization and activated ester chemistry. J. Polym. Sci. Polym. Chem. 48, 4758, doi: 10.1002/pola.24267 (2010).
Li, H., Bapat, A. P., Li, M. & Sumerlin, B. S. Protein conjugation of thermoresponsive amine-reactive polymers prepared by RAFT. Polym. Chem. 2, 323, doi: 10.1039/C0PY00178C (2011).
Chen, M., Moad, G. & Rizzardo, E. Thiocarbonylthio end group removal from RAFT-synthesized polymers by a radical-induced process. J. Polym. Sci. Polym. Chem. 47, 6704, doi: 10.1002/pola.23711 (2009).
Ellman, G. L. A colorimetric method for determining low concentrations of mercaptans. Arch. Biochem. Biophys. 74, 443, doi: 10.1016/0003-9861(58)90014-6 (1958).
Segui, F., Qiu, X.-P. & Winnik, F. M. An efficient synthesis of telechelic poly (N-isopropylacrylamides) and its application to the preparation of α, ω-dicholesteryl and α, ω-dipyrenyl polymers. J. Polym. Sci. Polym. Chem. 46, 314, doi: 10.1002/pola.22382 (2008).
Wijnen, J. W., Zavarise, S., Engberts, J. B. F. N. & Charton, M. Substituent effects on an inverse electron demand hetero Diels-Alder reaction in aqueous solution and organic solvents: Cycloaddition of substituted styrenes to di(2-pyridyl)-1,2,4,5-tetrazine. J. Org. Chem. 61, 2001, doi: 10.1021/jo9518563 (1996).
Eberhardt, M., Mruk, R., Zentel, R. & Théato, P. Synthesis of pentafluorophenyl(meth)acrylate polymers: New precursor polymers for the synthesis of multifunctional materials. Eur. Polym. J. 41, 1569, doi: 10.1016/j.eurpolymj.2005.01.025 (2005).
Pötzsch, R., Fleischmann, S., Tock, C., Komber, H. & Voit, B. I. Combining RAFT and Staudinger ligation: A potentially new synthetic tool for bioconjugate formation. Macromolecules 44, 3260, doi: 10.1021/ma2000724 (2011).
Stamenovic, M. M., Espeel, P., Camp, W. V. & Du Prez, F. E. Norbornenyl-based RAFT agents for the preparation of functional polymers via thiol-ene chemistry. Macromolecules 44, 5619, doi: 10.1021/ma200799b (2011).
Knall, A.-C. & Slugovc, C. Inverse electron demand Diels-Alder (iEDDA)-initiated conjugation: A (high) potential click chemistry scheme Chem. Soc. Rev. 42, 5131, doi: 10.1039/C3CS60049A (2013).
Foster, R. A. A. & Willis, M. C. Tandem inverse-electron-demand hetero-/retro-Diels-Alder reactions for aromatic nitrogen heterocycle synthesis Chem. Soc. Rev. 42, 63, doi: 10.1039/C2CS35316D (2013).
Acknowledgements
T.W. and R.O’R. thank the EPSRC and the University of Warwick for funding. We thank Prof. Filip du Prez for providing polymer 8.
Author information
Authors and Affiliations
Contributions
T.W. and R.O’R. conceived the experiments, T.W. conducted the experiments and analysed the results. Both authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wilks, T., O’Reilly, R. Efficient DNA–Polymer Coupling in Organic Solvents: A Survey of Amide Coupling, Thiol-Ene and Tetrazine–Norbornene Chemistries Applied to Conjugation of Poly(N-Isopropylacrylamide). Sci Rep 6, 39192 (2016). https://doi.org/10.1038/srep39192
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep39192
This article is cited by
-
Modular self-assembly of gamma-modified peptide nucleic acids in organic solvent mixtures
Nature Communications (2020)
-
Glycopolymer preparation via post-polymerization modification using N-succinimidyl monomers
Polymer Journal (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.